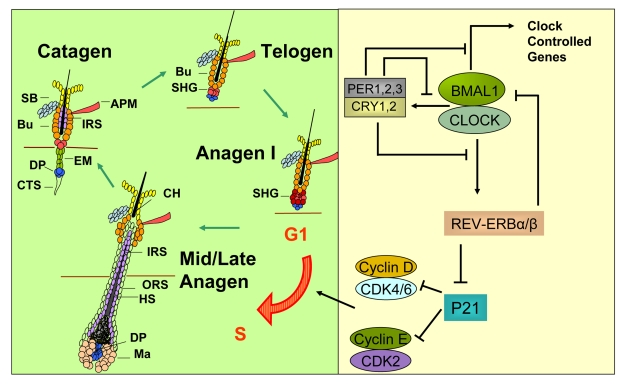
Figure 2.A model for how circadian clock genes participate in regulation of the synchronized hair growth cycle through regulation of cell cycle progression in the secondary hair germ.The hair growth cycle (left panel) is a
continuous process consisting of the quiescent telogen phase followed by
the growth phase (anagen) where signals, presumably originating in the
dermal papilla, activate stem and progenitor cell proliferation leading to
growth and differentiation of the hair shaft. Anagen is followed by catagen
where the lower two-thirds of the follicle undergo apoptosis, sparing the
stem cell compartments and the dermal papilla. The CLOCK/BMAL1 complex is
at the core of the mammalian circadian clock mechanism (right panel). It
activates multiple genes, collectively referred to as clock controlled
genes. Among these genes are Per1,2,3 and Cry1,2 whose protein
products translocate into the nucleus to inhibit the transcriptional
activity of the CLOCK/BMAL1 complex. Rev-erbα is another
clock controlled gene whose protein product negatively regulates expression
of Bmal1. Additionally, REV-ERBα
directly inhibits expression of the G1-S cell cycle inhibitor p21WAF1/CIP.
In the absence of BMAL1, downregulation of Rev-erbα leads to high P21
expression and G1 arrest in the hair germ cells during anagen I of the hair
follicle cycle, thus delaying anagen progression. APM - arrector pili
muscle, Bu - bulge, CH - club hair, CTS - connective tissue sheath, DP -
dermal papilla, EM - epithelial membrane, HS - hair shaft, IRS - inner root
sheath, Ma - matrix, ORS - outer root sheath, SB - sebaceous gland, SHG -
secondary hair germ.