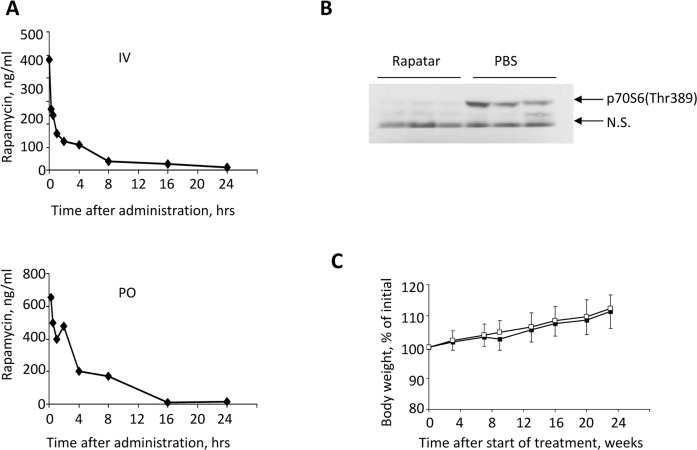
Figure 1.Pharmacokinetic and biological characteristics of Rapatar. (A) Rapamycin concentration-time profile in blood after intravenous (IV, top) and oral (PO, bottom) administration of Rapatar to mice (mean values, n = 3). A single dose of Rapatar was administered either IV (0.4mg/kg) or PO (4mg/kg). Blood samples were collected at designated times and analyzed for rapamycin by LC/MS/MS. (B) Rapatar blocks mTOR activation in vivo. Six C57/Bl/6J mice were food-deprived for 18 hrs. At the end of fasting period animals received either Rapatar (0.5mg/kg) or PBS via gavage and were allowed access to food. One hour later animals were sacrificed, livers were dissected and protein lysates were analyzed for mTOR activity by probing with p70S6(Thr389) antibody. (C) No acute or long-term toxicity are associated with PO administration of Rapatar. C57Bl/6J male mice received either Rapatar or PBS starting 8 weeks of age (10 mice/group) for 24 weeks according to the protocol described above. No loss in body weight was detected in experimental group throughout the treatment period. Both experimental and control groups showed similar gain in body weight with age.