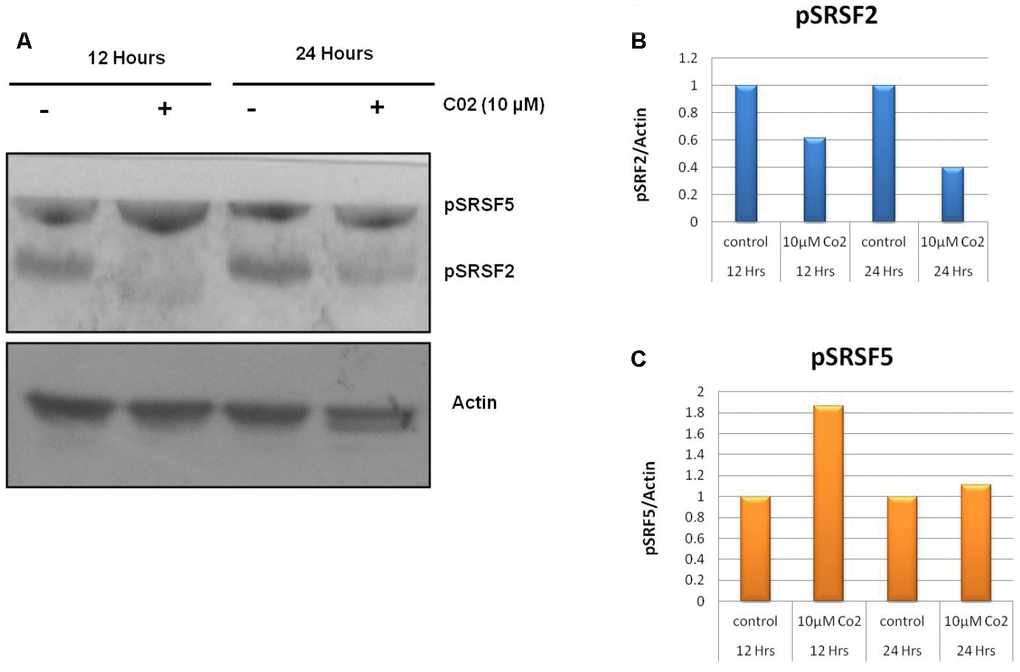
Figure 3.Effects of compound C02 on the SRPK1 mediated phosphorylation of SR proteins. (A) Western blotting analysis after treatment with compound C02 (10 μM) and negative control (DMSO) for 12 and 24 h on Jurkat cells is shown. SR protein phosphorylation was detected using mAb1H4, which recognizes phosphorylated serine arginine epitopes present on SR factors. The blot was re-probed with actin to be used as endogenous control. (B, C) The protein bands were also determined relative to the endogenous control Actin by using densitometry software.