MAP kinase phosphatase-1 - a new player at the nexus between sarcopenia and metabolic disease
Abstract
Sarcopenia, which is defined by the loss of skeletal muscle mass, predisposes skeletal muscle to metabolic dysfunction which can precipitate metabolic disease. Similarly, overnutrition, which is a major health problem in modern society, also causes metabolic dysfunction in skeletal muscle and predisposition to metabolic disease. It is now the prevailing view that both aging and overnutrition negatively impact skeletal muscle metabolic homeostasis through deleterious effects on the mitochondria. Accordingly, interplay between the molecular pathways implicated in aging and overnutrition that induce mitochondrial dysfunction are apparent. Recent work from our laboratory has uncovered the stress-responsive mitogen-activated protein kinase (MAPK) phosphatase-1 (MKP-1) as a new player in the regulation of metabolic homeostasis in skeletal muscle and mitochondrial dysfunction caused by overnutrition. These observations raise the intriguing possibility that MKP-1 may function as a common target in the convergence between sarcopenia and overnutrition in a pathophysiological pathway that leads to a loss of skeletal muscle mitochondrial function. With the increasing aging population it will become more important to understand how MKP-1, and possibly other phosphatases, operate at the nexus between sarcopenia and metabolic disease.
Skeletal muscle is a major contributor to
resting levels of energy expenditure [1], and reduced
skeletal muscle function contributes to age-induced obesity and diabetes.
Disruption of skeletal muscle function may also contribute to insulin
resistance and diabetes caused by overnutrition and obesity [2]. There is
increasing evidence that similar molecular pathways are at work in promoting
age-related and obesity-related muscle dysfunction. These pathways seem to
converge at the level of mitochondrial dysfunction.
The overall metabolic capacity of
skeletal muscle is determined by the myofiber type of the particular muscle.
Though less abundant, oxidative/slow twitch (MHC type I, IIa) muscle fibers are
important for overall metabolic wellness. However, the most abundant myofibers
are glycolytic or fast twitch (MHC type IIx/IIB) muscle fibers which mainly
utilize glycolysis to metabolize glucose. Oxidative myofibers have increased
mitochondria, more blood flow, and are resistant to fatigue. In contrast,
glycolytic fibers have the least amount of mitochondria, and fatigue easily [2,3].
Skeletal
muscle fiber type establishment is regulated by changes in calcium flux,
nervous system inputs, and transcriptional events and is a dynamic process.
Endurance exercise increases oxidative capacity of skeletal muscle, and to this
end, human studies have shown that endurance athletes have increased amounts of
MHC type I [2]. During
periods of inactivity, fibers switch from oxidative to more glycolytic types [4].
Furthermore, an increased proportion of glycolytic fibers have been
demonstrated in the elderly population as well as diet-induced obese
populations [2,3]. However,
it is not well understood whether these changes in myofiber content are causal
to age- and overnutrition-induced obesity and insulin resistance, or whether
they are merely consequences of these events.
One
hallmark of the aging process is the progressive atrophy of skeletal muscle
leading to weakness and frailness, a condition termed sarcopenia, which is also
associated with increased obesity in elderly [5]. It is
thought that the occurrence of sarcopenia in the elderly is
related to a switch in myofiber composition, a loss of muscle stem cell number,
and decreased mitochondrial function [6,7]. To this
end, mitochondrial number and function decreases with age in human subjects [8,9]. Numerous
hypotheses have surfaced as to why mitochondrial function decreases with age. These
hypotheses include increased reactive oxygen species (ROS) production, chronic
inflammation and/or mitochondrial DNA damage [8,10-14]. One
may draw many comparisons between the decline in skeletal muscle function that
occurs with aging and mitochondrial dysfunction in the skeletal muscle of
individuals that consume high levels of calories. Recent work has demonstrated
that mitochondrial damage occurs in obesity due to enhanced ROS and chronic
inflammation caused by increased fatty acid load [15-17]. These
data suggest that modulation of fatty acid-driven signaling pathways,
inflammatory pathways, or ROS may prevent mitochondrial damage and thus,
enhance skeletal muscle oxidative capacity, therefore improving overall metabolic
performance.
