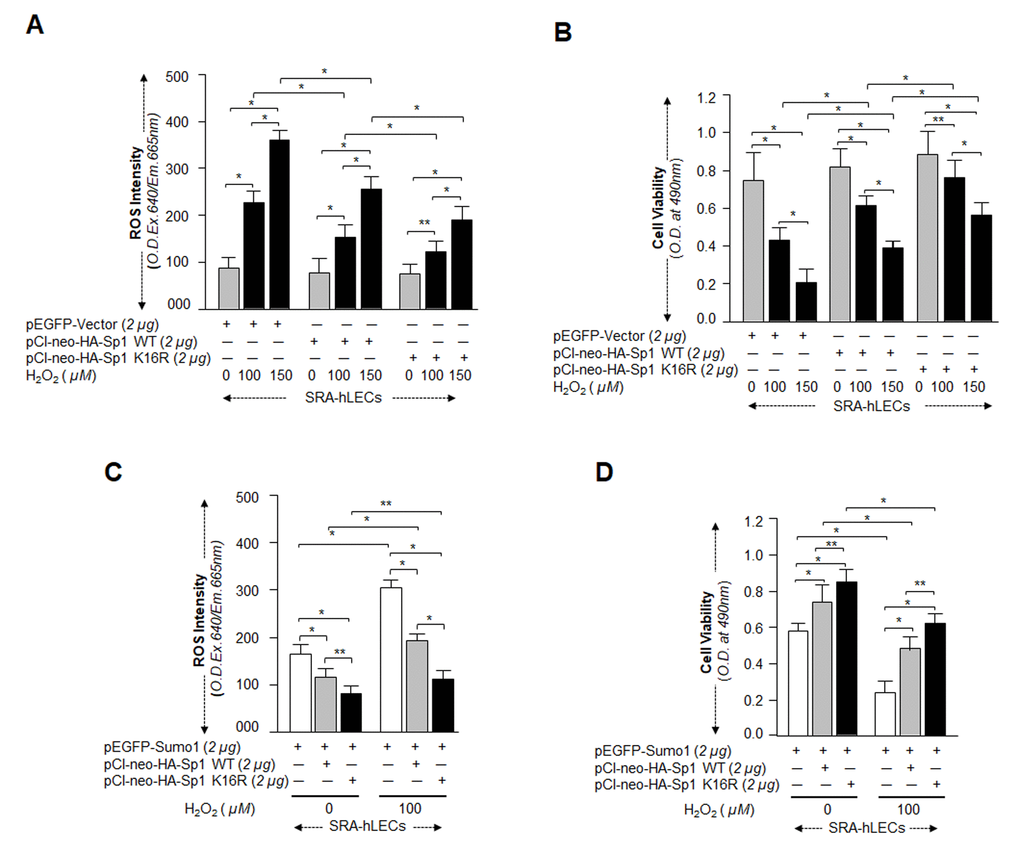
Figure 10.Sp1K16R mutated at Sumo1 binding site provided enhanced cytoprotection against oxidative stress. (A and B) SRA-hLECs were transfected with either pEGFP-Vector, pCl-neo-HA-Sp1, or pCl-neo-HA-Sp1K16R and then exposed to different concentrations of H2O2 as indicated. After 8h of H2O2 exposure, ROS intensity was quantified with CellROX deep red reagent (A). 24h later viability of cells was analyzed by MTS assay (B) as shown. Histogram values represent mean ± SD of three independent experiments. 0 vs 100 vs 150µM H2O2 and pEGFP-Vector vs pCl-neo-HA-Sp1 WT vs pCl-neo-HA-Sp1K16R (**p<0.05; *p<0.001). (C and D) SRA-hLECs were transfected with pEGFP-Sumo1 along with either pCMV-Vector (open bars), pCl-neo-HA-Sp1 (gray bars), or pCl-neo-HA-Sp1K16R (black bars), and then exposed to oxidative stress. ROS intensity (C) and cell viability (D) are presented as histograms. Values represent mean ± SD of three independent experiments. 0 vs 100µM H2O2 and pEGFP-Sumo1 vs pEGFP-Sumo1 plus pCl-neo-HA-Sp1 WT vs pEGFP-Sumo1 plus pCl-neo-HA-Sp1K16R (**p<0.05; *p<0.001). Sumoylation-deficient Sp1K16R (black bars) showed significantly higher protection and reduced ROS production, indicating that mutant Sp1K16R was more effective in protecting cells from oxidative stress Sumoylation-mediated insults.