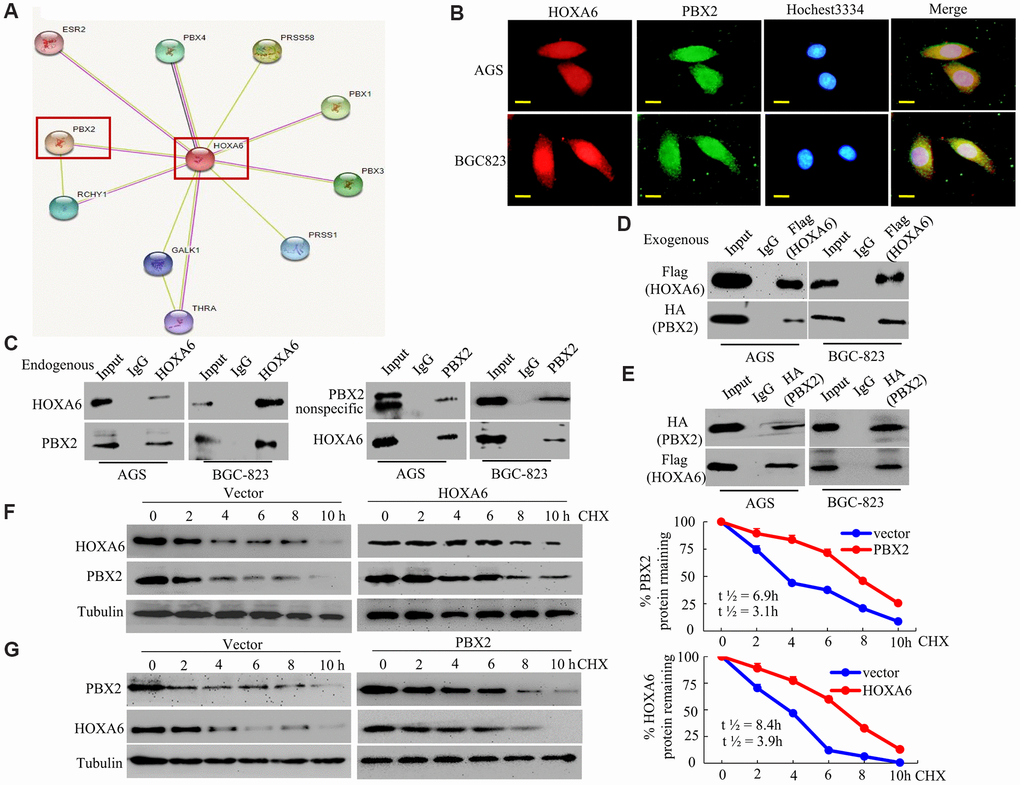
Figure 5.HOXA6 physically interacts with PBX2 in GC cells. (A) HOXA6-related protein-protein interaction (PPI) network from the STRING database. (B) Confocal microscopy detected the interaction of HOXA6 and PBX2 in GC AGS and BCG-823 cells. HOXA6 and PBX2 colocalization was observed in the nuclear, perinuclear and cytoplasmic regions of both cell lines. (C) To conduct immunoprecipitation with anti-HOXA6 (PBX2) antibody, either normal mouse IgG (nmIgG) or anti-HOXA6 (PBX2) was immunoprecipitated from whole-cell lysate antibody, followed by western blotting with anti-PBX2 (HOXA6) antibody. (D) HOXA6-Flag was co-transfected with PBX2-HA into AGS and BCG-823 cells. Whole-cell extracts were immunoprecipitated with an anti-Flag antibody and blotted with an anti-HA antibody. (E) PBX2-HA was co-transfected with HOXA6-Flag into AGS and BCG-823 cells. Anti-HA antibody was performed with immunoprecipitation, and pre-immune normal mouse immunoglobulin G (nm IgG) was designated as a reference control. Western blot analysis was used for anti-Flag antibody. (F, G) Regulation of PBX2 (HOXA6) protein stability by HOXA6 (PBX2). 50 μg/mL cycloheximide (CHX) was added to vector-transfected AGS cells overexpressing HOXA6 (PBX2) after the start of the experiment. The cells were harvested at the indicated times and lysates were prepared after the addition of cycloheximide. Western blotting was used to perform. Scale bars in B represent 25 μm.