Sleep and feeding are mutually exclusive behaviors. Consequently, an animal must decide which behavior to express based on internal drives and environmental cues. These behaviors are also functionally interconnected: food-deprivation suppresses sleep, while sleep loss induces hunger [1,2]. Extreme dysregulation of either behavior on its own is deleterious. Longitudinal studies in humans have revealed increased Body Mass Index in short sleeping individuals [3]. The neuropeptides Orexin and neuropeptide Y (NPY) both suppress sleep and promote feeding [4,5], while mice mutant for the leptin receptor have disrupted sleep patterns [6].
Sleep loss potently affects insulin function and has been clinically linked to Diabetes mellitus, metabolic syndromes, like Diabetes mellitus and obesity. It is possible that the interplay between sleep and metabolic syndromes occurs through the direct effect of sleep on metabolism or indirectly through the dysregulation of appetite [7]. Understanding the molecular and neural link between sleep and feeding will aid in our understanding of obesity and sleep-linked disorders.
Much of the genetic architecture controlling sleep, feeding and metabolism is conserved across phyla. A powerful genetic toolkit has been developed in the fruit fly, Drosophila melanogaster, that allows for the manipulation of genes and neural circuits with regional and temporal specificity [8]. Genetic screens in Drosophila have led to the identification of many genes affecting sleep, feeding and metabolism with conserved function in mammals. For example the Dopamine transporter promotes sleep [9,10] and a genome-wide obesity screen identified the hedgehog pathway as a conserved determinant of fat generation [11].
To gain insight into the genetic and neural basis of sleep-feeding interactions we investigated the effects of food-deprivation on Drosophila sleep. Energy stores and sleep needs are linked suggesting a link between metabolism and sleep [12]. In addition, because starved flies only survive 1-2 days, we reasoned they might be particularly sensitive to the sleep-suppressing effects of food-deprivation.
We therefore monitored flies' activity over a 24-hour period in small tubes with either standard fly food or agar as a feeding substrate (Figure 1A, B). We found that wild-type flies robustly suppress sleep following 12-hours of starvation on agar (Figure 1C, D), suggesting that the effect of food-deprivation on sleep that was previously documented in mammals is conserved in Drosophila.
Mammalian sleep is composed of distinct stages that can be characterized by unique electrophysiological properties. Sleep in flies is a also accompanied by alterations in neural activity [13], yet the relevance of these changes to mammalian sleep states remains unclear. Consolidation of sleep can be measured behaviorally in flies by determining the average length and total number of individual sleep bouts. Disruption in bout number suggests difficulty in initiating sleep while shortened bout length indicates a failure to maintain sleep. We found that 24 hours of starvation decreases bout number without affecting bout length (Figure 1E, F). Therefore, food-deprivation specifically affects the onset of sleep without affecting sleep maintenance.
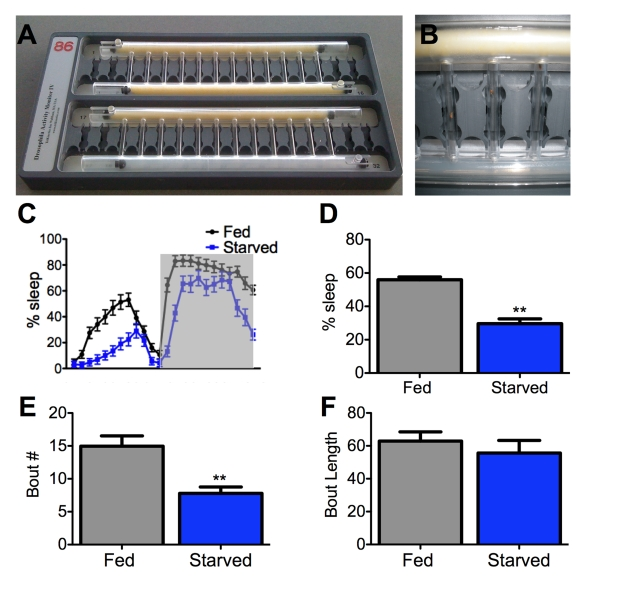
Figure 1. Starvation impairs sleep initiation but not maintenance. (A,B)
A Drosophila activity monitor typically used for sleep studies
can record up to 32 flies simultaneously. An individual fly is housed in
each vertical tube and an infrared beam detects activity. The large
horizontal tubes contain either food (yellow) or agar (translucent).