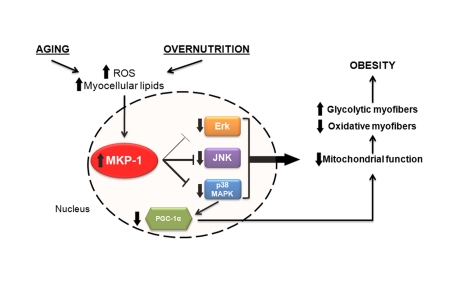
Figure 1. Potential relationship between aging, overnutrition and MKP-1-mediated regulation of skeletal muscle mitochondrial function.
Molecular
targets controlling skeletal muscle function and their impact on aging
Calcineurin/NFAT - Through transgenic and knockout mouse models,
calcineurin, a calcium-activated phosphatase (also known as protein phosphatase
2B), has been shown to be necessary to drive the slow twitch MHC phenotype [18-20].
Calcineurin does this by controlling the phosphorylation status of a
transcription factor called nuclear factor of activated T-cells (NFAT), which
is activated upon dephosphorylation. Active NFAT enhances transcription of
genes that promote a slow-twitch myofiber phenotype [18,21].
Interestingly, calcineurin has been demonstrated to increase in activity with
age in a ROS-dependent manner, and correlates with age-induced muscle
dysfunction [22].
MAP
Kinases - The MAPKs consist of growth factor-regulated
extracellular signal related kinases 1 and 2 (ERK1/2), and the stress-activated
MAPKs, c-jun NH2-terminal kinase (JNK) and p38 MAPK [23]. MAPKs are
activated by phosphorylation on regulatory tyrosine and threonine residues by
upstream MAP kinase kinases (MKKs), and are inactivated by dephosphorylation on
these same regulatory residues by MAPK phosphatases (MKPs) [23,24].
Although it is appreciated that MAPKs play an essential role in myogenesis,
relatively little is known about the involvement of the MAPKs in fiber type
establishment. A role for ERK1/2 has been demonstrated in slow-fiber type
expression, as constitutively active Ras increases MHC type I expression [25]. There is
also evidence that p38 MAPK is able to drive MHC type IIx (intermediate) gene
expression in myoblasts [26].
Increased
MAPK activity after exercise has been shown to be important for
exercise-mediated gene expression, which may contribute to the role of exercise
ameliorating the effects of aging in skeletal muscle [27]. Though the
contribution of MAPKs in aging has not been convincingly addressed in
vertebrates, some studies in drosophila as well as c. elegans
have demonstrated a role for MAPKs for enhancement of lifespan. Whereas the
role of p38 MAPK in promotion of lifespan is controversial [28,29], JNK
activity enhances lifespan by antagonizing insulin signaling [30,31].
Additionally, decreased ERK1/2 activity throughout aging is known to promote
senescence [32]. These data
suggest the possibility that decreased MAPK activity during aging may have
deleterious effects on metabolic health and lifespan.
PGC-1α - The transcriptional co-activator peroxisome
proliferator-activated receptor gamma co-activator 1-α (PGC-1α)
drives mitochondrial biogenesis and function and additionally is important in
the cellular defense against reactive oxygen species [33,34].
Specifically, in skeletal muscle, the expression of PGC-1α drives not only
mitochondrial biogenesis and oxidative myofiber establishment but also
vascularization [35-37]. It has
been found that a high fat diet or fatty acid treatment causes a reduction in
the expression of PGC-1α and other mitochondrial genes in skeletal muscle [38-40], which
may be a mechanism through which excess caloric intake impairs skeletal muscle
function.
Recent
work has also demonstrated that transgenic overexpression of PGC-1α in
skeletal muscle improves sarcopenia and obesity associated with aging in mice [41]. This
implies that pharmacological control of stability or expression of PGC-1α
may be a therapeutic option for the elderly to ameliorate metabolic
dysfunction. Pharmacological modulation of protein stability remains somewhat
challenging and so alternative strategies targeting regulators of PGC-1α
function are likely to meet with more success. Stability of PGC-1α has
been shown to be driven by both acetylation as well as phosphorylation [42,43].
Stability and activity of PGC-1α is enhanced by phosphorylation by p38
MAPK [43] which again raises the interesting issue of the role of the MAPKs in
both sarcopenia and in responses to overnutrition.
