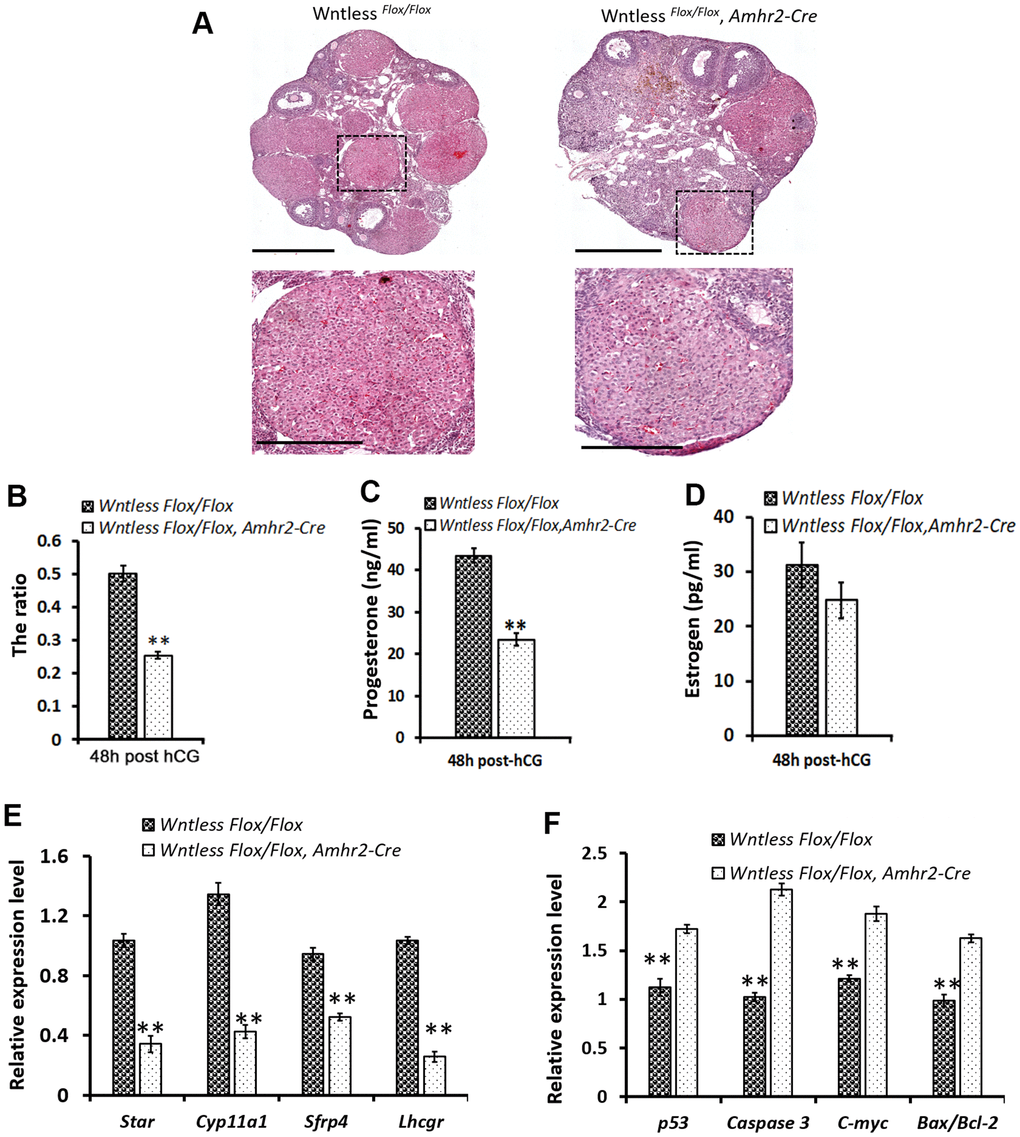
Figure 5.Impaired CL formation in GC-specific Wntless knockout mice. (A) Top row, representative images of control and mutant ovarian morphology at 48 h post-hCG treatment, scale bar = 500 μm. Second row, magnified images of CL, scale bar = 250 μm (B). The area ratio of CL to ovary for Wntless Flox/Flox (n=6) and Wntless Flox/Flox, Amhr2-Cre (n=6) mice at 48 h post-hCG treatment. Blood serum progesterone (C) and estradiol (D) levels in Wntless Flox/Flox (n=6) and Wntless Flox/Flox, Amhr2-Cre (n=6) mice at 48 h post-hCG treatment. Expression levels of luteal cell marker (E) and apoptotic genes (F), analyzed by qRT-PCR, in ovaries from 48 h post-hCG treated control and mutant mice. Gapdh served as the internal control gene. In B-F, **P<0.01, mean ± SEM. Experiments were replicated a minimum of 4 times.