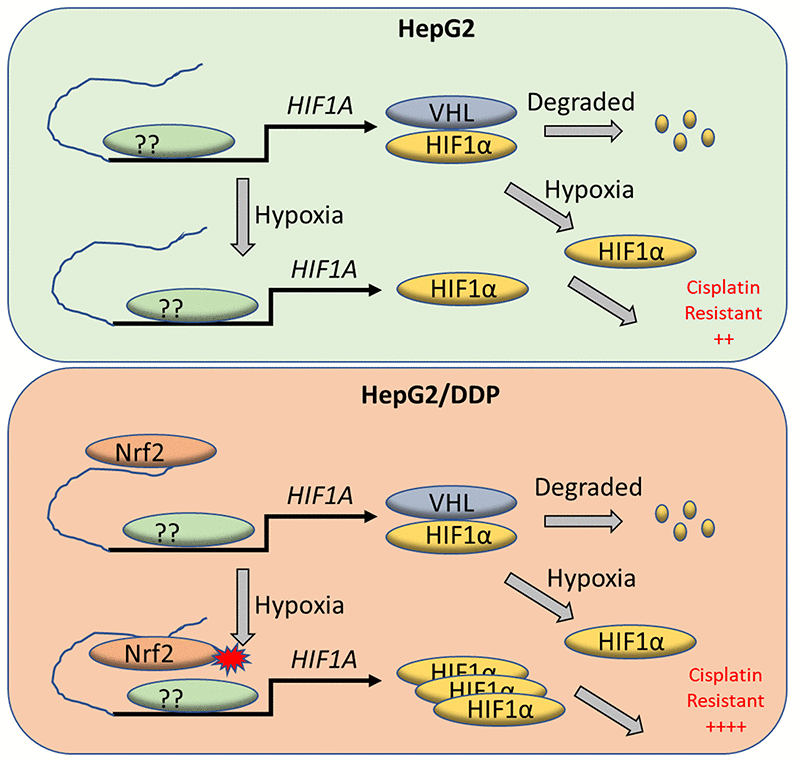
Figure 5.A working model for Nrf2 regulation of HIF-1α for cisplatin-resistance in HepG2/DDP cells under hypoxia. Tumor micro-environment is hypoxic, which contributes to cancerous progression and chemo-resistance through complex mechanisms. HIF-1α protein is normally translated then rapidly degraded by proteasome through VHL-mediated ubiquitination. Under hypoxia conditions however, HIF-1α degradation is inhibited and accumulates in the nucleus to induce adaptive transcriptional programs, leading to cisplatin resistance. We find in the cisplatin-resistant hepatocellular carcinoma cell line HepG2/DDP that Nrf2 binds to HIF-1α enhancer. The enhancer-bound Nrf2 serves to augment the transcription of HIF-1α by other transcription factors, leading to increased cisplatin resistance under hypoxic stress.