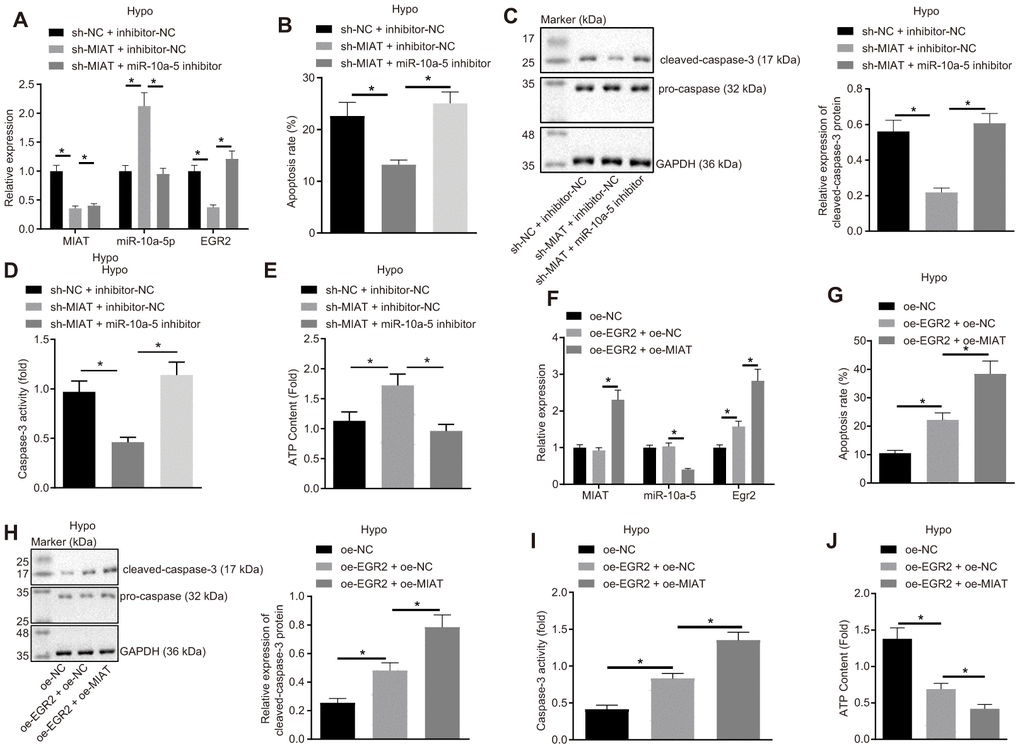
Figure 4.MIAT competitively binds to miR-10a-5p to upregulate EGR2 and further increase apoptosis of cardiomyocytes under exposure to hypoxia. (A) The expression of MIAT, miR-10a-5p and EGR2 in cardiomyocytes after inhibition of MIAT and/or miR-10a-5p determined by RT-qPCR. (B) Apoptosis of cardiomyocytes after inhibition of MIAT and/or miR-10a-5p detected by flow cytometry. (C) Western blot showing the expression patterns of cleaved-caspase-3 and caspase-3 in hypoxic cardiomyocytes after inhibition of MIAT and/or miR-10a-5p. The expression level is normalized to GAPDH. (D) The caspase-3 activity of cardiomyocytes under hypoxic conditions after inhibition of MIAT and/or miR-10a-5p. (E) The ATP content in cardiomyocytes after inhibition of MIAT and/or miR-10a-5p under hypoxic conditions. (F) The expression patterns of MIAT, miR-10a-5p and EGR2 in cardiomyocytes after MIAT and/or EGR2 overexpression determined by RT-qPCR normalized to GAPDH and U6. (G) The apoptosis of cardiomyocytes under hypoxic conditions after MIAT and/or EGR2 overexpression detected by flow cytometry. (H) The expression of cleaved-caspase-3 and pro-caspase-3 in the hypoxic cardiomyocytes after MIAT and/or EGR2 overexpression normalized to GAPDH determined by Western blot analysis. (I) The caspase-3 activity of cardiomyocytes under hypoxic conditions after MIAT and/or EGR2 overexpression. (J) The ATP content in cardiomyocytes under hypoxic conditions in response to MIAT and EGR2 overexpression relative to ATP content after EGR2 overexpression alone (fold). * p < 0.05, the above data were all measurement data, and expressed as mean ± standard deviation. The data comparison among multiple groups was performed using one-way ANOVA and Tukey’s post hoc test. The experiment was repeated 3 times independently.