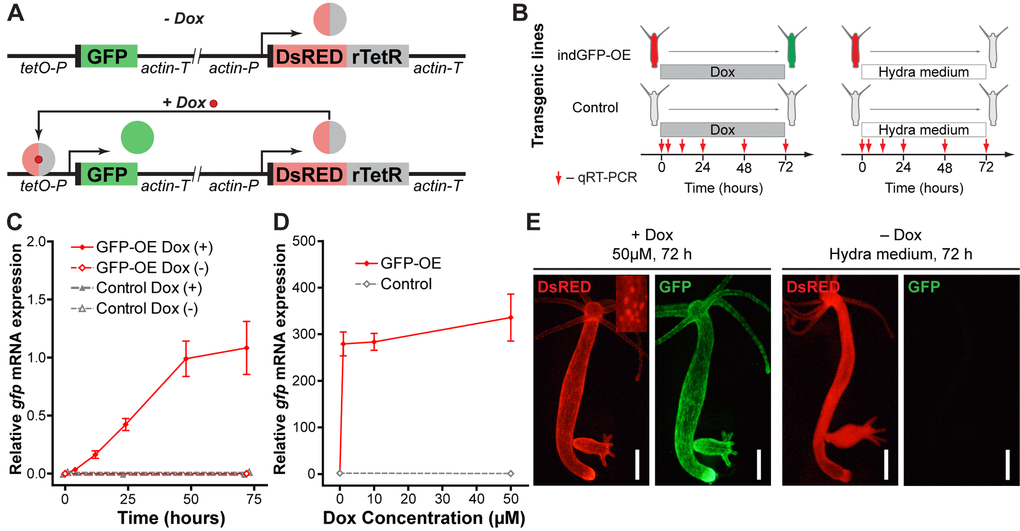
Figure 9.Development and validation of the inducible gene expression system. (A) Construct design and function principle of the inducible gene expression system. The gene of interest (GFP) is cloned under the control of a tetracycline-sensitive promotor (tetO-P, see Methods). Constitutive ubiquitous actin promotor (actin-P) drives expression of a tetracycline-responsive transcriptional transactivator (rTetR) fused to DsRED fluorescent protein. In the absence of doxycycline (-Dox, top), the protein is not able to bind to DNA. In the presence of doxycycline (+Dox, bottom) DsRED-rTetR fusion protein binds the tetO-P and activates expression of GFP. (B) Experimental setup for validation of the inducible system by qRT-PCR. Transgenic polyps containing the inducible construct (indGFP-OE) and corresponding empty controls were incubated in 10 μM doxycycline solution (Dox) or Hydra medium. Samples were taken at indicated time points for expression analysis by qRT-PCR. (C) Evaluation of the gfp expression level by qRT-PCR reveals time-dependent response of the transgenic indGFP-OE polyps to Dox treatment (n=3 for each time point, mean±S.E.M.). (D) Evaluation of the gfp expression level by qRT-PCR reveals efficient induction after 72 h treatment with Dox concentrations from 1 to 50 μM (n=3 for each point, mean±S.E.M.). (E) Life imaging of representative polyps of the indGFP-OE line treated for 72 h with doxycycline (50 μM, +Dox) or with Hydra medium (Dox-). DsRED fluorescence (red) is present in all epithelial cells of the polyps and localized to the nuclei (inset). Only in the presence of doxycycline (+Dox, left) GFP fluorescence (green) is detected. Scale bar: 300 μm.