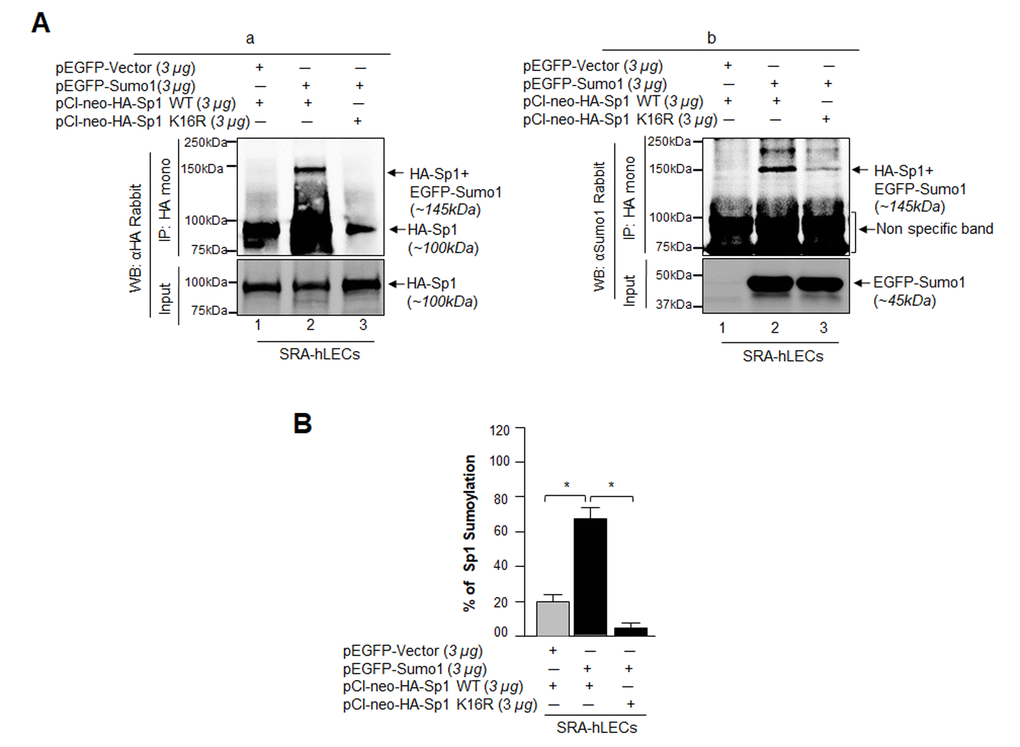
Figure 7.Sumo1 modified Sp1 at K16 residue in vivo. (A) SRA-hLECs (1.2X106) were transfected with pEGFP-Sumo1 (3µg) along with Sp1 WT (3µg) or its mutant Sp1K16R (mutated at Sumoylation sites) using (3µg) plasmid as indicated. Exogenous Sp1 was immunoprecipitated (IP) from cell lysates containing equal amounts of proteins, and its Sumoylated form was detected with anti-HA (Ba) and anti-Sumo1 (Bb) rabbit polyclonal antibodies as indicated. Cell lysates were prepared and subjected to IP using anti-HA monoclonal antibody. IP with anti-HA monoclonal antibody shows a single-exogenous Sumoylated band at ~145 kDa (lane 2, pEGFP-Sumo1+pCl-neo-HA-Sp1WT). No Sumoylation band could be detected in cell extracts of pEGFP-Vector+pCl-neo-HA-Sp1WT or pEGFP-Sumo1+pCl-neo-HA-Sp1-K16R transfected cells (B, a and b; lanes 1 and 3). (B) SRA-hLECs were transfected with pCl-neo-HA-Sp1WT plus pEGFP-Vector, or pCl-neo-HA-Sp1WT plus pEGFP-Sumo1, or pCl-neo-HA-Sp1-K16R plus pEGFP-Sumo1. 48h later, total cell lysates were prepared and processed for Sumo1-ELISA assay according to the manufacturer’s protocol (EpiQuikTM) to measure the Sumoylated form of Sp1. Data represent mean ± SD from three independent experiments: pCl-neo-HA-Sp1WT plus pEGFP-Vector, vs pCl-neo-HA-Sp1WT plus pEGFP-Sumo1, vs pCl-neo-HA-Sp1 K16R plus pEGFP-Sumo1 (*p<0.001).