MAP
Kinase Phosphatases - MKPs are dual-specificity phosphatases that constitute
a sub-family of the protein tyrosine phosphatase (PTP) superfamily [44]. MKPs are
classified into three subgroups based on subcellular localization, tissue
distribution and substrate preference [44,45]. MKP-1
is the founding member of this family of enzymes and is a nuclear-localized,
immediate-early gene that is responsive to numerous stimuli including ROS,
cytokines, growth factors, and fatty acids [15,46]. MKP-1
expression is confined to the nucleus and therefore is restricted to the
dephosphorylation of the nuclear pool of active MAPKs [47]. Though
MKP-1 has the ability to dephosphorylate all three MAPKs, it displays substrate
preference to the stress-responsive MAPKs, p38 MAPK and JNK [48,49].
As
discussed, enhanced ROS, cytokines and fatty acids can induce the expression of
MKP-1 [15,46]. Therefore, it is conceivable that increased MKP‑1
expression in response to stresses such as these might play a role in the
demise of skeletal muscle mitochondrial function. Indeed, we had provided the
first insight into a potential connection between mitochondrial function and
MKP-1 [47]. We showed
that in mice lacking MKP-1 mitochondrial oxidative phosphorylation was enhanced
in the oxidative portions of skeletal muscle [47]. Consistent
with this MKP-1-deficient mice exhibit enhanced levels of energy expenditure
and were resistant to diet-induced obesity [47]. These
results provide genetic evidence that MKP-1 is connected, through modulation of
the MAPKs, to mitochondrial function in skeletal muscle as well as whole body
energetics. It is tempting to speculate that age- and diet-induced stresses that
promote deleterious mitochondrial function could be attributed to the
convergence, at least in part, to the overexpression of MKP-1 in skeletal
muscle.
More
recently we have been able to provide mechanistic insight in to the pathway
linking MKP-1 to mitochondrial function. We have found that MKP-1 is a key
regulator of the master controller of mitochondrial biogenesis, PGC-1α [15]. When mice
are fed a high-fat diet, the expression levels of MKP-1 in skeletal muscle
increase which results in a reduction in PGC-1α levels. In contrast,
PGC-1α levels are maintained in mice lacking MKP-1 [15]. Consistent
with the notion that PGC-1α promotes oxidative myofiber composition
MKP-1-deficient mice are refractory to the switch from oxidative to glycolytic
myofibers seen in wild type mice fed a high fat diet and remain lean. These
observations support the idea that MKP-1 plays an important role in maintaining
skeletal muscle health. Mechanistically, MKP-1 appears to control a pool of
nuclear p38 MAPK activity that is responsible for phosphorylating PGC-1α
on residues that promote its stability. Therefore, increased MKP-1 expression
levels result in a reduction of p38 MAPK-mediated PGC-1α phosphorylation
and ultimately, stability. Collectively, these observations raise the
possibility that impairment in mitochondrial function might arise through
disturbing the dynamic homeostatic balance between MKP-1 and p38 MAPK activity
in the nucleus. The precise contribution of MKP-1, as well as other MKPs which
have the capacity to dephosphorylate p38 MAPK in the nucleus, to mitochondrial
dysfunction in either aging or overnutrition still remains to be fully
realized. Nevertheless, these recent insights provide a platform from which
these questions can be addressed.
Phosphatases
in aging
The role of PTPs in the aging process is
largely unknown. On a broad scale, enhanced phosphatase activity has been
associated with cellular senescence [32,50]. It is
also recognized that ROS, which inhibit PTPs by modification of the catalytic
cysteine [51], are
increased during the aging process. The enhanced levels of ROS during aging are
likely due to the increased production of pro-inflammatory cytokines [13,52], and
reduced antioxidant defenses [13]. However,
even though ROS inhibit phosphatase activity, enhanced ROS levels are also
known to drive cellular senescence [53], suggesting
a potential role for PTPs both in promoting as well as inhibiting senescence.
ROS also elicits a cellular stress response, which may induce stress-responsive
phosphatases, such as the MKPs, to serve as negative regulators of excessive
MAPK activity [54,55]. More
studies are warranted to determine what role enhanced ROS in a whole organism
has on modulation of PTP activity during aging.
In
support of the data that similar mechanisms are at work in overnutrition as
well as age-induced metabolic dysfunction, our laboratory has demonstrated that
mice lacking MKP-1 are resistant to age-induced obesity, in the absence of a
high fat diet [47]. Moreover,
MKP-1 expression in skeletal muscle of aged human patients has been shown to be
enhanced [56]. Though the
mechanisms behind the resistance to age-induced obesity in mice lacking MKP-1
have not yet been defined, one may hypothesize that similar mechanisms also
apply as those in a high fat diet scenario. For example, increases in cellular
insults such as ROS and intramyocellular lipids, which increase during aging [57,58], may
trigger increased MKP-1expression, thus negatively regulating p38 MAPK
activity, and therefore, PGC-1α stability. Consistent with this notion, as
previously discussed, PGC-1α expression is sufficient to relieve rodents
of age-related sarcopenia [41]. It is
unclear however, how ROS may promote increased levels of MKP-1 expression
without affecting catalytic activity. Nevertheless, modulation of p38 MAPK
activity by MKP-1 may be important in the progression of age-related sarcopenia
and obesity.
