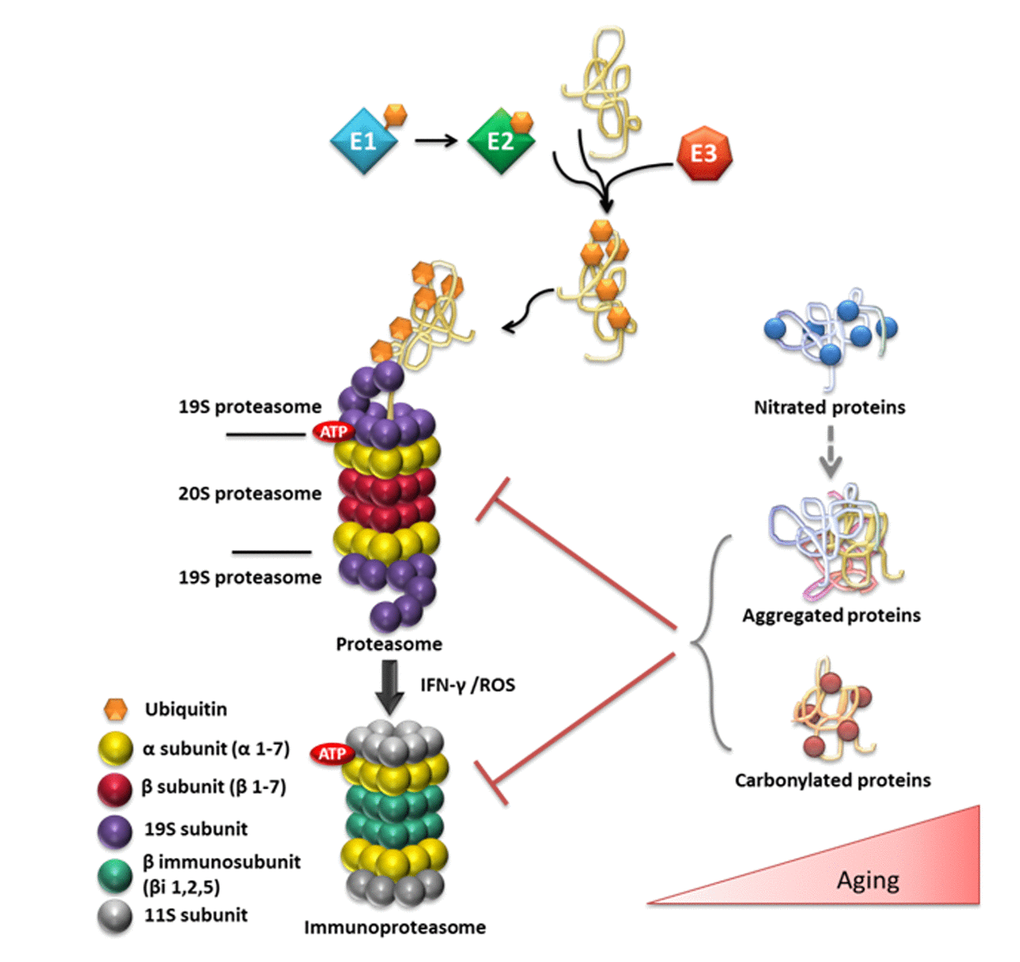
Figure 3.Overview of the ubiquitin (Ub)/proteasome system and its substrates in relation to aging. Ub conjugation is mediated by a series of enzymes. The Ub-activating enzyme E1 transfers Ub to the active site of the E2 Ub-conjugating enzyme and the E3 Ub-ligase ligate Ub to the target protein. The ubiquitinated protein is targeted to the 26S proteasome for degradation. The 26S proteasome consists of the 20S catalytic core and of one or two 19S regulatory particles. The 20S proteasome consists of 28 subunits that are divided to two outer α and two central β rings. The immunoproteasome is induced in response to the immunomodulatory cytokine interferon-gamma (IFN-gamma) or in response to the increased OS that is observed during aging. The age-related elevation of OS also causes oxidative damage to proteins, such as carbonylation. In addition, the excessive •NO production during aging can lead to aberrant S-nitrosylation/tyrosine nitration. Nitrated proteins are prone to aggregation and may contribute to the onset and progression of various neurodegenerative diseases, including AD or PD. The accumulation of aggregated or carbonylated proteins inhibit proteasomal activity contributing the observed proteasomal dysfunction during aging and to the advancement of age-related pathologies.