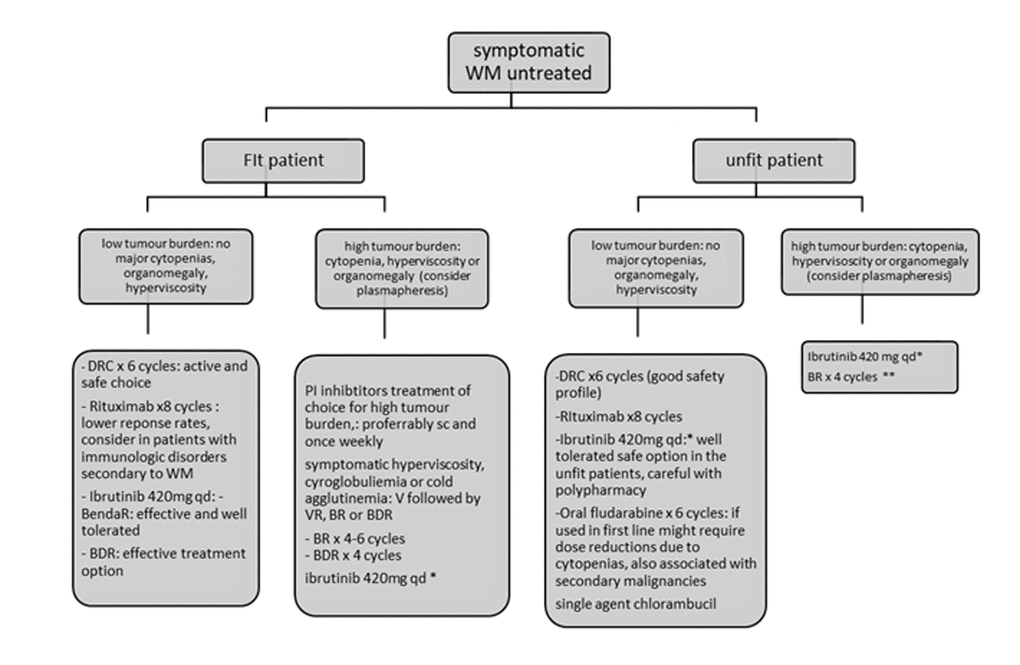
Figure 1.Recommendations for the treatment of newly diagnosed patients with Waldenstrom’s Macroglobulinemia. Figure adjusted from ESMO guidelines for WM 2018 and EMN recommendations for treatment of rare plasma cell dyscrasias. *Approved in USA by FDA for first line and only for patients unfit for immunochemotherapy in Europe by EMA. ** BR for unfit patients may require dose reductions for bendamustine and use of G-CSF and antibiotic prophylaxis. BDR: bortezomib, dexamethasone, rituximab, BR: bendamustine, rituximab; DRC: dexamethasone, rituximab, cyclophosphamide; AF: atrial fibrillation, V: bortezomib.