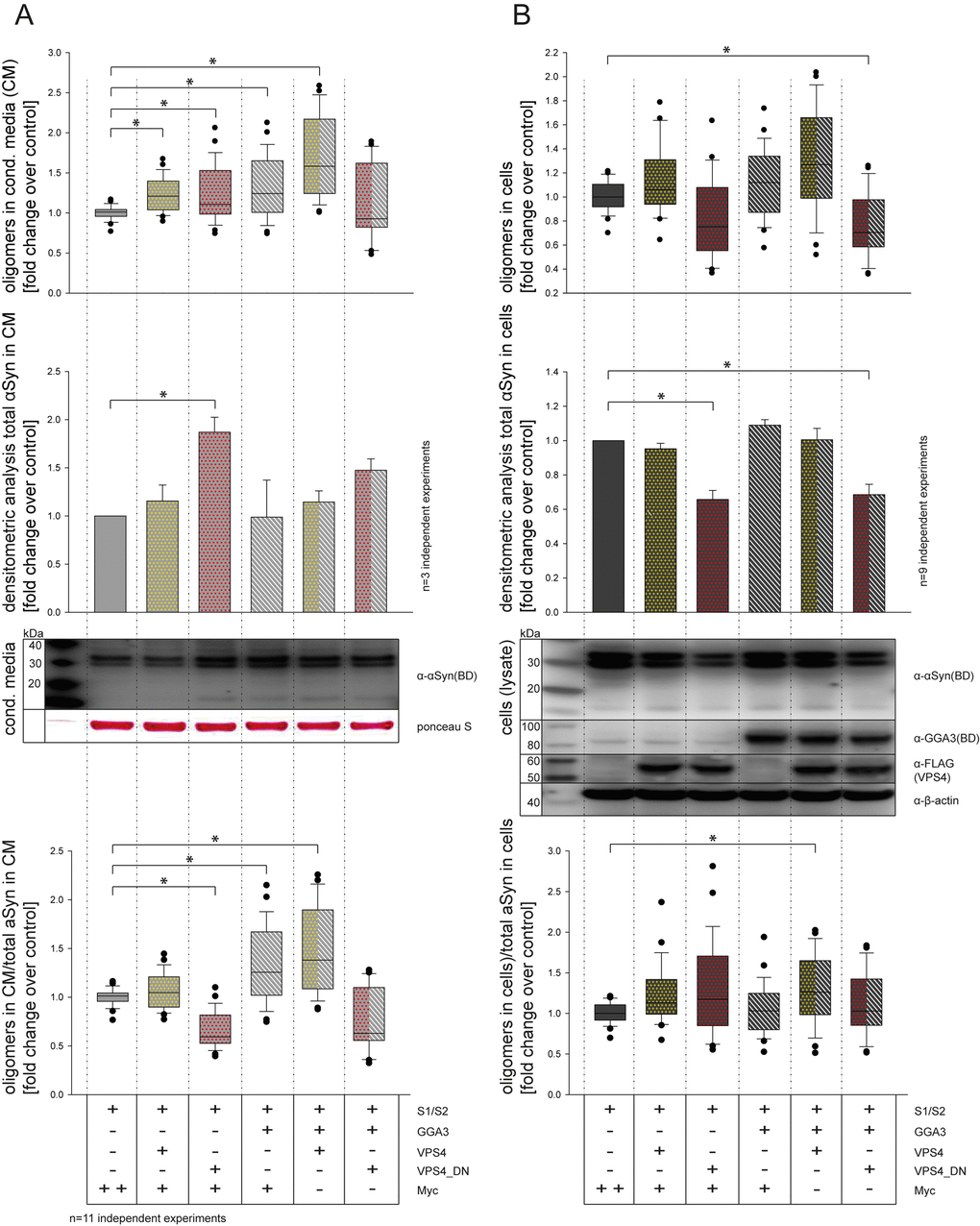
Figure 3.GGA3 and VPS4 have additive effects on α-syn oligomer secretion. CM from N2A cells co-expressing S1/S2 and either empty control vector, GGA3, VPS4 and VPS4_dn alone or in combination were collected 48h after transfection. α-syn oligomer levels were measured by luciferase assay in conditioned media (CM) (A) as well as cells (B). The upper panel of (A) shows α-syn oligomer levels in CM measured by luciferase assay as fold change to control (Myc). The middle panel of (A) shows the total α-syn levels in CM measured by densitometric analysis as fold change to control. The lower panel of (A) shows the proportion of α-syn oligomers in CM to total α-syn in CM as fold change to control. The upper panel of (B) shows α-syn oligomer levels in cells measured by luciferase assay as fold change to control (Myc). The middle panel of (B) shows the total α-syn levels in cells measured by densitometric analysis as fold change to control. The lower panel of (B) shows the proportion of α-syn oligomers in cells to total α-syn in cells as fold change to control. In proportion to total α-syn, VPS4 and GGA3 overexpression increased α-syn oligomer secretion (A, lower panel, lane 4+5). In contrast, expression of VPS4_dn resulted in decreased α-syn oligomerization in CM (A, lower panel, lane 3), whereas intracellular α-syn oligomer as well as total α-syn levels were decreased (B, upper and middle panel, lane 3+6). The results of n=11 independent experiments are shown. Statistical analysis was performed using Kruskal-Wallis one-way ANOVA on ranks followed by multiple comparisons versus control group (Dunn's Method) with *=p<0.05.