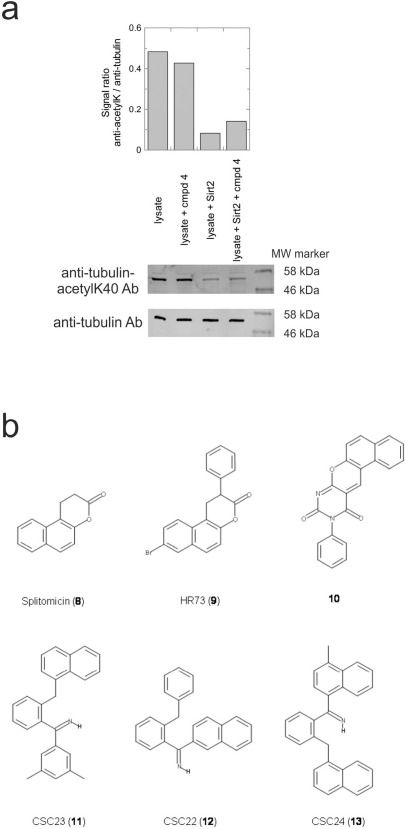
Figure 4.Effects of compounds 3 and 4 on Sirt2-dependent α-tubulin deacetylation and chemical structures of published Sirtuin inhibitors and compounds analyzed here(a) Lysates of HEK cells were incubated with Sirt2 in presence and absence of 100 μM compound 4 and then probed with anti-tubulin-acetylK40 antibody and anti-tubulin antibody, respectively. The bars show the signal ratios, indicating that the tubulin-deacetylating activity of Sirt2 is lowered by the addition of compound 4. (b) Splitomicin (8) is an established inhibitor for yeast Sir2, and HR73 (9) and the tetracyclic pyrimidinedione 10 for mammalian Sirt1. 11, 12, and 13 are identified here as Sirt2-specific inhibitors, which form a series of structurally related compounds yet show varying inhibition potencies.