Treatments
for sarcopenia
Caloric
Restriction - Calorie restriction is perhaps the most well
recognized anti-aging therapy, and exerts this effect in multiple ways [13]. Calorie
restriction has an anti-inflammatory effect and is important for decreasing
oxidant production during aging [13]. In
addition, calorie restriction is associated with decreased MAPK activity [13].
Provocatively, calorie restriction may serve to prevent the reported increase
in MKP-1 levels during aging. Increased MKP-1 levels during aging would reduce
PGC‑1α levels thereby removing the protective effects of stressors
on skeletal muscle. Calorie restriction also activates the deacetylase SIRT1,
which is a positive regulator of longevity in c. elegans[59]. To this
end, SIRT1 also promotes PGC-1α activity by deacetylation [42], thus
demonstrating a role for calorie restriction in promotion of PGC-1α
activity. Interestingly, acetylation is also known to enhance the phosphatase
function of MKP-1 [60]. One
hypothesis may be that SIRT1 deacetylates, and therefore decreases activity of
MKP‑1, leading to increased PGC-1α function, thus, preventing
myofiber dysfunction.
Exercise - Moderate
exercise retards the aging process, and additionally induces an oxidative
muscle phenotype [13]. Several
hypotheses have arisen regarding the role of exercise in aging. Leading
hypotheses include the fact that a moderate exercise regimen reduces
pro-inflammatory cytokine production and chronic inflammation. The reduced
level of inflammation leads to a decrease in ROS production, which lessens DNA
damage [13]. Other groups
have demonstrated that resistance training enhances myofiber size and
contractility, thus preventing age-mediated myofiber loss [61,62].
Importantly, exercise has been shown to induce MAPK activity in skeletal
muscle, which is important for exercise-mediated gene expression [27]. In
particular, p38 MAPK is known to drive PGC-1α transcription as well as
stability in skeletal muscle [43,63]. In
animals lacking MKP-1 expression, it would be important to determine if the
enhanced MAPK activity in skeletal muscle promotes similar gene expression
events as exercise. If this is the case, mice lacking MKP-1 may exhibit
characteristics similar to that of the molecular effects of exercise.
Perspective
It
is now well accepted that aging is an underlying predisposition to metabolic
disease. Not surprisingly, the pathways that succumb to the deleterious effects
of aging often seem to be those that also control metabolism. Protein tyrosine
phosphatases have not featured as prominent targets for convergence for these
two pathophysiological processes up until now. MKP-1 appears to exhibit
characteristics that afford it the ability to respond to age-related stresses
such as increased ROS and free fatty acids in skeletal muscle. If in fact MKP-1
plays a major role in the convergence in skeletal muscle between aging and
susceptibility to metabolic disease, how this phosphatase is regulated by such
external factors needs to be addressed in significantly more depth. One
intriguing, and as yet to be tested idea, is whether MKP-1 is more deeply
intertwined with other pathways that control longevity such as the SIRTs. As
such, it will be interesting to determine whether there is a longevity
phenotype in mice lacking MKP-1. Clearly, these ideas set the stage for further
work to be conducted on age- and nutrition-related consequences, not only on
MKP-1, but also other protein tyrosine phosphatases in skeletal muscle.
Acknowledgments
A.M.B. is supported by NIH grants AR46504
and DK57751.
R.J.R.F was supported by T32 DK07356.
Conflicts of Interest
The authors of this manuscript have no
conflict of interest to declare.
References
-
1.
Zurlo
F
, Larson
K
, Bogardus
C
and Ravussin
E.
Skeletal muscle metabolism is a major determinant of resting energy expenditure.
J Clin Invest.
1990;
86:
1423
-1427.
[PubMed]
.
-
2.
Zierath
JR
and Hawley
JA.
Skeletal muscle fiber type: influence on contractile and metabolic properties.
PLoS Biol.
2004;
2:
e348
[PubMed]
.
-
3.
