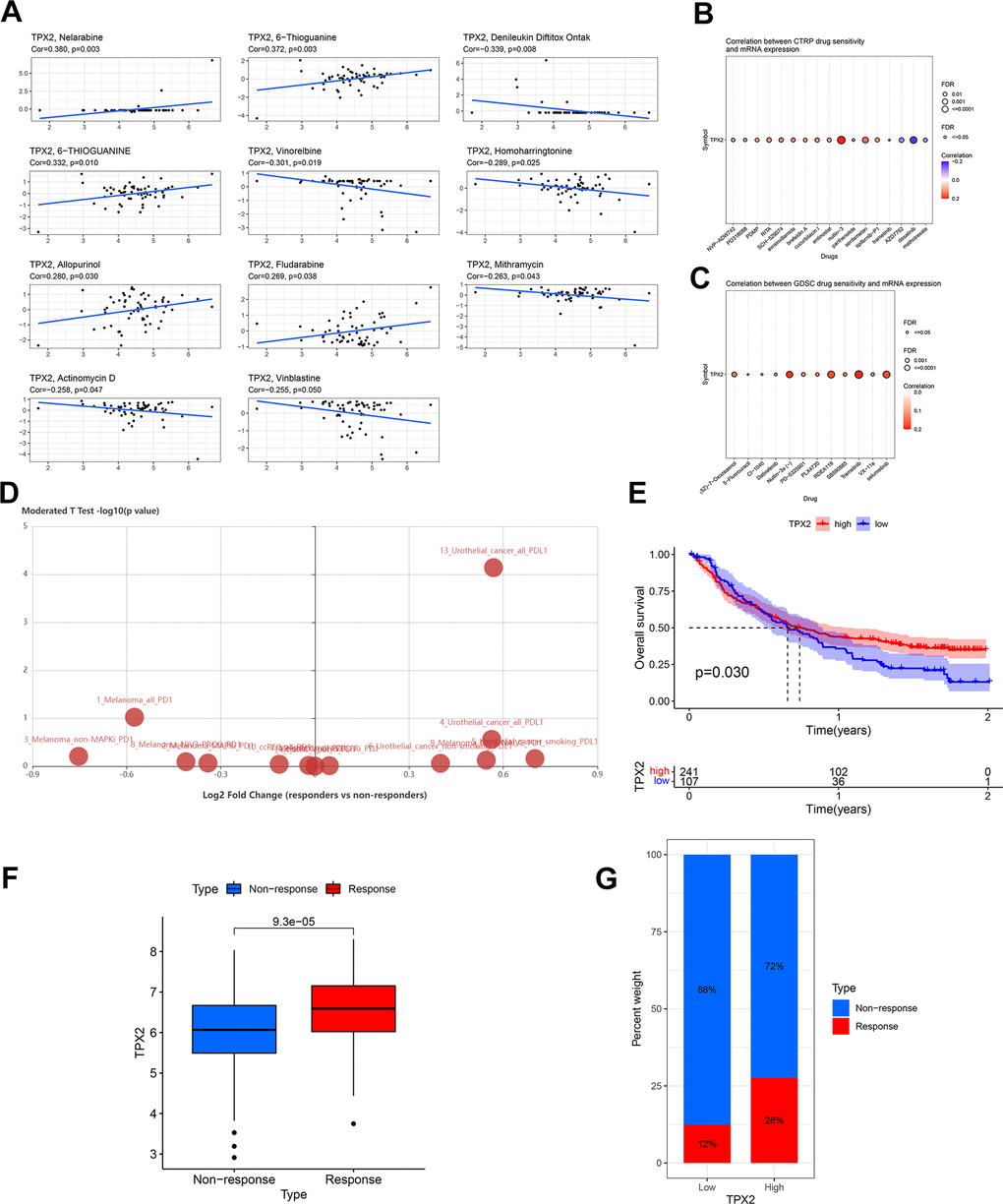
Figure 11.Drug sensitivity analysis and validation of the immunotherapeutic predictive value of TPX2. (A) Drug sensitivity analysis of TPX2 using the CellMiner database. (B) Correlation between CTRP drug sensitivity and TPX2 expression. (C) Correlation between GDSC drug sensitivity and TPX2 expression. Red dots indicate positive association. Blue dots indicate negative association. (D) Difference in expression of TPX2 between responders and non-responders undergoing anti-PD1/PD-L1 therapy using the TISIDB database. (E) Kaplan-Meier OS curves for TPX2 in IMvigor 210. (F) TPX2 expression was higher in responders than that in non-responders in IMvigor 210. (G) Treatment response rates with anti-PD-L1 therapy in patients with high and low expressions of TPX2 in IMvigor 210. p-value < 0.05 was considered statistically significant.