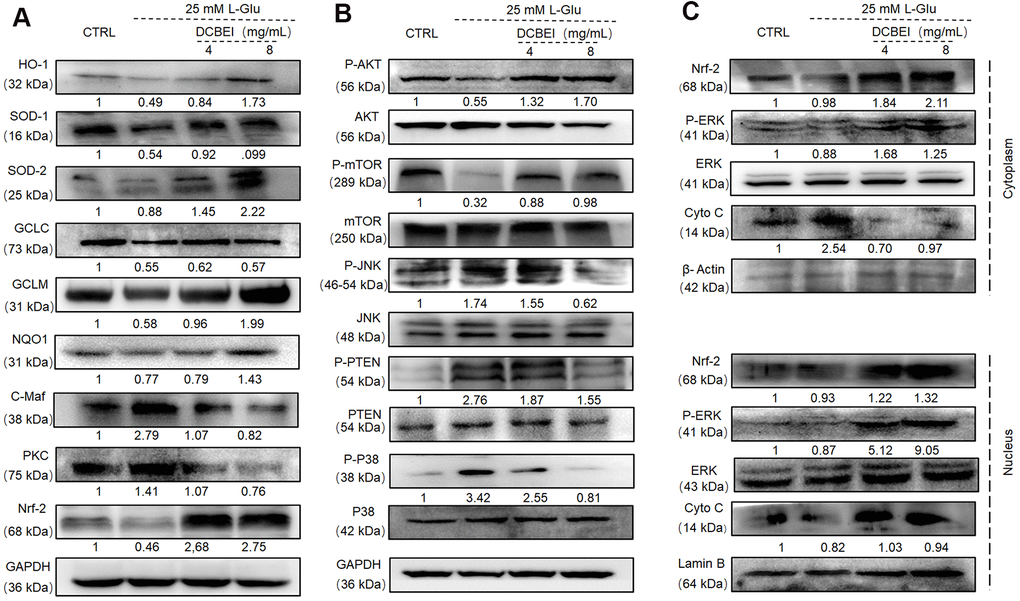
Figure 6.DCBEI ameliorates AD symptoms by reducing apoptosis, attenuating oxidative stress, and regulating neurotransmitter levels via Nrf2 signaling. (A) In L-Glu-exposed HT22 cells, DCBEI increased the expression of Nrf-2 and its downstream targets including HO-1, SOD-1, SOD-2, GCLC, GCLM and NQO1, and reduced the expression levels of C-Maf. (B) Analysis of MAPKs and Akt in L-Glu-treated HT22 cells. DCBEI increased the expression of p-AKT and p-mTOR, and reduced the expression of p-JNK, p-PTEN and p-P38 in L-Glu-treated HT22 cells. (C) DCBEI increases p-ERK levels and causes a redistribution of p-ERK, Nrf-2, and Cyto C from the cytoplasm to the nucleus. Quantification data were normalized to GAPDH and the corresponding total proteins, and are reported as fold change relative to CTRL (n = 3).