Sliding barriers control access to each substrate [32]. Both tubes
contain food for fed controls (A, top), while agar is provided to
the starved experimental group (A, bottom; and B) on day 2 of testing the
experiment (starved, experimental). (C,D) Female flies
starved for 24 hours sleep less than fed counterparts. Shaded area (C)
represents lights-off. (E, F) The total number of sleep
bouts (Bout #) is decreased in starved flies while average bout length does
not differ from fed counterparts. Asterisk denotes significant difference
(P<0.01, ANOVA) from control groups. Data are mean ± SEM.
We screened for mutants with aberrant sleep during starvation in order to identify genes linking sleep and feeding. We found that mutants for the genes Clock and cycle are hypersensitive to the wake-promoting effects of food-deprivation. Clock and cycle are transcriptional activators that are expressed in ~150 central brain neurons, multiple populations of sensory neurons and peripheral cells. Clock and cycle function as binding partners and are required for 24-hour transcriptional cycling of the core-circadian clock [14].
In addition to regulating circadian rhythms, Clock and cycle have been implicated in the regulation of sleep, feeding, olfaction, and starvation resistance [15-18]. Clock-dependent modulation of each behavior appears to be conferred through distinct neuronal populations. For example, Clock-regulated control of circadian behavior localizes to eight neurons termed the small ventrolateral neurons [15] while regulation of feeding and starvation resistance localize to the gustatory neurons and fat body bodies [16,18]. Through tissue-specific disruption of Clock function we probed populations of cells for their role in starvation-induced sleep suppression. Selectively disrupting Clock function in a population of dorsally located neurons in the central brain phenocopied the genetic mutant. However, eliminating Clock function in cells previously implicated in circadian locomotor behavior, feeding, olfaction, vision, or starvation-resistance did not affect sleep-suppression during starvation. Therefore, cellular control of sleep-feeding interactions appears to be distinct from those controlling other Clock-dependent behaviors.
The pleiotropic nature of behavior suggests many additional genes function in concert with Clock and cycle to modulate sleep-feeding interactions. Neuropeptide F, the Drosophila ortholog of Neuropeptide Y, has been implicated in control of feeding [19] and motivational behavior [20] and is an excellent candidate for modulating sleep-feeding interactions. The mammalian gastrointestinal satiety-inducing peptide cholecys tokinin (CCK) has been reported to induce sleep and a CCK-A receptor antagonist blocks this effect [21]. The function of drosulfakinin, the fly ortholog of CCK, is unknown. It is expressed in the brain [22] and represents a candidate for signaling nutrients cues to Clock-expressing neurons.
In mammals, hypothalamic Orexin regulates both sleep and feeding and Orexin signaling has been proposed as an attractive drug target for dysfunction of both sleep and feeding systems [23,24]. In addition to Orexin, T-type Ca2+ channels have been linked to regulation of sleep-feeding interactions. Administration of a selective T-Type Ca2+ channel antagonist increases sleep and reduces body fat in mice fed a high-fat diet [25]. In flies, Ca2+ homeostasis has been linked to sleep-wake regulation [26] and future investigation of the role of specific Ca2+ channels in the regulation of sleep and feeding may be informative.
Our study focused on the effect of food-deprivation on sleep. The consequences of sleep-deprivation on metabolism were not addressed. Loss of sleep has detrimental effects on metabolism and has been linked to conditions such as obesity and diabetes [27], and the Drosophila insulin-producing cells have been shown to regulate sleep. Hyperexcitation of insulin insulin- producing cells inhibits sleep [28] while activation of the Epidermal Growth Factor Receptor activation in these cells induces sleep [29]. These findings suggest a functional link between the systems controlling insulin and sleep. Furthermore, alterations in mice mutant for Clock and BMAL1, the mammalian orthologs of Clock and cycle, have significant metabolic defects that include decreased insulin release and a diminished ability to maintain normal blood glucose levels [30,31]. Future work examining the metabolism of short-sleeping Drosophila mutants may aid our understanding of the link between sleep loss and metabolic dysfunction.