Bassel-Duby
R
and Olson
EN.
Signaling Pathways in Skeletal Muscle Remodeling.
Annu Rev Biochem.
2006;
75:
19
-37.
[PubMed]
.
-
4.
Schiaffino
S
and Serrano
A.
Calcineurin signaling and neural control of skeletal muscle fiber type and size.
Trends Pharmacol Sci.
2002;
23:
569
-575.
[PubMed]
.
-
5.
Zamboni
M
, Mazzali
G
, Fantin
F
, Rossi
A
and Di Francesco
V.
Sarcopenic obesity: a new category of obesity in the elderly.
Nutr Metab Cardiovasc Dis.
2008;
18:
388
-395.
[PubMed]
.
-
6.
Johnston
AP
, De
Lisio M
and Parise
G.
Resistance training, sarcopenia, and the mitochondrial theory of aging.
Appl Physiol Nutr Metab.
2008;
33:
191
-199.
[PubMed]
.
-
7.
Hiona
A
and Leeuwenburgh
C.
The role of mitochondrial DNA mutations in aging and sarcopenia: implications for the mitochondrial vicious cycle theory of aging.
Exp Gerontol.
2008;
43:
24
-33.
[PubMed]
.
-
8.
Short
KR
, Bigelow
ML
, Kahl
J
, Singh
R
, Coenen-Schimke
J
and Raghavakaimal
S.
Decline in skeletal muscle mitochondrial function with aging in humans.
Proc Natl Acad Sci U S A.
2005;
102:
5618
-5623.
[PubMed]
.
-
9.
Petersen
KF
, Befroy
D
, Dufour
S
, Dziura
J
, Ariyan
C
and Rothman
DL.
Mitochondrial dysfunction in the elderly: possible role in insulin resistance.
Science.
2003;
300:
1140
-1142.
[PubMed]
.
-
10.
Johannsen
DL
and Ravussin
E.
The role of mitochondria in health and disease.
Curr Opin Pharmacol.
2009;
9:
780
-786.
[PubMed]
.
-
11.
Trifunovic
A
, Wredenberg
A
, Falkenberg
M
, Spelbrink
JN
, Rovio
AT
and Bruder
CE.
Premature ageing in mice expressing defective mitochondrial DNA polymerase.
Nature.
2004;
429:
417
-423.
[PubMed]
.
-
12.
Wilson
FH
, Hariri
A
, Farhi
A
, Zhao
H
, Petersen
KF
and Toka
HR.
A cluster of metabolic defects caused by mutation in a mitochondrial tRNA.
Science.
2004;
306:
1190
-1194.
[PubMed]
.
-
13.
Chung
HY
, Cesari
M
, Anton
S
, Marzetti
E
, Giovannini
S
and Seo
AY.
Molecular inflammation: underpinnings of aging and age-related diseases.
Ageing Res Rev.
2009;
8:
18
-30.
[PubMed]
.
-
14.
Scicchitano
BM
, Rizzuto
E
and Musaro
A.
Counteracting muscle wasting in aging and neuromuscular diseases: the critical role of IGF-1.
Aging.
2009;
1:
451
-457.
[PubMed]
.
-
15.
Roth
RJ
, Le
AM
, Zhang
L
, Kahn
M
, Samuel
VT
and Shulman
GI.
MAPK phosphatase-1 facilitates the loss of oxidative myofibers associated with obesity in mice.
J Clin Invest.
2009;
119:
3817
-3829.
[PubMed]
.
-
16.
Anderson
EJ
, Lustig
ME
, Boyle
KE
, Woodlief
TL
, Kane
DA
and Le
C.
Mitochondrial H2O2 emission and cellular redox state link excess fat intake to insulin resistance in both rodents and humans.
J Clin Invest.
2009;
10:
1
-9.
.
-
17.
Bonnard
C
, Durand
A
, Peyrol
S
, Chanseaume
E
, Chauvin
MA
and Morio
B.
Mitochondrial dysfunction results from oxidative stress in the skeletal muscle of diet-induced insulin-resistant mice.
J Clin Invest.
2008;
118:
789
-800.
[PubMed]
.
-
18.
Chin
ER
, Olson
EN
, Richardson
JA
, Yang
Q
, Humphries
C
and Shelton
JM.
A calcineurin-dependent transcriptional pathway controls skeletal muscle fiber type.
Genes Dev.
1998;
12:
2499
-2509.
[PubMed]
.
