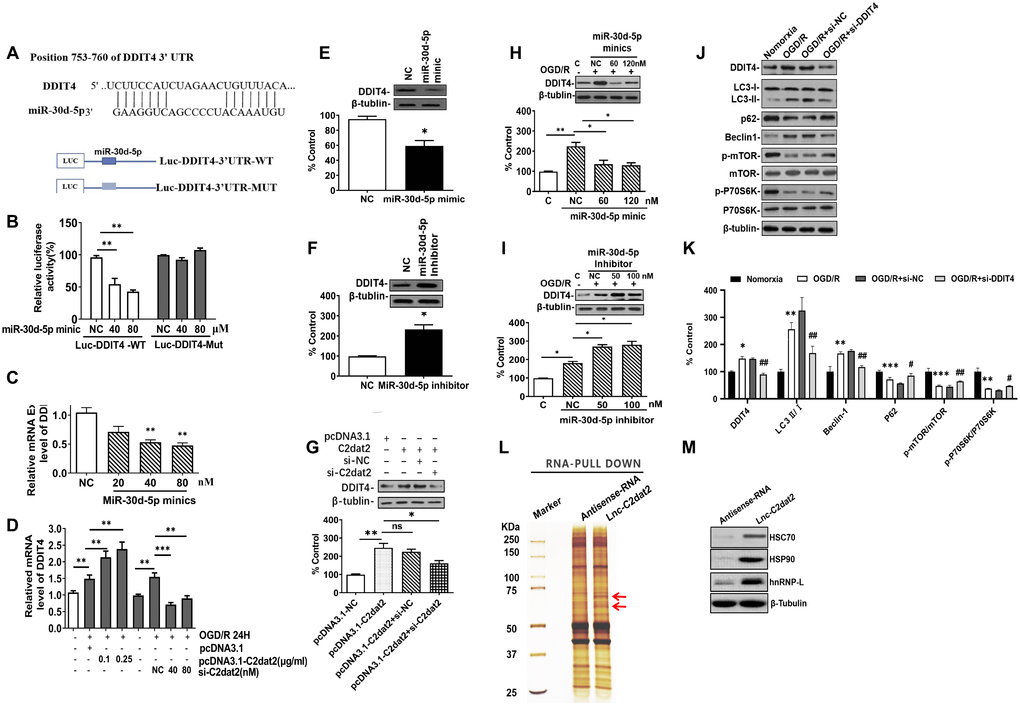
Figure 7.C2dat2 positively regulated DDIT4 expression by sponging miR-30d-5p. (A) Possible binding sites of miR-30d-5p in DDIT4 3′-UTR. (B) luc-DDIT4 3′-UTR-WT or luc-DDIT 3′-UTR-MUT plasmids were cotransfected with NC or miR-30d-5p mimic into HEK 293T cells for 24 h. Luciferase activity was determined. (C) After transfection with NC and miR-30d-5p mimic for 48 h, DDIT4 mRNA levels were detected by RT-qPCR. (D) N2a cells were treated with OGD/R for 24 h after C2dat2 overexpression or knockdown followed by RT-qPCR of DDIT4 mRNA levels. (E and F) After transfection with (E) miR-30d-5p mimic or (F) inhibitor for 48 h, DDIT4 protein levels were assessed via Western blotting. (G) After transfection with pcDNA3.1 or C2dat2 (0.1 μg/ml) and then cotransfected with C2dat2 (0.1 μg/ml) and NC or si-C2dat2 (60 nM) for 48 h, DDIT4 protein levels were assessed via Western blotting. (H and I) After transfection with NC, miR-30d-5p mimic, or inhibitor overnight, N2a cells were treated with OGD/R for 48 h. Western blotting of DDIT4 levels. *P < 0.05; **P < 0.01; ns, not significant vs. normoxia (n = 3 per group). (J) Relative protein expression levels of DDIT4, LC3, P62, Beclin-1, p-mTOR/mTOR, and p-P70S6K/P70S6K were assessed via Western blotting of N2a cells transfected with si-NT or si-DDIT4 and then treated with OGD/R for 24 h. (K) Relative protein levels were analyzed. Data are mean ± SEM. β-Tubulin was blotted as a loading control. (L) lncRNA C2dat2 binding with the HSC70/HSPA9 conjugate. RNA pull-down results of lncRNA C2dat2 via silver staining. (M) Western blotting confirmed C2dat2, HSC70, and HSP90 expression levels by RNA pull-down of the I/R tissue. *P < 0.05; ns, not significant vs. normoxia; ##P < 0.05 vs. si-NT (n = 3 per group).