Identifying the molecular basis of behavioral integration will pave the way for the development of drugs that act in a context-dependent fashion. Our findings that Clock and cycle regulate sleep during food-deprivation is a starting point for understanding the complex interactions regulating sleep and feeding. Utilizing currently available fly mutants to verify candidate genes identified in large-scale fly and mammalian analyses should significantly improve our understanding of sleep-feeding interactions and resulting pathologies.
References
- 1. MacFadyen UM , Oswald I and Lewis SA. Starvation and human slow-wave sleep. J Appl Physiol. 1973; 35: 391 -394. [PubMed] .
- 2. Spiegel K , Tasali E , Penev P and Van Cauter E. Brief communication: Sleep curtailment in healthy young men is associated with decreased leptin levels, elevated ghrelin levels, and increased hunger and appetite. Ann Intern Med. 2004; 141: 846 -850. [PubMed] .
- 3. Taheri S , Lin L , Austin D , Young T and Mignot E. Short sleep duration is associated with reduced leptin, elevated ghrelin, and increased body mass index. PLoS Med. 2004; 1: e62 [PubMed] .
- 4. Szentirmai E and Krueger JM. Central administration of neuropeptide Y induces wakefulness in rats. Am J Physiol Regul Integr Comp Physiol. 2006; 291: R473 -480. [PubMed] .
- 5. Chemelli RM , Willie JT , Sinton CM , Elmquist JK , Scammell T , Lee C , Richardson JA , Williams SC , Xiong Y and Kisanuki Y. Narcolepsy in orexin knockout mice: molecular genetics of sleep regulation. Cell. 1999; 98: 437 -451. [PubMed] .
- 6. Laposky AD , Bradley MA , Williams DL , Bass J and Turek FW. Sleep-wake regulation is altered in leptin-resistant (db/db) genetically obese and diabetic mice. Am J Physiol Regul Integr Comp Physiol. 2008; 295: R2059 -2066. [PubMed] .
- 7. Spiegel K , Knutson K , Leproult R , Tasali E and Van Cauter E. Sleep loss: a novel risk factor for insulin resistance and Type 2 diabetes. J Appl Physiol. 2005; 99: 2008 -2019. [PubMed] .
- 8. Venken KJ and Bellen HJ. Emerging technologies for gene manipulation in Drosophila melanogaster. Nat Rev Genet. 2005; 6: 167 -178. [PubMed] .
- 9. Kume K , Kume S , Park SK , Hirsh J and Jackson FR. Dopamine is a regulator of arousal in the fruit fly. J Neurosci. 2005; 25: 7377 -7384. [PubMed] .
- 10. Wu MN , Koh K , Yue Z , Joiner WJ and Sehgal A. A genetic screen for sleep and circadian mutants reveals mechanisms underlying regulation of sleep in Drosophila. Sleep. 2008; 31: 465 -472. [PubMed] .
- 11. Pospisilik JA , Knauf C , Joza N , Benit P , Orthofer M , Cani PD , Ebersberger I , Nakashima T , Sarao R and Neely G. Targeted deletion of AIF decreases mitochondrial oxidative phosphorylation and protects from obesity and diabetes. Cell. 2007; 131: 476 -491. [PubMed] .
- 12. Harbison ST and Sehgal A. Quantitative genetic analysis of sleep in Drosophila melanogaster. Genetics. 2008; 178: 2341 -2360. [PubMed] .
- 13. Nitz DA , van Swinderen B , Tononi G , Greenspan RJ: Electrophysiological correlates of rest and activity in Drosophila melanogaster. Curr Biol. 2002; 12: 1934 -1940. [PubMed] .
- 14. Rutila JE , Suri V , Le M , So WV , Rosbash M and Hall JC. CYCLE is a second bHLH-PAS clock protein essential for circadian rhythmicity and transcription of Drosophila period and timeless. Cell. 1998; 93: 805 -814. [PubMed] .
- 15. Tanoue S , Krishnan P , Krishnan B , Dryer SE and Hardin PE. Circadian clocks in antennal neurons are necessary and sufficient for olfaction rhythms in Drosophila. Curr Biol. 2004; 14: 638 -649. [PubMed] .
