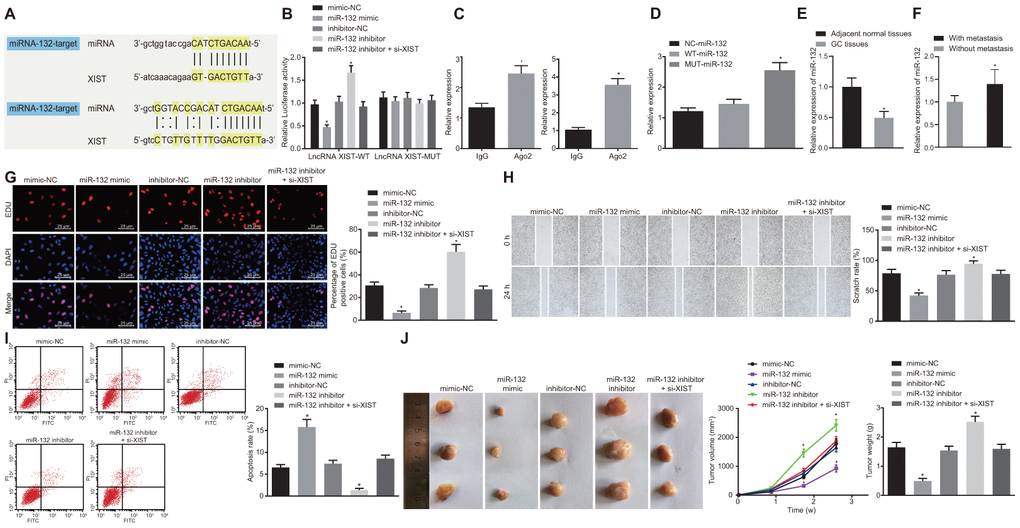
Figure 3.MiR-132 specifically binds to lncRNA XIST and its overexpression restrains tumor formation ability, cell proliferation and migration while promoting apoptosis in gastric cancer. (A) Binding region between miR-132 and lncRNA XIST; (B) Relative luciferase activity of miR-132 and lncRNA XIST; (C) Binding of lncRNA XIST to miR-132 and Ago2 detected by RIP assay; (D) RNA pull-down assay suggested binding relationship between lncRNA XIST and miR-132; (E) Relative expression of miR-132 in gastric cancer tissues (n = 65) and the adjacent normal tissues determined by RT-qPCR; *, p < 0.05 compared with adjacent normal tissues; (F) Relative expression of miR-132 in gastric cancer tissues from patients with metastasis (n = 45) and without metastasis (n = 20) determined by RT-qPCR; *, p < 0.05 compared with gastric cancer tissues from patients without metastasis; In the panels (G–I) SGC7901 cells were transfected with miR-132 mimic or inhibitor or co-transfected with miR-132 inhibitor and si-XIST. (G) Cell proliferation ability evaluated by EdU assay (× 400); (H) Cell migration ability assessed by scratch test (× 40); (I) Cell apoptosis ability in each group detected by flow cytometry; (J) The effect of miR-132 and lncRNA XIST on tumor formation in nude mice injected with the stably transfected SGC7901 cells. All measurement data and statistical results were expressed as mean ± standard deviation. A value of p < 0.05 was considered as statistically significant and a value of p < 0.001 was considered statistically highly significant; *, p < 0.05 compared with the NC group; comparisons between two groups were analyzed by t-test, while comparison among multiple groups were analyzed using one-way ANOVA. The experiment was repeated 3 times.