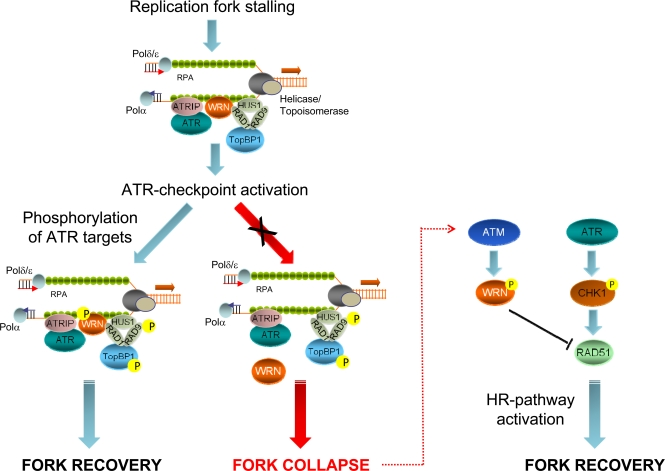
Figure 3.Role of ATR- or ATM-dependent modulation of WRN function in promoting correct recovery from replication arrestIn response to replication fork stalling, the ATR-ATRIP and 9.1.1 complexes are independently loaded at RPA-bound ssDNA regions to activate the replication checkpoint. Early after checkpoint activation, WRN is recruited to fork stalling sites through its interaction with RAD1, a subunit of the 9.1.1 complex. This stage corresponds to formation of WRN foci, which co-localise with RPA. After 9.1.1-dependent relocalisation in foci, WRN is phosphorylated by ATR in a manner that could be dependent on TopBP1. Phosphorylation of WRN by ATR is instrumental for preventing DSBs accumulation at stalled forks and and ensuring faithful recovery of replication forks. Degeneration of the stalled forks into breakage, such as in the absence of ATR phosphorylation of WRN, can cause the activation of an alternative pathway: in this case, ATM-dependent phosphorylation promotes de-localization of WRN from collapsed forks to prepare the way for RAD51-mediated replication recovery, which is also dependent on RAD51 phosphorylation by CHK1.