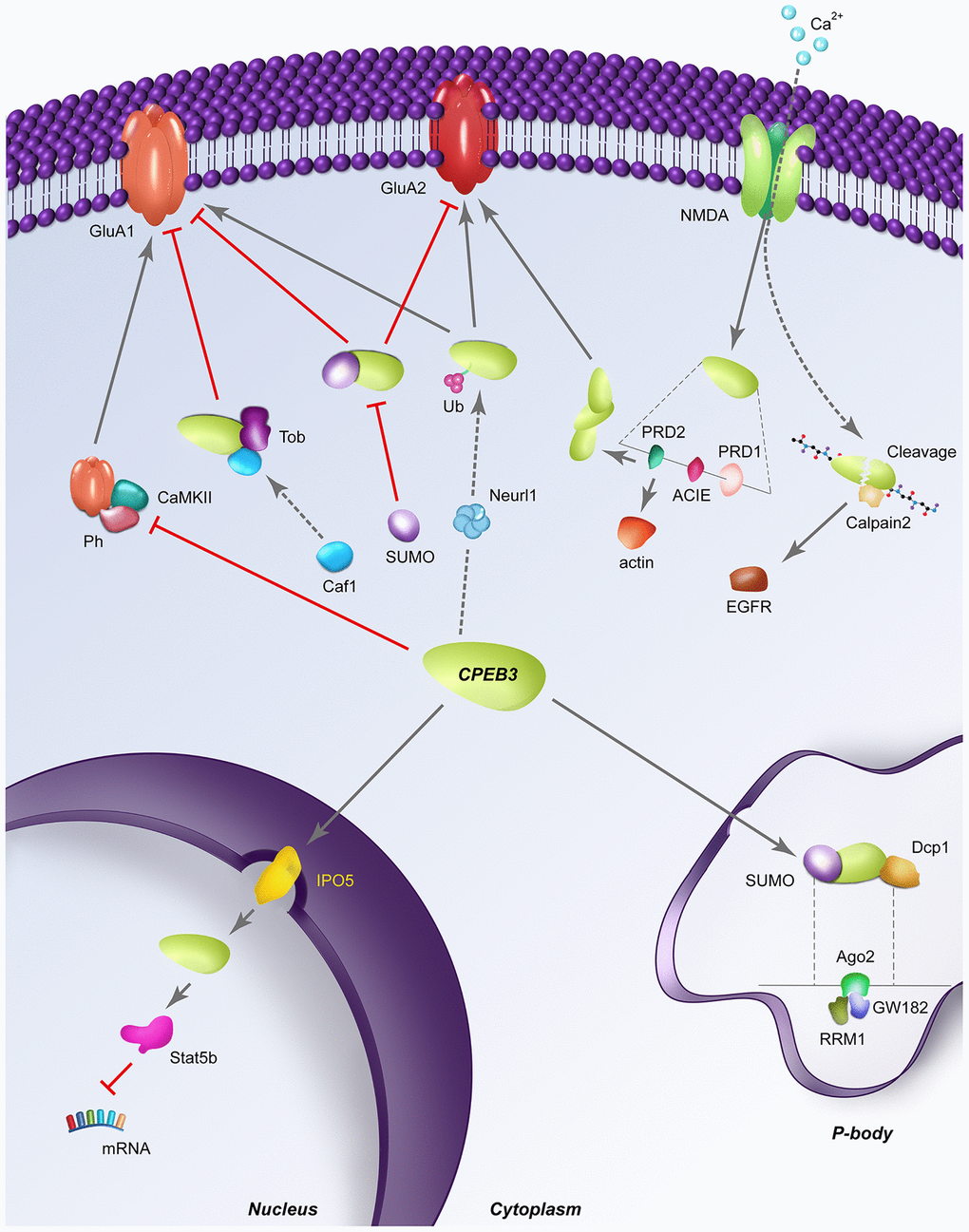
Figure 3.Dynamic regulatory pathways of CPEB3. Red perpendicular lines represent an inhibitory relationship; Arrows indicate a stimulatory relationship; SUMO: sumoylation; Ub: ubiquitination; Ph: phosphorylation; RRM1: RNA recognition motif 1; dashed lines indicate a structural expansion of a portion of the CPEB3 gene is located below. (A) CPEB3 inhibits the NMDA-dependent phosphorylation by CAMKII, allowing the expression of CPEB3 targets in a stimulation-dependent manner. (B) In its basal state, Tob binds CPEB3, recruiting the deadenylase Caf1 for the deadenylation of CPEB3 targets like GluA1. (C) SUMOylated CPEB3 (depicted in the cytoplasm) inhibits the expression of downstream targets GluA1 and GluA2; Sumoylated CPEB3 also downregulates further CPEB3 sumoylation in a negative feedback loop. SUMOylated CPEB3 (depicted in P body) is associated with the P-body protein mRNA de-capping enzyme 1 (Dcp1); sequestration of CPEB3 to P-bodies is specifically driven by the interaction of the RRM1 domain of CPEB3 to Ago2 and GW182 (D) Neurl1 induces CPEB3 monoubiquitination, inversing the basal repressive role of CPEB3 on targets like GluA2. (E) NMDA stimulation induces CPEB3 activation translation of actin through direct binding of the actin cytoskeleton interaction element (ACIE). The PRD1 domain flanking the ACIE promotes aggregation of CPEB3s to one another, further promoting the translation of other CPEB3 targets like GluA2. (F) Stimulation-induced calcium influx triggers the cleavage of CPEB3 by calpain 2, leading to proteolysis of the N-terminal repression motif to activate translation of CPEB3 targets like EGFR. (G) Nuclear translocation of CPEB3 from the cytoplasm occurs through the karyopherins IPO5, after which CPEB3 associates with the transcription factor Stat5b to downregulate transcription of Stat5b targets like EGFR.