-
19.
Naya
FJ
, Mercer
B
, Shelton
J
, Richardson
JA
, Williams
RS
and Olson
EN.
Stimulation of slow skeletal muscle fiber gene expression by calcineurin in vivo.
J Biol Chem.
2000;
275:
4545
-4548.
[PubMed]
.
-
20.
Parsons
SA
, Millay
DP
, Wilkins
BJ
, Bueno
OF
, Tsika
GL
and Neilson
JR.
Genetic loss of calcineurin blocks mechanical overload-induced skeletal muscle fiber type switching but not hypertrophy.
J Biol Chem.
2004;
279:
26192
-26200.
[PubMed]
.
-
21.
Serrano
AL
, Murgia
M
, Pallafacchina
G
, Calabria
E
, Coniglio
P
and Lomo
T.
Calcineurin controls nerve activity-dependent specification of slow skeletal muscle fibers but not muscle growth.
Proc Natl Acad Sci U S A.
2001;
98:
13108
-13113.
[PubMed]
.
-
22.
Dai
DF
, Santana
LF
, Vermulst
M
, Tomazela
DM
, Emond
MJ
and MacCoss
MJ.
Overexpression of catalase targeted to mitochondria attenuates murine cardiac aging.
Circulation.
2009;
119:
2789
-2797.
[PubMed]
.
-
23.
Raman
M
, Chen
W
and Cobb
MH.
Differential regulation and properties of MAPKs.
Oncogene.
2007;
26:
3100
-3112.
[PubMed]
.
-
24.
Owens
DM
and Keyse
SM.
Differential regulation of MAP kinase signalling by dual-specificity protein phosphatases.
Oncogene.
2007;
26:
3203
-3213.
[PubMed]
.
-
25.
Murgia
M
, Serrano
AL
, Calabria
E
, Pallafacchina
G
, Lomo
T
and Schiaffino
S.
Ras is involved in nerve-activity-dependent regulation of muscle genes.
Nat Cell Biol.
2000;
2:
142
-147.
[PubMed]
.
-
26.
Meissner
JD
, Chang
KC
, Kubis
HP
, Nebreda
AR
, Gros
G
and Scheibe
RJ.
The p38alpha/beta mitogen-activated protein kinases mediate recruitment of CREB-binding protein to preserve fast myosin heavy chain IId/x gene activity in myotubes.
J Biol Chem.
2007;
282:
7265
-7275.
[PubMed]
.
-
27.
Sakamoto
K
and Goodyear
LJ.
Invited review: intracellular signaling in contracting skeletal muscle.
J Appl Physiol.
2002;
93:
369
-383.
[PubMed]
.
-
28.
Troemel
ER
, Chu
SW
, Reinke
V
, Lee
SS
, Ausubel
FM
and Kim
DH.
p38 MAPK regulates expression of immune response genes and contributes to longevity in C. elegans.
PLoS Genet.
2006;
2:
e183
[PubMed]
.
-
29.
Alper
S
, McElwee
MK
, Apfeld
J
, Lackford
B
, Freedman
JH
and Schwartz
DA.
The Caenorhabditis elegans germ line regulates distinct signaling pathways to control lifespan and innate immunity.
J Biol Chem.
2010;
285:
1822
-1828.
[PubMed]
.
-
30.
Oh
SW
, Mukhopadhyay
A
, Svrzikapa
N
, Jiang
F
, Davis
RJ
and Tissenbaum
HA.
JNK regulates lifespan in Caenorhabditis elegans by modulating nuclear translocation of forkhead transcription factor/DAF-16.
Proc Natl Acad Sci U S A.
2005;
102:
4494
-4499.
[PubMed]
.
-
31.
Wang
MC
, Bohmann
D
and Jasper
H.
JNK extends life span and limits growth by antagonizing cellular and organism-wide responses to insulin signaling.
Cell.
2005;
121:
115
-125.
[PubMed]
.
-
32.
Tresini
M
, Lorenzini
A
, Torres
C
and Cristofalo
VJ.
Modulation of replicative senescence of diploid human cells by nuclear ERK signaling.
J Biol Chem.
2007;
282:
4136
-4151.
[PubMed]
.
-
33.
Wu
Z
, Puigserver
P
, Andersson
U
, Zhang
C
, Adelmant
G
and Mootha
V.
Mechanisms controlling mitochondrial biogenesis and respiration through the thermogenic coactivator PGC-1.
Cell.
