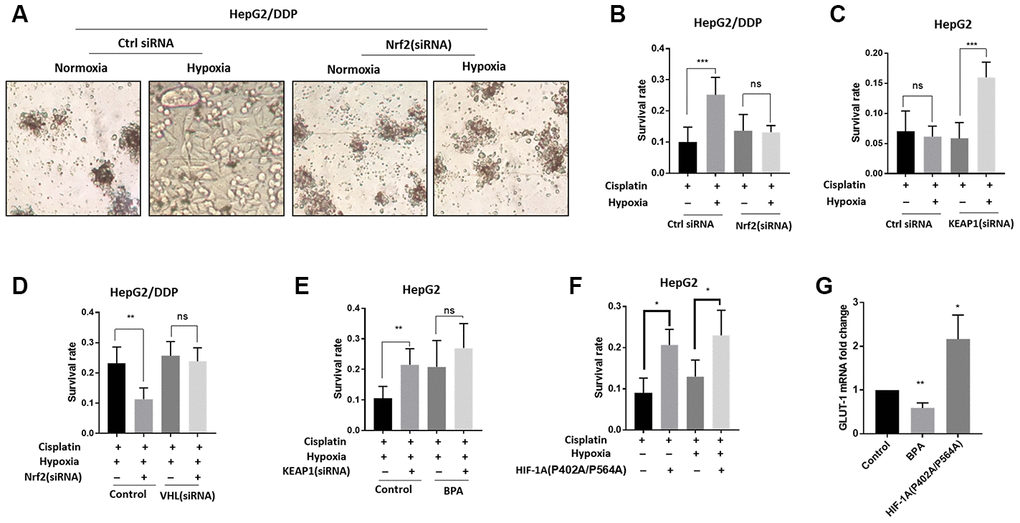
Figure 3.Nrf2 promotes hypoxia-induced cisplatin resistance in HepG2/DDP cells through HIF-1α. (A) Nrf2 knockdown blocked the hypoxia-induced cisplatin-resistance in HepG2/DDP cells. Cells were transfected with Nrf2-specific siRNA for 24 hours, then incubated under normoxic (21% O2) or hypoxic conditions (5% O2) for 36 hours in the presence of 10ug/ml cisplatin. Cells were imaged in the 96-well plate. Representative images were shown. (B) CellTiter-Glo viability assay for cells shown in (A). HepG2/DDP cells treated with cisplatin for 36 hours or non-treated controls were directly lyzed with CellTiter-Glo reagent and read by luminescence reader. Data collected from 3 biological repeats were normalized to non-treated control. Student’s t-test was performed to evaluate the statistical significance. ns, not significant, ***P<0.0001. (C) Nrf2 activation by KEAP1 knockdown promoted hypoxia-induced cisplatin resistance in HepG2 cells. KEAP1 was knocked down as in (A) and cell viability was measured as in (B). Student’s t-test: ns, not significant, ***P<0.0001. (D) HIF-1α activation prevented Nrf2 knockdown from sensitizing HepG2/DDP cells to cisplatin. Cells were transfected with indicated siRNA and treated with 5% O2 as in (A). CellTiter-Glo viability assay was conducted as in (B) for 3 biological replicates. Student’s t-test: **P<0.001, ns, not significant. (E) HIF-1α inhibition prevented KEAP1 knockdown from increasing cisplatin resistance in HepG2 cells. HIF-1α was inhibited with an established chemical inhibitor dimethyl-bisphenol A (BPA). HepG2 cells with or without KEAP1 knockdown were treated with cisplatin and BPA as indicated and incubated under normoxia or hypoxia for 36 hours. CellTiter-Glo viability assay was performed as in (B) for 3 biological replicates. Student’s t-test: **P<0.001, ns, not significant. 100uM BPA increased cisplatin resistance for unknown reasons. (F) Expression of constitutively active HIF-1α increased cisplatin resistance in HepG2 cells. Cells were transfected with plasmid expressing HIF-1α (P402A/P564A) for 24 hours then treated with cisplatin and hypoxia as indicated for 36 hours. Viability was measured by CellTiter-Glo. Student’s t-test: **P<0.001, ns, not significant. (G) HIF-1α transcriptional activity was down-regulated and upregulated by BPA and HIF-1α (P402A/P564A), respectively. HepG2 cells were treated with BPA or transfected with HIF-1α (P402A/P564A). Total mRNA was extracted and reversed transcribed. GLUT-1 cDNA was examined by RT-qPCR. Data from 3 experiments were plotted and analyzed with Student’s t-test: *P<0.01, **P<0.001.