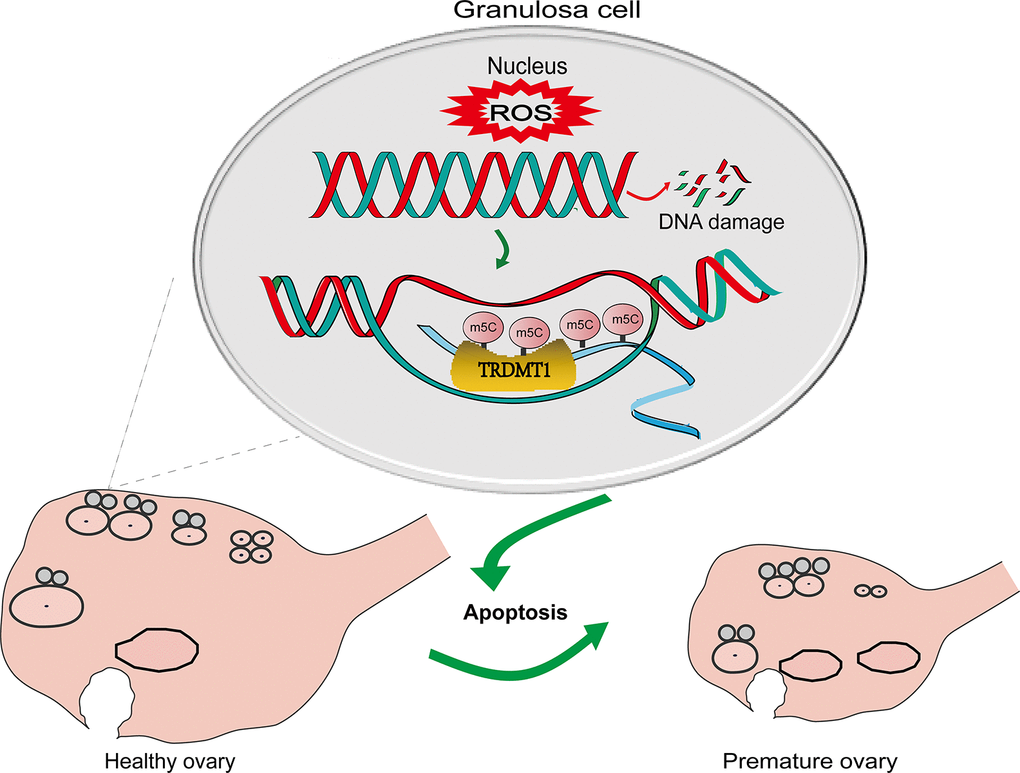
Figure 6.A schematic diagram showing the possible mechanism of TRDMT1 on GCs. CTX induced oxidative stress by generating ROS and thereby GCs apoptosis via DNA damage. The increased level of ROS leads to DNA damage. The increase in DNA damage promotes the apoptosis of GCs, thus promoting POF. TRDMT1 is important for DNA damage repair efficiency and survival. TRDMT1 participated in the DNA damage repair of GCs through methylation. m5C mRNA methylation is enriched at transcriptionally active sites with DNA damage. In short, oxidative damage to DNA induced the apoptosis of GCs and promoted the development of POF. TRDMT1 promoted DNA damage repair by regulating the methylation level, which reduced the apoptosis of GCs and inhibited the occurrence of POF. The regulation of oxidative DNA damage repair mediated by TRDMT1 was significantly correlated with its methylation activity.