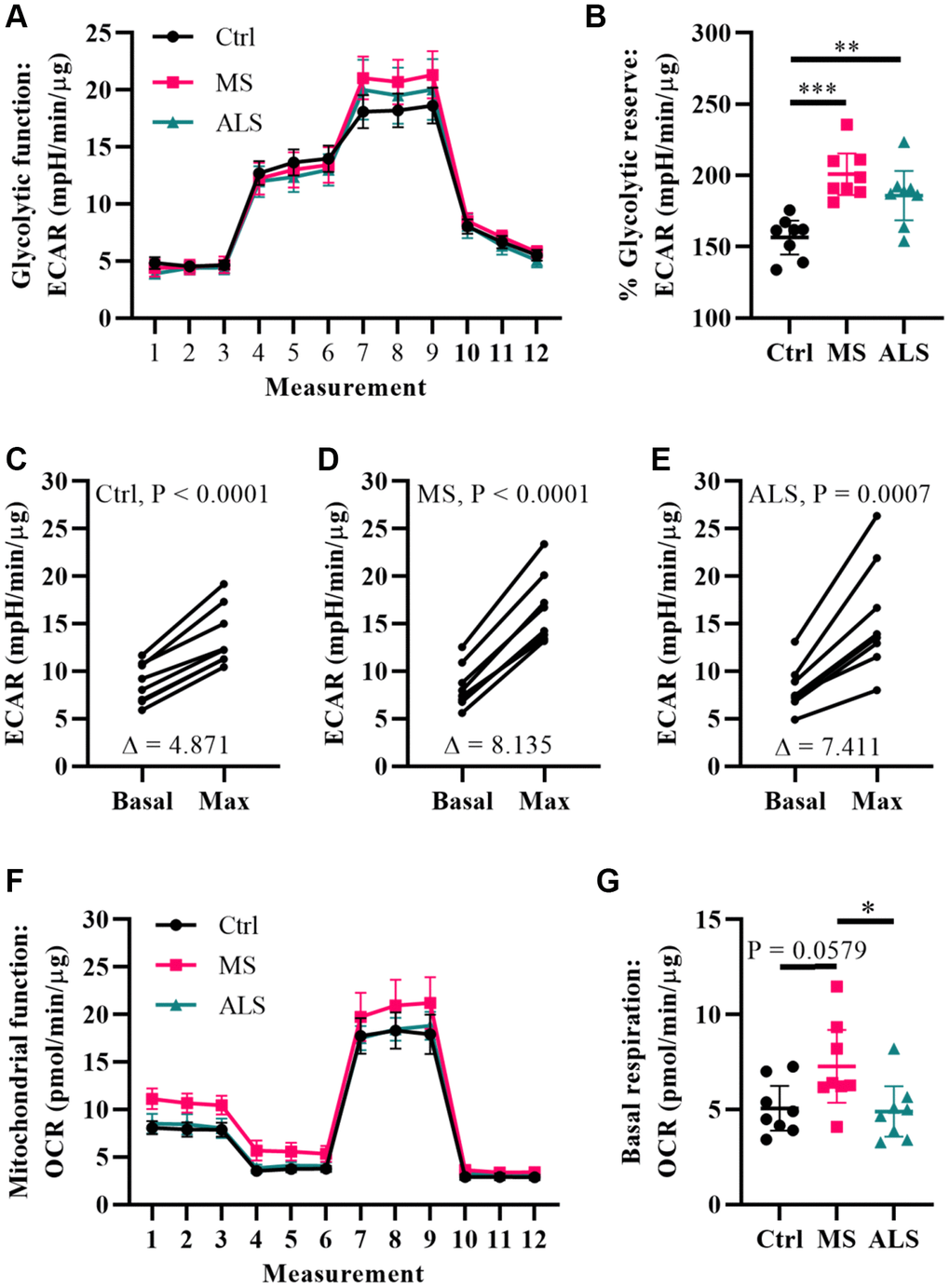
Figure 4.Multiple sclerosis skin fibroblasts have altered bioenergetics. (A) Glycolytic function of MS, ALS, and control skin fibroblasts were assayed using the Seahorse Glycolytic Stress Test. The cells were incubated in glucose-free media containing 2 mM glutamine for one hour prior to measuring glycolytic function. Sequential addition of 5.5 mM glucose, 1.5 μM oligomycin, and 50 mM 2-deoxy-D-glucose were added to the cells while the ECAR was monitored. (B) The percent glycolytic reserve (glycolytic capacity/basal glycolysis) increase after treatment with oligomycin. (C–E) Paired analysis of skin fibroblasts treated with oligomycin reflects individual changes from basal glycolysis to maximum glycolytic capacity. The average change (Δ) is indicated. (F) Mitochondrial function of MS, ALS, and control skin fibroblasts were assayed using the Seahorse Mito Stress Test. All cells were incubated in assay medium containing 5.5 mM glucose and 2 mM glutamine for one hour prior to performing the assay. The OCR was monitored during sequential injection of 1.5 μM oligomycin, 1 μM FCCP, and 1 μM rotenone plus 1μM antimycin A. (G) The average basal respiration detected in skin fibroblasts. (A and F) Each dot represents the average rates measured ± SEM from all eight unique cell lines within each indicated group. (B and G) Each dot represents a unique patient-derived skin fibroblast (n = 8 per group). The graphs show the average with the 95% confidence interval. Statistical significance was determined using one-way ANOVA post hoc Tukey test. *P < 0.05, **P < 0.01, ***P < 0.001. (C–E) Significance was determined using paired t test analysis (two tailed) using averaged data. All averages were determined from two independent experiments each containing triplicates using all three measurements per injection group. All data was normalized to total protein content (μg).