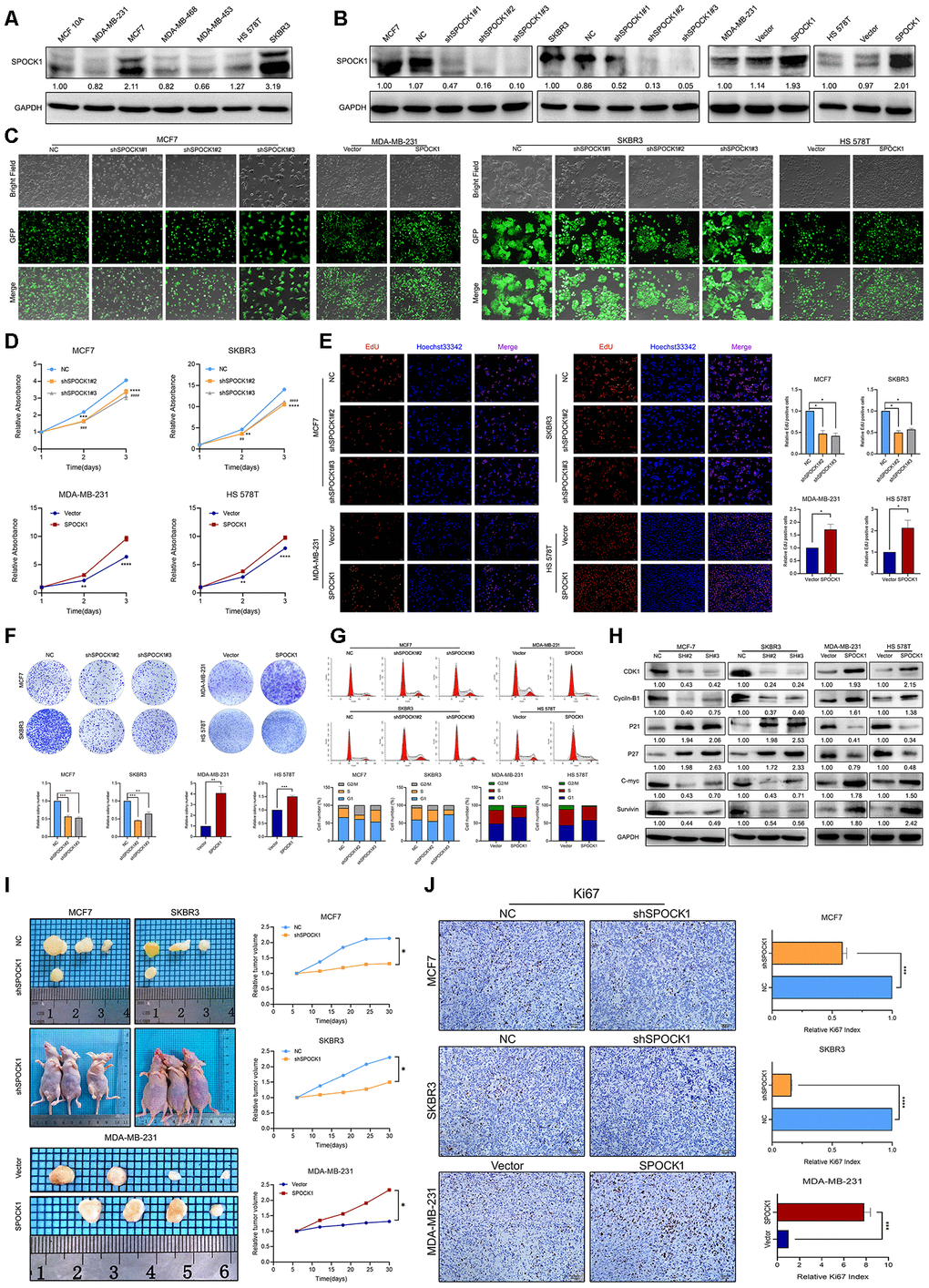
Figure 2.SPOCK1 influences BC cell growth. (A) Protein expression levels of SPOCK1 in BC cell lines as determined by western blot analysis. (B) MCF7/SKBR3 cells with SPOCK1 silencing and MDA-MB-231/HS 578T cells with SPOCK1 overexpression were established by viral transduction. The SPOCK1 levels in these established cell lines were verified by western blot analysis at 48 h after transfection. (C) Cells in bright light and GFP were captured to merge for displaying the transfection efficiency. (D) Cell viability was examined by MTT assay. (E) Results of EdU assay on BC cells. Representative photographs are shown at the original magnification, ×100. (F) Cell clonogenic capacity was measured by colony formation assay. (G) Flow-cytometry analysis was performed to detect cell cycle progression. (H) The expression of proteins related cell cycle (CDK1, Cyclin-B1, P21, P27, C-mvc and Survivin) was determined by western blot analysis. GAPDH was used as a loading control. (I) Xenograft tumors formed by injecting the indicated cells. Relative tumor volume curves were summarized in the line chart (*P<0.05). (J) IHC staining of the proliferation marker Ki67 in xenograft tumors. The relative percentage of Ki67-positive cells was summarized in the bar charts. The P values were obtained using t-tests (***P<0.001, ****P<0.0001). All results are from three independent experiments. The error bars represent the SD.