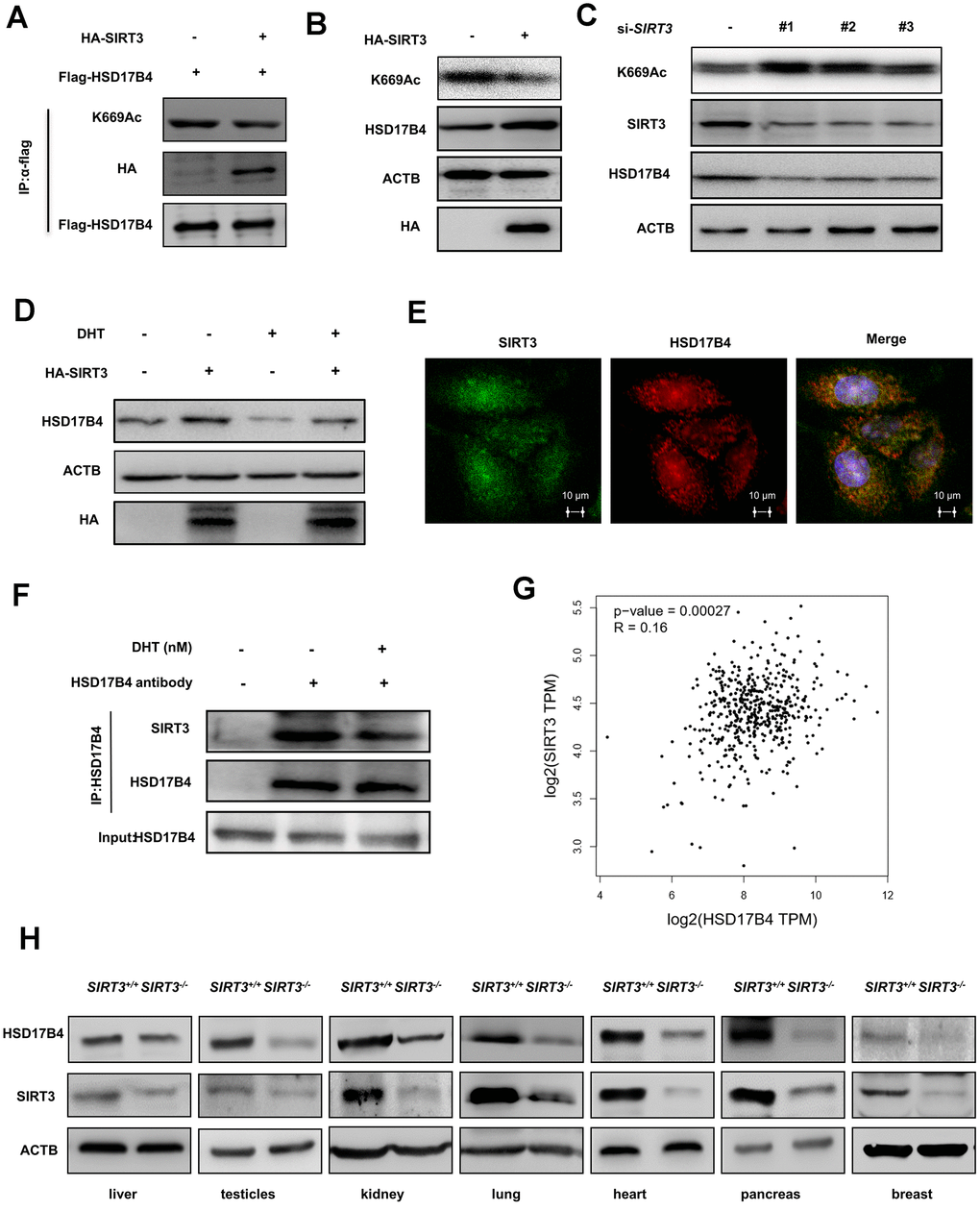
Figure 6.SIRT3 decreases the K669 acetylation level and stabilizes the protein level of HSD17B4. (A) SIRT3 overexpression decreases HSD17B4 acetylation. HA-tagged SIRT3 plasmid was cotransfected with FLAG-tagged HSD17B4 plasmid into LNCaP cells. Proteins were immunoprecipitated for western blotting. (B) SIRT3 overexpression causes accumulation of HSD17B4 protein. LNCaP cells were transfected with HA-SIRT3, and the protein levels of endogenous HSD17B4 were determined by western blotting. (C) SIRT3 knockdown decreases HSD17B4 protein. LNCaP cells were transfected with siSIRT3 or control. Cells were harvested 48 h after transfection, and cell lysates were analyzed by western blotting. (D) SIRT3 can rescue the protein levels of HSD17B4 that are decreased by DHT. LNCaP cells were transfected with HA-SIRT3 and untreated or treated with DHT. HSD17B4 protein levels were determined by western blotting. (E) SIRT3 colocalizes with HSD17B4. Cultured LNCaP cells were paraformaldehyde fixed, blocked, and processed for double immunofluorescence with antibodies against SIRT3 (green) and HSD17B4 (red). Merged images of both channels are shown on the right. Bar: 10 μm. (F) DHT decreases endogenous HSD17B4 binding with SIRT3. LNCaP cells were cultured with or without DHT for 24 h before harvest. The interaction between endogenous HSD17B4 and SIRT3 was determined by co-IP and western blotting. (G) SIRT3 is positively correlated with HSD17B4 in PCa. The correlation between the mRNA expression levels of SIRT3 and HSD17B4 in patients with PCa was analyzed using the public web server GEPIA. (H) The expression of SIRT3 and HSD17B4 in the SIRT3 knockout mouse model. Whole cell lysates were prepared from different mouse tissues, followed by western blotting analysis. Data are shown as the mean ± SD (n = 3) or typical photographs of one representative experiment. Similar results were obtained in three independent experiments. *p < 0.05, **p < 0.01, ***p <0.001.