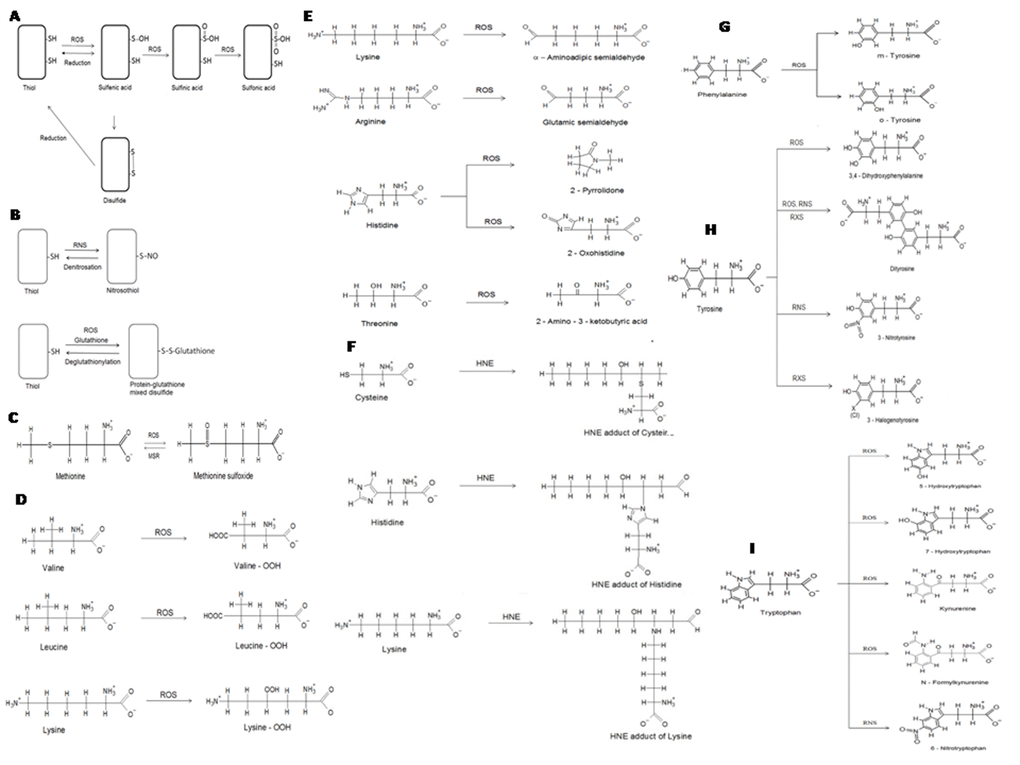
Figure 1.Selected non-enzymatic protein modifications. (A) oxidation of cysteine residues in proteins. Cysteine residues may be oxidized to sulfenic, sulfinic and sulfonic derivatives or form disulfide bonds. Oxidation to sulfenic acid and formation of disulfides is reversible; (B) modifications of cysteine residues in proteins: formation of nitrosocysteine and S-glutathionylation; (C) oxidation of methionine forms methionine sulfoxide, which may be reduced back to methionine by methionine sulfoxide reductases (MSR); (D) formation of hydroperoxides of valine, lysine and leucine; (E) formation of carbonyl derivatives of lysine, arginine, His and threonine; (F) formation of 4-hydroxynonenal adducts of cysteine, His and lysine; (G) oxidative modifications of phenylalanine; (H) modifications of tyrosine; (I) modifications of tryptophan.