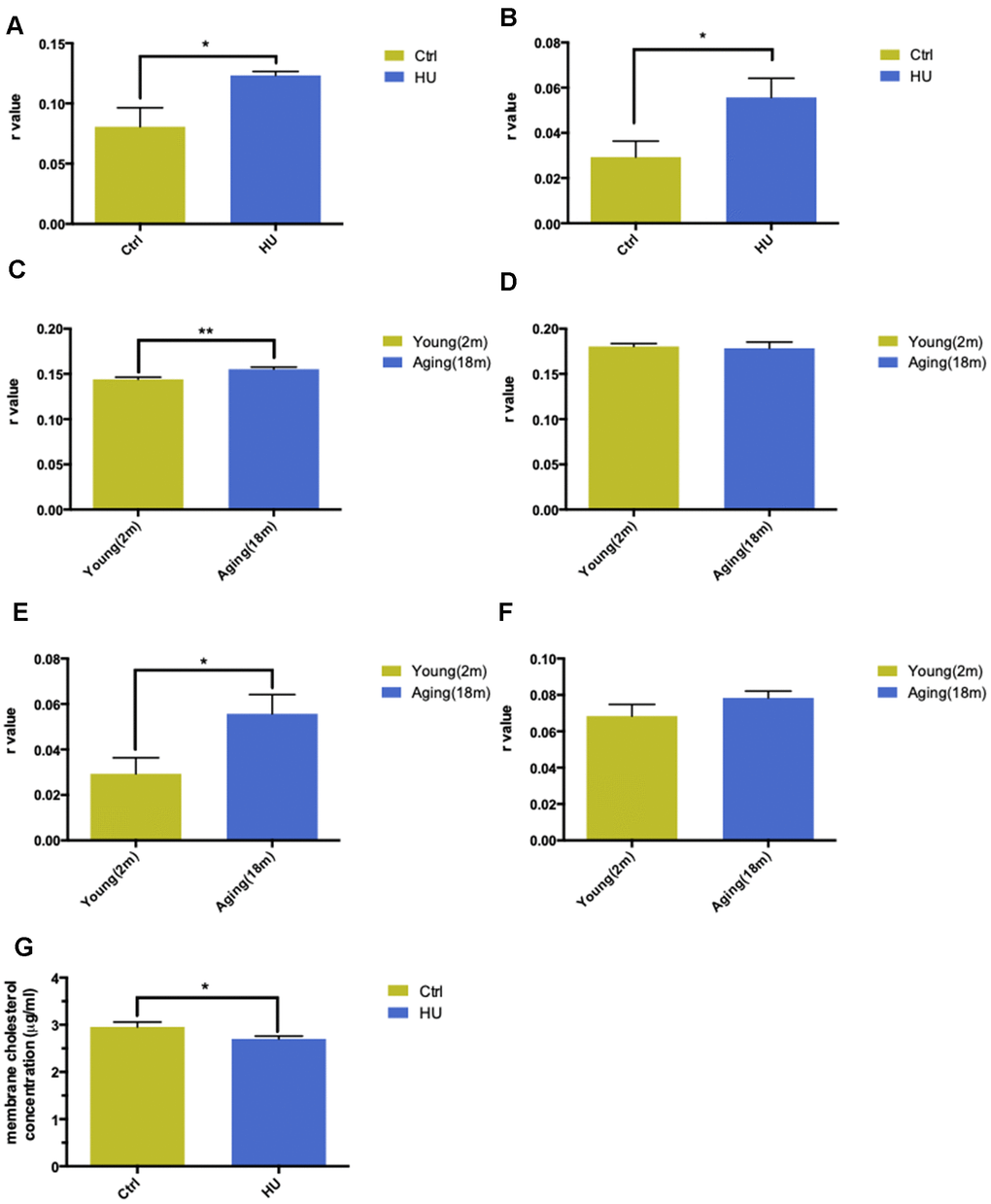
Figure 2.Validation of “membrane aging” in vivo and in vitro. Changes in membrane fluidity and cholesterol levels in membrane pellets are two main characteristics of membrane aging. Fluorescent probe polarization is one of the most direct measurements representing the alterations in membrane fluidity. HU treatment significantly increased the r value compared with the control in vitro via applying the DPH fluorescent probe (P < 0.05) (A). The same trend was observed while probing with TMA-DPH (P < 0.01) (B). The results demonstrated that HU treatment decreased neuronal membrane fluidity dramatically because the r value was inversely proportional to membrane mobility. In vivo, native membrane pellets were carefully extracted, followed by hippocampus and cortex isolation in 18-month-old male SD rats (n = 6) and 2-month-old male SD rats (n = 6). When probing with DPH in cortical membrane pellets, the r value increased significantly in the aging group compared with the young group (P < 0.01) (C). In hippocampal regions, no difference was observed between two groups (D). Similar results were obtained by applying a TMA-DPH fluorescent probe in cortical and hippocampal native membrane pellets in vivo. The r value considerably increased in elderly rats only in the cortical regions (P < 0.05) but without apparent changes in the hippocampus (E, F). Regarding the alterations in membrane lipid composition, HU-treated neurons showed a decreased cholesterol level in neuronal membrane pellets (P < 0.05) (G). All the data are expressed as mean ± SD from three independent experiments (N = 3). *P < 0.05, **P < 0.01, ***P < 0.001. The Student t test was used to determine the statistical significance of the differences.