- 16. Chatterjee A , Tanoue S , Houl JH and Hardin PE. Regulation of Gustatory Physiology and Appetitive Behavior by the Drosophila Circadian Clock. Curr Biol. 2010; 20: 300 -309. [PubMed] .
- 17. Hendricks JC , Lu S , Kume K , Yin JC , Yang Z and Sehgal A. Gender dimorphism in the role of cycle (BMAL1) in rest, rest regulation, and longevity in Drosophila melanogaster. J Biol Rhythms. 2003; 18: 12 -25. [PubMed] .
- 18. Xu K , Zheng X and Sehgal A. Regulation of feeding and metabolism by neuronal and peripheral clocks in Drosophila. Cell Metab. 2008; 8: 289 -300. [PubMed] .
- 19. Wu Q , Zhao Z and Shen P. Regulation of aversion to noxious food by Drosophila neuropeptide Y- and insulin-like systems. Nat Neurosci. 2005; 8: 1350 -1355. [PubMed] .
- 20. Krashes MJ , DasGupta S , Vreede A , White B , Armstrong JD and Waddell S. A neural circuit mechanism integrating motivational state with memory expression in Drosophila. Cell. 2009; 139: 416 -427. [PubMed] .
- 21. Shemyakin A and Kapas L. L-364,718, a cholecystokinin-A receptor antagonist, suppresses feeding-induced sleep in rats. Am J Physiol Regul Integr Comp Physiol. 2001; 280: R1420 -1426. [PubMed] .
- 22. Nichols R and Lim IA. Spatial and temporal immunocytochemical analysis of drosulfakinin (Dsk) gene products in the Drosophila melanogaster central nervous system. Cell Tissue Res. 1996; 283: 107 -116. [PubMed] .
- 23. Willie JT , Chemelli RM , Sinton CM and Yanagisawa M. To eat or to sleep? Orexin in the regulation of feeding and wakefulness. Annu Rev Neurosci. 2001; 24: 429 -458. [PubMed] .
- 24. Hungs M and Mignot E. Hypocretin/orexin, sleep and narcolepsy. Bioessays. 2001; 23: 397 -408. [PubMed] .
- 25. Uebele VN , Gotter AL , Nuss CE , Kraus RL , Doran SM , Garson SL , Reiss DR , Li Y , Barrow JC and Reger TS. Antagonism of T-type calcium channels inhibits high-fat diet-induced weight gain in mice. J Clin Invest. 2009; 119: 1659 -1667. [PubMed] .
- 26. Zimmerman JE , Rizzo W , Shockley KR , Raizen DM , Naidoo N , Mackiewicz M , Churchill GA and Pack AI. Multiple mechanisms limit the duration of wakefulness in Drosophila brain. Physiol Genomics. 2006; 27: 337 -350. [PubMed] .
- 27. Knutson KL , Spiegel K , Penev P and Van Cauter E. The metabolic consequences of sleep deprivation. Sleep Med Rev. 2007; 11: 163 -178. [PubMed] .
- 28. Crocker A , Shahidullah M , Levitan IB and Sehgal A: Identification of a neural circuit that underlies the effects of octopamine on sleep:wake behavior. Neuron. 2010; 65: 670 -681. [PubMed] .
- 29. Foltenyi K , Greenspan RJ and Newport JW. Activation of EGFR and ERK by rhomboid signaling regulates the consolidation and maintenance of sleep in Drosophila. Nat Neurosci. 2007; 10: 1160 -1167. [PubMed] .
- 30. Marcheva B , Ramsey KM , Buhr ED , Kobayashi Y , Su H , Ko CH , Ivanova G , Omura C , Mo S and Vitaterna MH. Disruption of the clock components CLOCK and BMAL1 leads to hypoinsulinaemia and diabetes. Nature. 2010; 466: 627 -631. [PubMed] .
- 31. Froy O and Miskin R. Effect of feeding regimens on circadian rhythms: implications for aging and longevity. Aging. 2010; 2: 7 -27. [PubMed] .
- 32. Agosto J , Choi JC , Parisky KM , Stilwell G , Rosbash M and Griffith LC. Modulation of GABAA receptor desensitization uncouples sleep onset and maintenance in Drosophila. Nat Neurosci. 2008; 11: 354 -359. [PubMed] .



