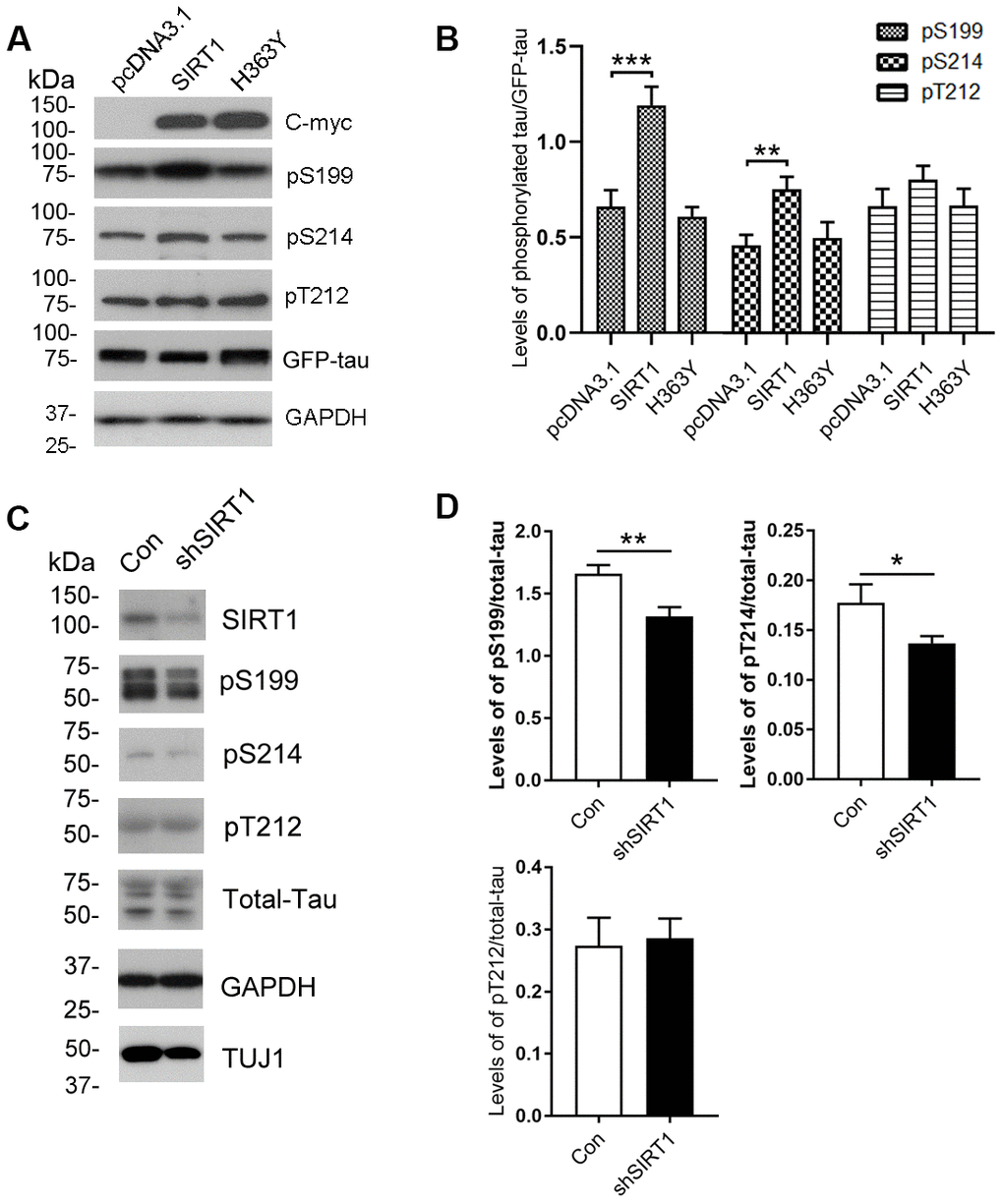
Figure 2.Changes of site-specific phosphorylation levels of tau in HEK-293A cells and primary cortical neurons. (A) The levels of total tau and the indicated site-specific phosphorylation levels of tau in the extracts of HEK-293A cells transfected with GFP-tau441 together with SIRT1 or H363Y were analyzed by western blot developed with anti-GFP antibody and with several phosphorylation-dependent/site-specific tau antibodies shown in right side of the panel. (B) Blots in panel A were quantified after normalization with the GFP-tau level, and the relative levels of site-specific tau phosphorylation are shown as mean ± S.D. (n=3), **, p < 0.01; ***, p < 0.001. (C) The levels of total tau and tau phosphorylated at the indicated phosphorylation sites in the extracts of cortical neurons of E18 rats were analyzed by western blots developed with R134d against total tau and with several phosphorylation-dependent/site-specific tau antibodies shown in right side of the panel. Tuj1 was used as a neuronal cell marker for western blot. The cortical neurons of E18 rats were infected with lentiviral-shSIRT1 or its empty vectors for 3 days to knockdown the endogenous expression level of SIRT1. The virus containing empty vectors were used as controls. (D) Blots in panel C were quantified after normalization with the total tau level, and the relative levels of site-specific tau phosphorylation are shown as mean ± S.D. (n=3), *, p < 0.05, **, p < 0.01.