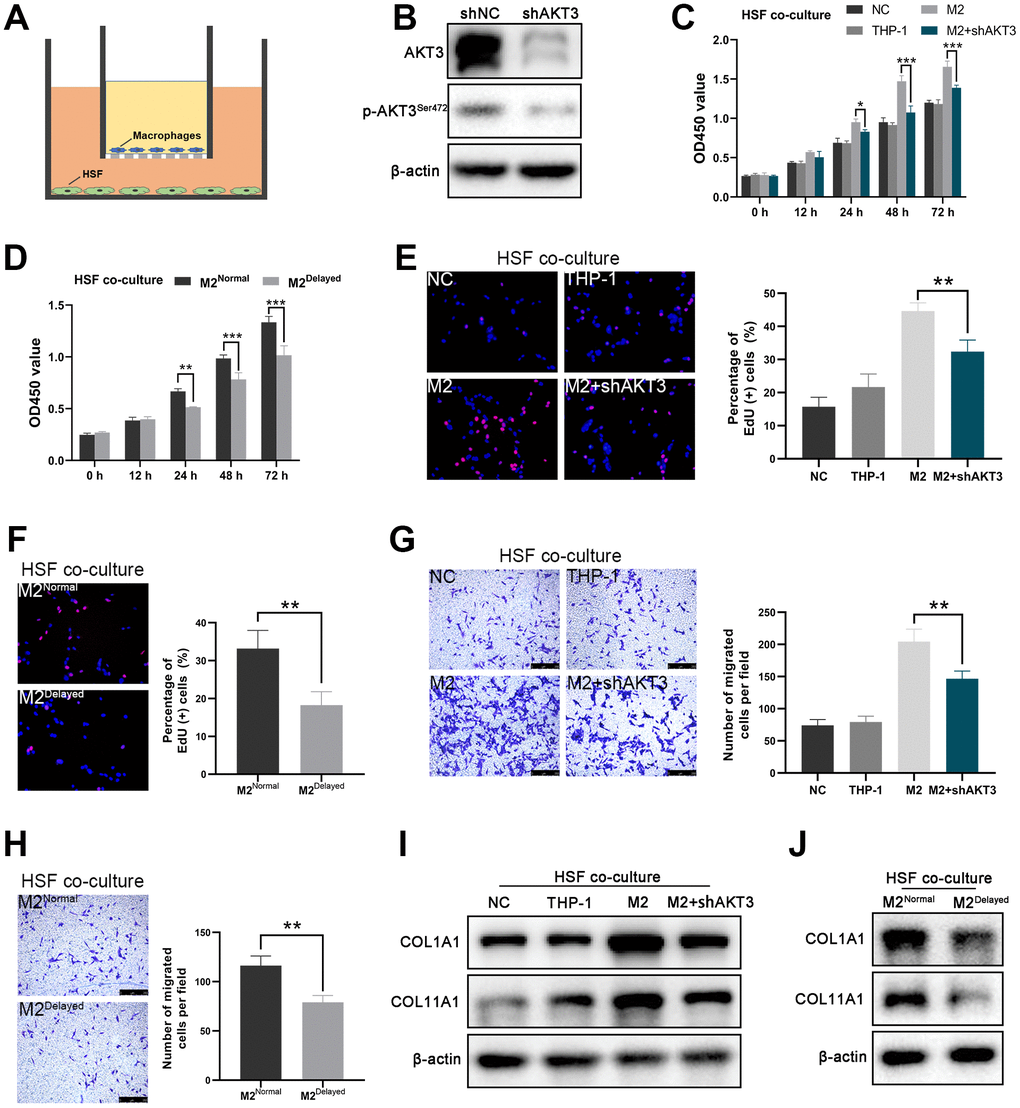
Figure 5.AKT3 knockdown in M2 macrophages suppressed proliferation and migration as well as COL1A1 and COL11A1 expression ex vivo. (A) Schematic of the M2 macrophage-HSF co-culture model. (B) Total AKT3 and associated phosphorylated AKT3Ser472 levels in THP-1-derived M2 macrophages following AKT3 knockdown. (C–D) CCK-8 assay of the co-culture model. (C) Proliferation of co-cultured HSFs was impaired following AKT3 knockdown in THP-1-derived M2 macrophages. (D) M2 macrophages isolated from delayed cutaneous wound tissue also lost their ability to facilitate HSF proliferation compared to M2 macrophages derived from normal wound tissue. (E, F) EdU assay of the co-culture model. (E) DNA replication induced by M2 macrophages in HSFs was abrogated by AKT3 knockdown in these macrophages. (F) M2 macrophages from delayed cutaneous wound tissue were incapable of promoting HSF DNA replication. (G, H) Transwell migration assay of the co-culture model. (G) HSF migration was impaired after co-culture with AKT3 knockdown in THP-1-derived M2 macrophages. (H) M2 macrophages isolated from delayed cutaneous wound tissue could not promote HSF migration. (I) COL1A1 and COL11A1 protein levels were increased in HSFs co-cultured with THP-1-derived M2 macrophages. AKT3 knockdown in the M2 macrophages decreased COL1A1 and COL11A1 expression in the co-cultured HSFs. (J) M2 macrophages from delayed cutaneous wound tissue were incapable of inducing COL1A1 and COL11A1 expression in co-cultured HSFs compared to normal wound tissue-derived M2 macrophages. All the experiments were repeated at least three times.