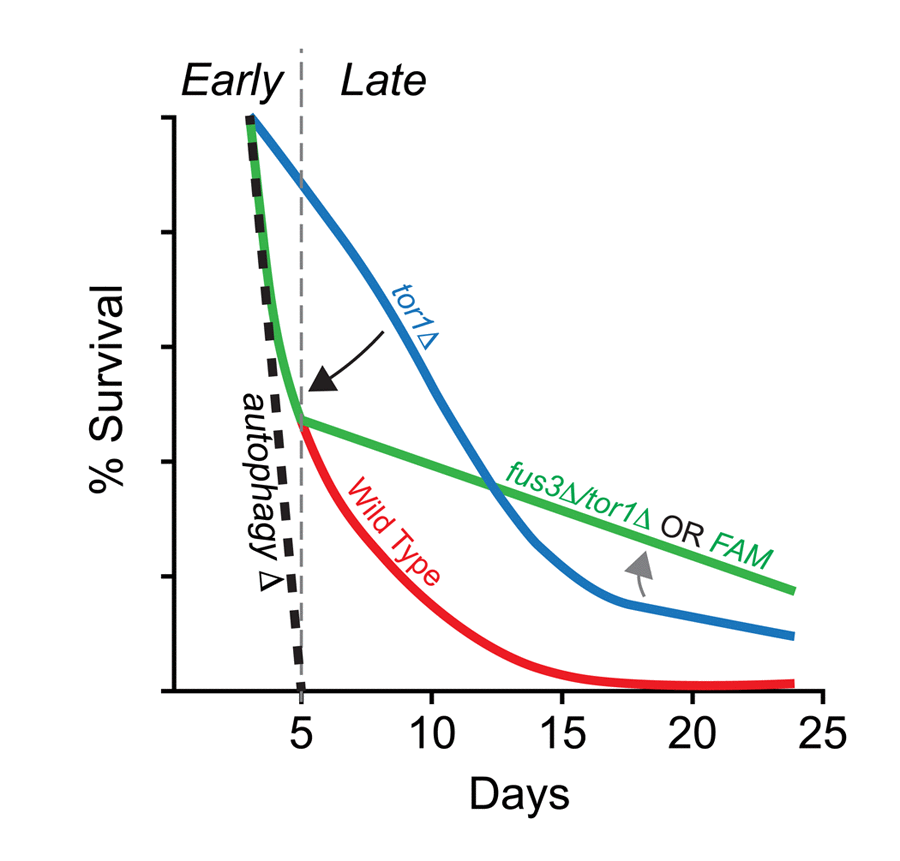
Figure 8.Summary of the combined effects of FUS3 and TOR1 on CLS in yeast. (A) Diagram indicating the dynamic CLS response of mapkΔ and tor1Δ cells. The elongated lifespan of tor1Δ cells is dependent on the ability of cells to carry out autophagocytosis, since cells lacking a functional autophagosome (e.g. atg1Δ) die rapidly, regardless of the functional activity (or presence) of Tor1 in cells. Thus, the elongated lifespan phenotype of tor1Δ cells can be nullified by autophagy-null mutations (e.g. atg1Δ; black dashed line) (taken from Alvers et al. [16]). Early in the CLS experiment, deletion of FUS3 (which is required for efficient autophagy [42]) has a similar effect by reducing the survival of tor1Δ yeast (fus3Δ/tor1Δ; black arrow). This effect is similar to the decay rate of wild type cells within the first week of the CLS experiment, which may or may not be coincidental. The effect is also not as extreme as is observed for autophagy nullification. In later stages of the CLS assay, fus3Δ/tor1Δ cells exhibit considerable change in decay rate, indicating a possible transition to another stage in which the balance between antagonistic autophagy regulators (as well as other CLS-controlling processes) shifts (grey arrow) in a manner that is not normally observed when both genes are present in wild type form. This two-stage response likely requires the ability of Fus3 to be activated by phosphorylation since substitution of Fus3 with Fus3T180A,Y182F (i.e. FAM) exhibits the same CLS response as deletion of both FUS3 and TOR1 together.