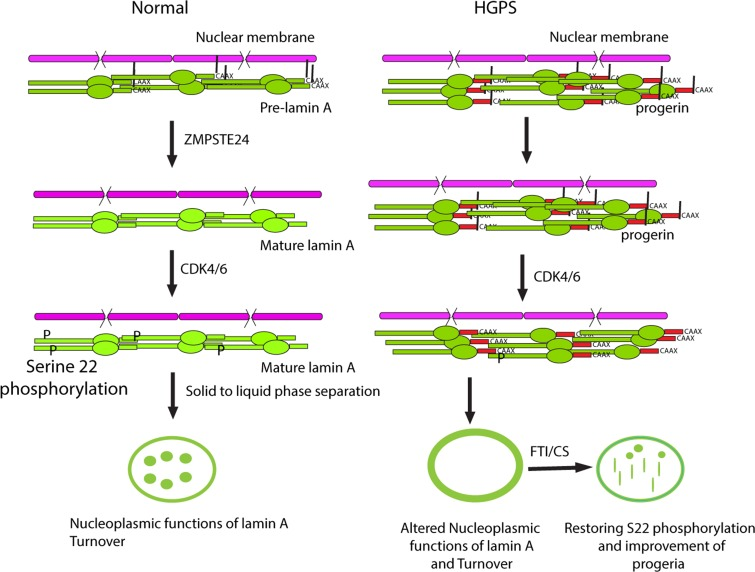
Figure 7.Cleavage and defarnesylation of lamin A allows serine 22 phosphorylation, a modification that promotes a solid to liquid phase separation of lamin polymersThis phase separation can make soluble lamin A available for nucleoplasmic functions and allows lamin A turnover, regulating the overall size and stiffness of the envelope. Progerin is not defarnesylated and is resistant to phosphorylation at serine 22, compromising its solubilization, nucleoplasmic functions and turnover. FTI-277 or the mutation of the farnesylation site (CS) partially correct the defects in progerin but the solid to liquid transition is defective and intranuclear progerin forms mostly fibrous rods. New treatments are required for a full normalization of lamin A in progeria.