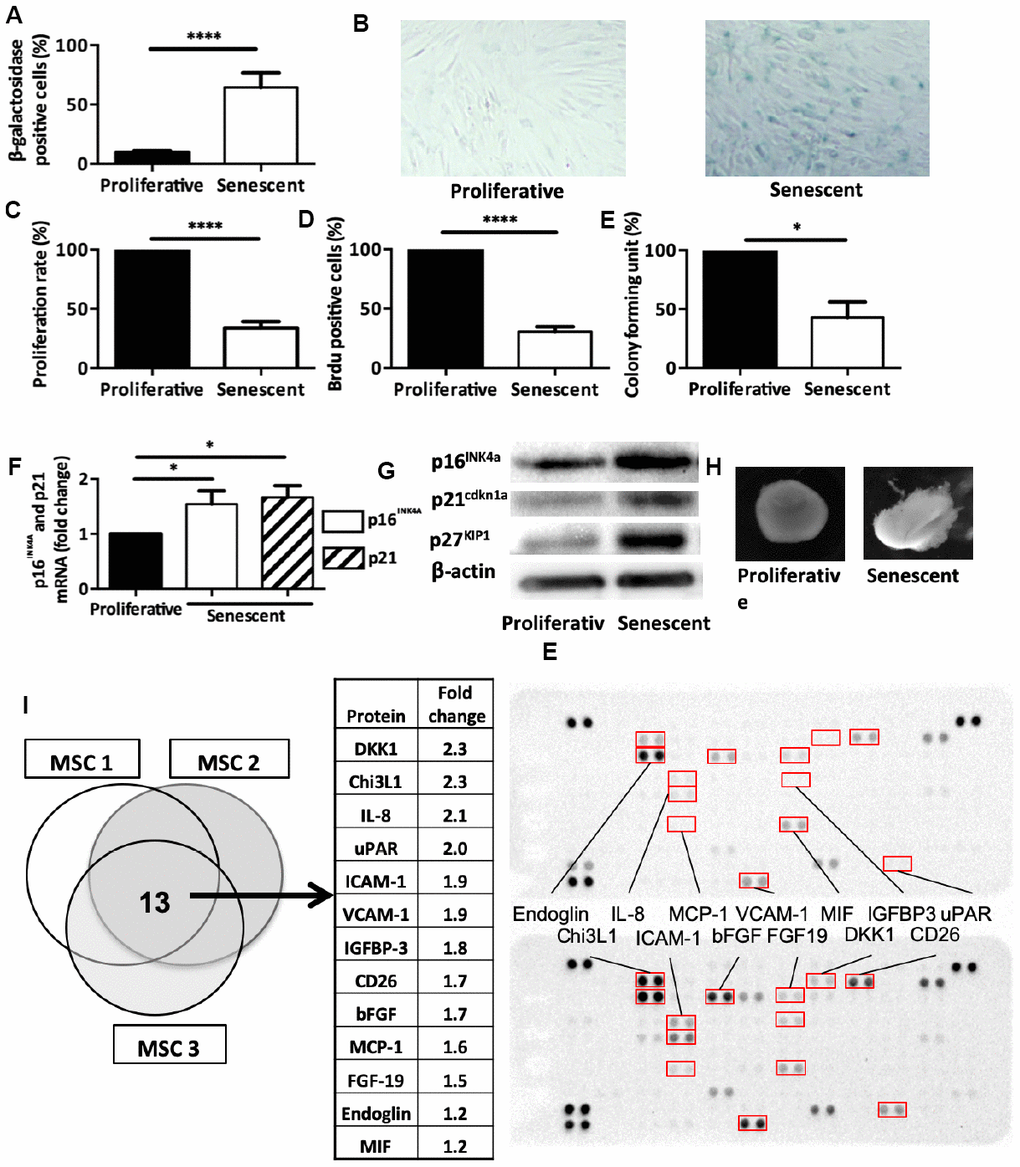
Figure 2.Senescence modulates MSCs intrinsic properties. (A) Beta-galactosidase staining in human MSCs at day 14 after DNA damaged-induced senescence (Senescent) or not (Proliferative). Data are the mean ± SEM (n=5); ****=p<0.0001. (B) Representative images of beta-galactosidase staining in proliferative and senescent human MSCs. (C) Proliferation rate (mean ± SEM) in proliferative and senescent human MSCs (n=6); ****=p<0.0001. (D) BrdU incorporation in proliferative and senescent human MSCs. BrdU-positive cells relative to all DAPI-positive cells were counted using an optical microscope (mean ± SEM; n=5). ****=p<0.0001. (E) Colony forming units in proliferative and senescent human MSCs (mean ± SEM; n=3); *=p<0.05. (F) p16INK4a and p21cdkn1a mRNA expression in human MSCs at day 14 after DNA damaged-induced senescence (Senescent) or not (Proliferative) by RT-qPCR. Data are the fold change relative to proliferative cells (mean ± SEM; n=4 for each condition); *=p<0.05. (G) p16INK4a, p21cdkn1a and p27KIP1 protein expression in human MSCs at day 14 after DNA damaged-induced senescence (Senescent) or not (Proliferative) by western blotting. Representative images of MSCs from n=3 independent donors. (H) Representative images of one cartilage pellet after chondrogenesis induction in proliferating and senescent human MSCs (from n=3). (I) Protein expression profiles of total cell extracts from senescent (bottom) and proliferating (top) human MSCs from three different healthy donors. The table showed the 13 proteins that were overexpressed in all three senescent MSC samples.