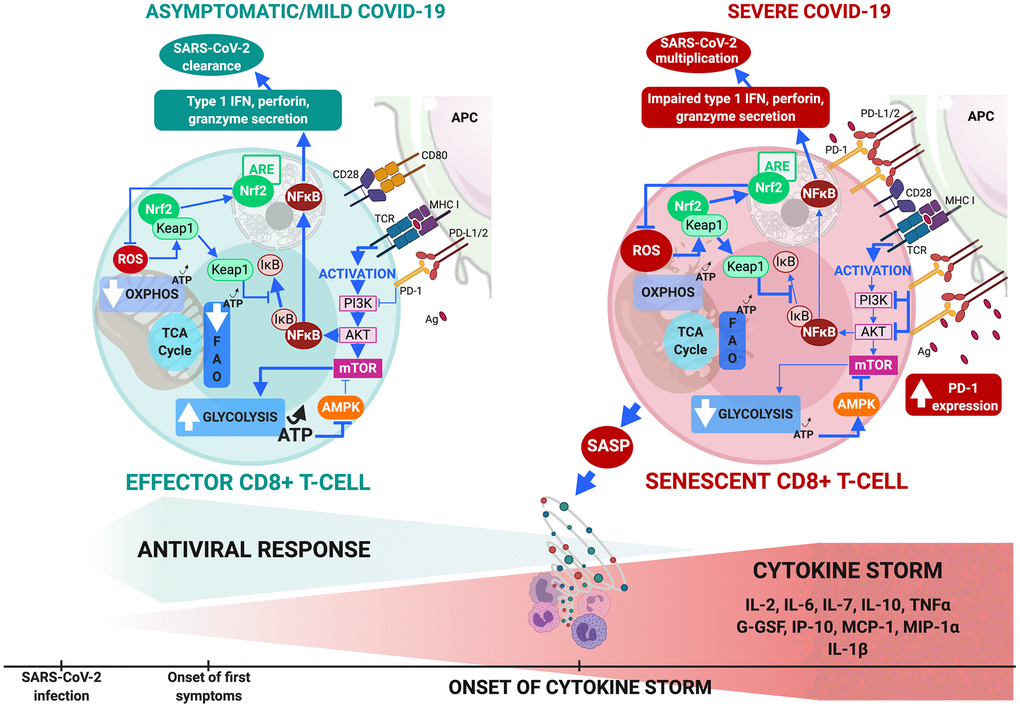
Figure 2.CD8+ T-cell metabolism in COVID-19. (A) In comorbidity-free patients developing asymptomatic/mild symptomatic forms of COVID-19, cytotoxic immune response mediated by effector CD8+ T-cells results in eradication of virus and patient recovery. Normal antigen levels in antigen-presenting cells and cytokine levels (interleukin IL-2 released by helper CD4+ T-cells) stimulate TCRs and co-receptors such as CD28, thus enhancing mTOR signaling via PI3K and protein kinase B that increases glycolysis. Cells shift from OXPHOS/FAO to glycolytic-based metabolism, whereby anabolic processes activate effector cells to clear infection. This includes production of cytotoxic factors (type 1 Interferon, granzyme, perforin) and enhanced proliferation. Massive increase in glycolysis results in production of ATP (less than OXPHOS but sufficient to inhibit AMPK, preventing mTOR pathway blockade. ROS production activates Nrf2, reducing inflammation and apoptosis by inhibiting NF-κB and pro-inflammatory cytokine production. (B) Aging and age-related disorders cause CD8+ T-cell senescence in severe COVID-19. Excess antigens upregulate inhibitory receptors (programmed death-1: PD-1) that block TCR activation, thus reducing signaling required for glycolytic metabolic phenotype itself crucial to proper effector functioning. Malfunction is compounded by upregulation of PD-1 expression-enhancing transcription factors, reduction in helper cell survival and proliferation signaling (IL-2), and increase in inhibitory signals. Senescent CD8+ T cells secrete SASP, paracrinely amplifying production of inflammatory cytokines and triggering cytokine storm. Massive decrease in glycolysis causes fall in ATP production and fails to sufficiently inhibit AMPK which then partially inhibits mTOR pathway. Substantial ROS production activates Nrf2 but fails to inhibit NF-κB pathway and pro-inflammatory cytokine production. These events combined make cells malfunction metabolically, inhibit cytotoxic function and exhaust the phenotype.