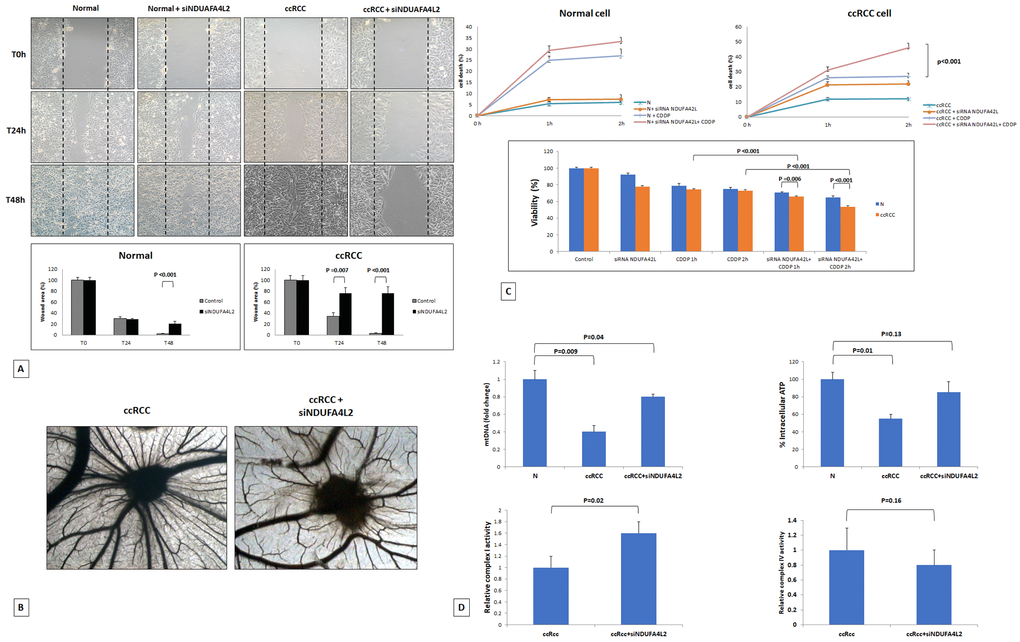
Figure 5.Wounded normal and tumor cell monolayers were photographed 24 and 48 hours after the mechanical scratch and the area of the wounds was measured in 3 independent wound sites per group. When specified, the cells were treated with small interfering RNA targeting NDUFA4L2 (siNDUFA4L2). RCC cells treated with siNDUFA4L2 have decreased cell migratory capabilities compared with untreated tumor cells (A). Chick embryo chorioallantoic membrane angiogenic assay: when tumor cell are treated with siNDUFA4L2, a lower vascular reaction is detectable (B). NDUFA4L2 has a role in RCC resistance to cisplatin (CDDP)-induced cytotoxicity (C). The death rate of treated tumor cells (tumor+ siNDUFA4L2+CDDP) is significantly higher than that of untreated cells (tumor+CDDP) (P<0.001). No difference is observed in normal cells. MTT assay reveals significantly decreased cell viability when RCC cells are treated with siNDUFA4L2 before cisplatin incubation (C). RCC cells exhibit reduced levels of mitochondrial DNA, and produced lower levels of ATP, as compared to normal cells. These levels are rescued when cancer cells are treated with siNDUFA4L2 (D). NDUFA4L2 specifically inhibits mitochondrial complex I but not complex IV activity (D).