1999;
98:
115
-124.
[PubMed]
.
-
34.
St-Pierre
J
, Drori
S
, Uldry
M
, Silvaggi
JM
, Rhee
J
and Jager
S.
Suppression of reactive oxygen species and neurodegeneration by the PGC-1 transcriptional coactivators.
Cell.
2006;
127:
397
-408.
[PubMed]
.
-
35.
Arany
Z
, Foo
SY
, Ma
Y
, Ruas
JL
, Bommi-Reddy
A
and Girnun
G.
HIF-independent regulation of VEGF and angiogenesis by the transcriptional coactivator PGC-1alpha.
Nature.
2008;
451:
1008
-1012.
[PubMed]
.
-
36.
Lin
J
, Wu
H
, Tarr
PT
, Zhang
CY
, Wu
Z
and Boss
O.
Transcriptional co-activator PGC-1 alpha drives the formation of slow-twitch muscle fibres.
Nature.
2002;
418:
797
-801.
[PubMed]
.
-
37.
Handschin
C
, Chin
S
, Li
P
, Liu
F
, Maratos-Flier
E
and Lebrasseur
NK.
Skeletal muscle fiber-type switching, exercise intolerance, and myopathy in PGC-1alpha muscle-specific knock-out animals.
J Biol Chem.
2007;
282:
30014
-30021.
[PubMed]
.
-
38.
Patti
ME
, Butte
AJ
, Crunkhorn
S
, Cusi
K
, Berria
R
and Kashyap
S.
Coordinated reduction of genes of oxidative metabolism in humans with insulin resistance and diabetes: Potential role of PGC1 and NRF1.
Proc Natl Acad Sci U S A.
2003;
100:
8466
-8471.
[PubMed]
.
-
39.
Mootha
VK
, Lindgren
CM
, Eriksson
KF
, Subramanian
A
, Sihag
S
and Lehar
J.
PGC-1alpha-responsive genes involved in oxidative phosphorylation are coordinately downregulated in human diabetes.
Nat Genet.
2003;
34:
267
-273.
[PubMed]
.
-
40.
Crunkhorn
S
, Dearie
F
, Mantzoros
C
, Gami
H
, da
Silva WS
and Espinoza
D.
Peroxisome proliferator activator receptor gamma coactivator-1 expression is reduced in obesity: potential pathogenic role of saturated fatty acids and p38 mitogen-activated protein kinase activation.
J Biol Chem.
2007;
282:
15439
-15450.
[PubMed]
.
-
41.
Wenz
T
, Rossi
SG
, Rotundo
RL
, Spiegelman
BM
and Moraes
CT.
Increased muscle PGC-1alpha expression protects from sarcopenia and metabolic disease during aging.
Proc Natl Acad Sci U S A.
2009;
106:
20405
-20410.
[PubMed]
.
-
42.
Gerhart-Hines
Z
, Rodgers
JT
, Bare
O
, Lerin
C
, Kim
SH
and Mostoslavsky
R.
Metabolic control of muscle mitochondrial function and fatty acid oxidation through SIRT1/PGC-1alpha.
Embo J.
2007;
26:
1913
-1923.
[PubMed]
.
-
43.
Puigserver
P
, Rhee
J
, Lin
J
, Wu
Z
, Yoon
JC
and Zhang
CY.
Cytokine stimulation of energy expenditure through p38 MAP kinase activation of PPARgamma coactivator-1.
Mol Cell.
2001;
8:
971
-982.
[PubMed]
.
-
44.
Dickinson
RJ
and Keyse
SM.
Diverse physiological functions for dual-specificity MAP kinase phosphatases.
J Cell Sci.
2006;
119:
4607
-4615.
[PubMed]
.
-
45.
Theodosiou
A
and Ashworth
A.
MAP kinase phosphatases.
Genome Biol.
2002;
3:
REVIEWS3009
[PubMed]
.
-
46.
Boutros
T
, Chevet
E
and Metrakos
P.
Mitogen-activated protein (MAP) kinase/MAP kinase phosphatase regulation: roles in cell growth, death, and cancer.
Pharmacol Rev.
2008;
60:
261
-310.
[PubMed]
.
-
47.
Wu
JJ
, Roth
RJ
, Anderson
EJ
, Hong
EG
, Lee
MK
and Choi
CS.
Mice lacking MAP kinase phosphatase-1 have enhanced MAP kinase activity and resistance to diet-induced obesity.
