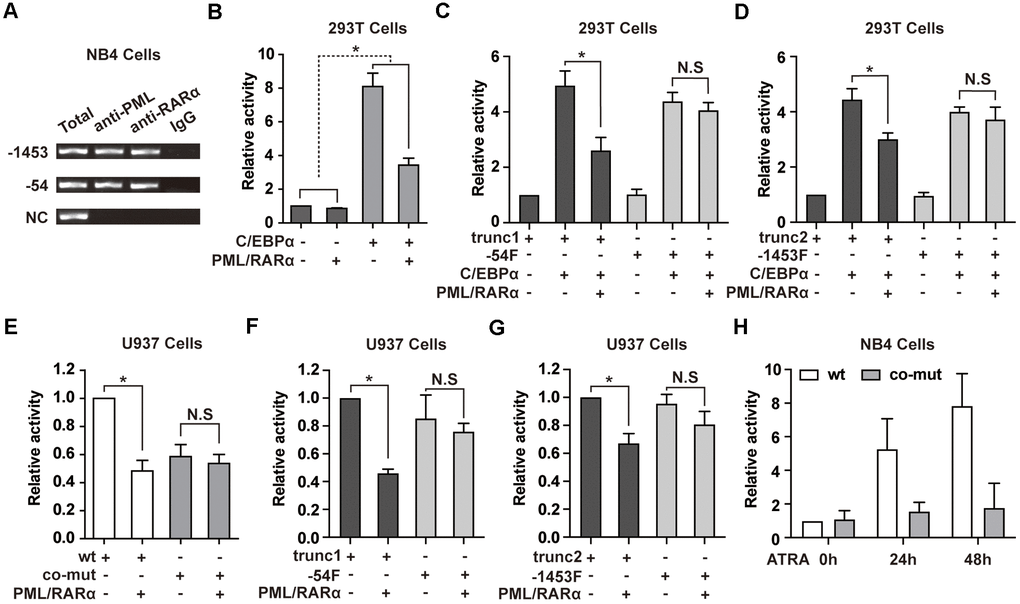
Figure 3.PML/RARα binds to NEAT1 promoter and represses the C/EBPα-mediated transactivation of NEAT1. (A) ChIP was performed in NB4 cells with anti-PML, anti-RARα, or nonspecific (normal immunoglobulin G (IgG)) antibodies. The immunoprecipitated DNA was amplified by PCR, followed by agarose electrophoresis. (B) The promoter of NEAT1 was co-transfected into 293T cells along with pcDNA3.1 vector or pcDNA3.1-PML/RARα expression plasmid in the absence or presence of C/EBPα. (-) and (+) represent the absence or presence of the indicated plasmid. (C, D) NEAT1 promoter truncation plasmids that contain (trunc1 and trunc2) or do not contain RARE and RARE half motifs (-54F and -1453F) were co-transfected with pcDNA3.1 vector or pcDNA3.1-C/EBPα and with or without PML/RARα-expression construct. Luciferase activity was detected 24 h after transfection. (E) The wild-type (wt) or double mutated (co-mut) NEAT1 promoter construct was co-transfected into U937 cells along with pcDNA3.1 vector or pcDNA3.1-PML/RARα expression plasmid. (F, G) NEAT1 promoter truncation plasmids in the presence (trunc1 and trunc2) or absence of RARE and RARE half motifs (-54F and -1453F) were co-transfected with pcDNA3.1 vector or pcDNA3.1-PML/RARα expression construct. (H) The wild-type (wt) or double mutated (co-mut) NEAT1 promoter construct was transfected into NB4 cells. Six hours later, cells were treated with ATRA and tested at the indicated time points. The error bar represents the standard error of the mean (S.E.M.) (n=3). * indicates p<0.05.