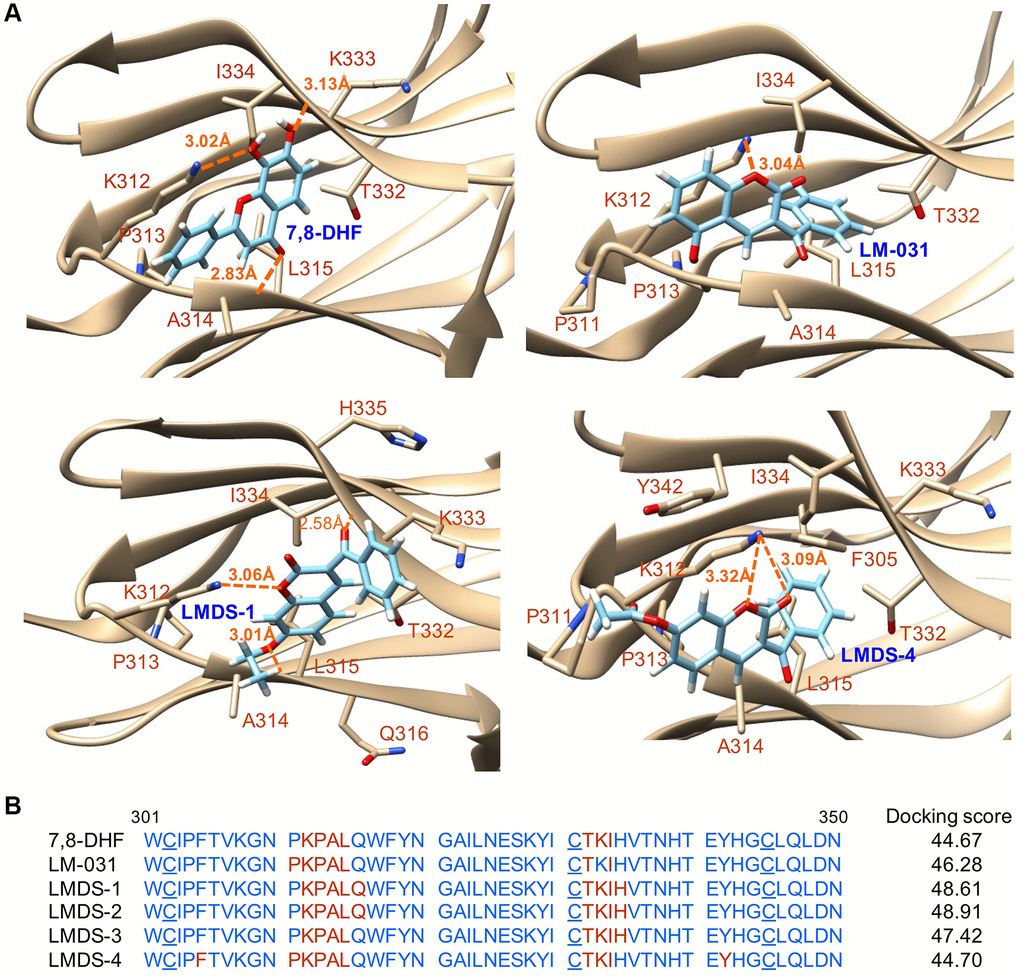
Figure 2.Docking computations of 7,8-DHF, LM-031 and analogs. (A) The docking conformations of 7,8-DHF (as a positive control), LM-031, LMDS-1 and LMDS-4 binding to extracellular d5 domain (the second immunoglobulin-like domain, residues 250–340) of TRKB receptor. The TRKB-d5 domain (ribbon structures) is colored in beige and the wire-frame structures denote the compounds. The labeled amino acids were within 10 Å radii of examined compounds. Carbon, oxygen, hydrogen and nitrogen atoms of compounds or side chains of surrounding amino acids are shown in light blue, red, white and blue, respectively. The dotted orange lines indicate hydrogen bond interactions between compounds and protein. (B) Amino acid residues 301–350 of d5 domain. The amino acids within 10 Å radii of examined compounds are colored in red; the cysteines involved in disulfide linkage are underlined. Shown on the right were docking scores of 7,8-DHF, LM-031 and analogs calculated by the GOLD program.