Cell Metab.
2006;
4:
61
-73.
[PubMed]
.
-
48.
Wu
JJ
and Bennett
AM.
Essential role for mitogen-activated protein (MAP) kinase phosphatase-1 in stress-responsive MAP kinase and cell survival signaling.
J Biol Chem.
2005;
280:
16461
-16466.
[PubMed]
.
-
49.
Franklin
CC
and Kraft
AS.
Conditional expression of the mitogen-activated protein kinase (MAPK) phosphatase MKP-1 preferentially inhibits p38 MAPK and stress-activated protein kinase in U937 cells.
J Biol Chem.
1997;
272:
16917
-16923.
[PubMed]
.
-
50.
Bernier
L
and Wang
E.
A prospective view on phosphatases and replicative senescence.
Exp Gerontol.
1996;
31:
13
-19.
[PubMed]
.
-
51.
Meng
TC
, Fukada
T
and Tonks
NK.
Reversible oxidation and inactivation of protein tyrosine phosphatases in vivo.
Mol Cell.
2002;
9:
387
-399.
[PubMed]
.
-
52.
Kamata
H
, Honda
S
, Maeda
S
, Chang
L
, Hirata
H
and Karin
M.
Reactive oxygen species promote TNFalpha-induced death and sustained JNK activation by inhibiting MAP kinase phosphatases.
Cell.
2005;
120:
649
-661.
[PubMed]
.
-
53.
Rai
P
, Onder
TT
, Young
JJ
, McFaline
JL
, Pang
B
and Dedon
PC.
Continuous elimination of oxidized nucleotides is necessary to prevent rapid onset of cellular senescence.
Proc Natl Acad Sci U S A.
2009;
106:
169
-174.
[PubMed]
.
-
54.
Zhou
JY
, Liu
Y
and Wu
GS.
The role of mitogen-activated protein kinase phosphatase-1 in oxidative damage-induced cell death.
Cancer Res.
2006;
66:
4888
-4894.
[PubMed]
.
-
55.
Kuwano
Y
, Kim
HH
, Abdelmohsen
K
, Pullmann
R Jr
, Martindale
JL
and Yang
X.
MKP-1 mRNA stabilization and translational control by RNA-binding proteins HuR and NF90.
Mol Cell Biol.
2008;
28:
4562
-4575.
[PubMed]
.
-
56.
Williamson
D
, Gallagher
P
, Harber
M
, Hollon
C
and Trappe
S.
Mitogen-activated protein kinase (MAPK) pathway activation: effects of age and acute exercise on human skeletal muscle.
J Physiol.
2003;
547:
977
-987.
[PubMed]
.
-
57.
Morino
K
, Petersen
KF
and Shulman
GI.
Molecular mechanisms of insulin resistance in humans and their potential links with mitochondrial dysfunction.
Diabetes.
2006;
55 Suppl 2:
S9
-S15.
[PubMed]
.
-
58.
Thompson
LV
Age-related muscle dysfunction.
Exp Gerontol.
2009;
44:
106
-111.
[PubMed]
.
-
59.
Bordone
L
and Guarente
L.
Calorie restriction, SIRT1 and metabolism: understanding longevity.
Nat Rev Mol Cell Biol.
2005;
6:
298
-305.
[PubMed]
.
-
60.
Cao
W
, Bao
C
, Padalko
E
and Lowenstein
CJ.
Acetylation of mitogen-activated protein kinase phosphatase-1 inhibits Toll-like receptor signaling.
J Exp Med.
2008;
205:
1491
-1503.
[PubMed]
.
-
61.
Adamo
ML
and Farrar
RP.
Resistance training, and IGF involvement in the maintenance of muscle mass during the aging process.
Ageing Res Rev.
2006;
5:
310
-331.
[PubMed]
.
-
62.
Ljubicic
V
, Joseph
AM
, Adhihetty
PJ
, Huang
JH
, Saleem
A
and Uguccioni
G.
Molecular basis for an attenuated mitochondrial adaptive plasticity in aged skeletal muscle.
Aging.
2009;
1:
818
-830.
[PubMed]
.
-
63.
Akimoto
T
, Pohnert
SC
, Li
P
, Zhang
M
, Gumbs
C
and Rosenberg
PB.
Exercise stimulates Pgc-1alpha transcription in skeletal muscle through activation of the p38 MAPK pathway.
J Biol Chem.
2005;
280:
19587
-19593.
[PubMed]
.