FoxO TFs in longevity and aging-associated disease
The role of FoxO TFs in lifespan control was first established in C. elegans, where reduction of IIS or ablation of the germline extends lifespan in a DAF-16/FoxO-dependent manner [1,2]. FoxO also promotes lifespan extension in Drosophila [3,4]. Although reduction of IIS also promotes longevity in mammals [5,6,7], whether or not this requires FoxO TFs remains to be determined. Multiple studies indicate that specific FoxO polymorphisms are associated with longevity in cohorts of extremely long-lived humans [8,9,10,11,12,13], suggesting that FoxO TFs may also regulate lifespan in humans.
While a direct connection between mammalian FoxO TFs and lifespan control has not yet been established experimentally, recent work in conditional knockout mice supports a role for FoxO TFs in modulating phenotypes that are reminiscent of aging-associated diseases in humans. For example, mice lacking FoxO1, FoxO3, and FoxO4 develop thymic lymphomas and hemangiomas, indicating that FoxO TFs are bonafide tumor suppressors [14]. Furthermore, deletion of FoxO TFs in osteoblasts results in reduced bone mass secondary to increased osteoblast apoptosis [15,16], suggesting that FoxO TFs are protective against osteoporosis. In contrast to these apparent salubrious effects of FoxO TFs, FoxO1 can contribute to metabolic dysregulation similar to that observed in Type 2 diabetes, as FoxO1 haploinsufficiency protects mice against insulin resistance induced by a high-fat diet [17], and both liver-specific and osteoblast-specific FoxO1 deletion ameliorate glucose intolerance in mouse models of insulin resistance [18,19,20]. Thus, in mammals, FoxO TFs have context-dependent effects on the development of phenotypes associated with age-related disease. Elucidating the regulatory mechanisms that maintain the balance of FoxO TF activity may prove to be crucial for understanding and combating the progression of age-related disease.
DAF-16/FoxO regulation in the control of C. elegans lifespan
DAF-16/FoxO is required for C. elegans lifespan modulation by IIS and the germline [1,2], as well as in some contexts of dietary restriction [21]. Reduction of IIS and ablation of the germline both extend lifespan by increasing DAF-16/FoxO activity. Neither intervention increases lifespan in daf-16/FoxO null mutants [1,2], indicating that DAF-16/FoxO is the critical target of IIS and the germline in lifespan control.
IIS and the germline both inhibit DAF-16/FoxO by promoting its cytoplasmic sequestration. When IIS is reduced, or when the germline is ablated, DAF-16/FoxO translocates to the nucleus [22,23,24], where it executes gene regulatory programs that promote longevity. IIS induces the phosphorylation of DAF-16/FoxO by Akt/Protein Kinase B (PKB) [23,24], which results in the cytoplasmic sequestration of DAF-16/FoxO through its direct association with 14-3-3 pro- teins [25,26]. The molecular requirements for DAF-16/FoxO nuclear translocation in the context of reduced IIS have not been fully delineated but may include serine-threonine kinases such as JNK-1 [27] and CST-1 [28].
How the germline promotes the cytoplasmic sequestration of DAF-16/FoxO is not entirely understood. Notably, germline ablation extends lifespan in animals with reduced IIS [2]. In animals lacking a germ-line, translocation of cytoplasmic DAF-16/FoxO to the nucleus requires the nuclear receptor DAF-12 [29] and its steroid hormone ligands [29,30], which are known as dafachronic acids (DAs) [31]. The conserved protein KRI-1 is required for DAF-16/FoxO nuclear translocation in animals lacking a germline but largely dispensable for DAF-16/FoxO nuclear localization in animals with reduced IIS [29]. In aggregate, these observations suggest that IIS and the germline control the subcellular localization of DAF-16/FoxO via distinct mechanisms.
Nuclear translocation is not sufficient for full DAF-16/FoxO activation
Although nuclear localization of DAF-16/FoxO is clearly necessary for DAF-16/FoxO-dependent lifespan extension, multiple lines of evidence indicate that it is not sufficient for full DAF-16/FoxO activation. For example, a DAF-16/FoxO mutant lacking all four canonical Akt/PKB phosphorylation sites localizes to the nucleus but fails to fully extend lifespan [24,29]. This indicates that a second pathway acts in parallel to Akt/PKB and the germline to inhibit the activity of nuclear DAF-16/FoxO.
A genetic screen for molecules that regulate nuclear DAF-16/FoxO activity
To identify components of this parallel DAF-16/FoxO regulatory pathway, we exploited the fact that in larvae, DAF-16/FoxO promotes developmental arrest in an alternative larval stage called dauer that is morphologically distinct from reproductively developing larvae [32]. We performed a genetic screen for mutants that enhance the weak dauer-constitutive phenotype of an akt-1 null mutant (eak screen). We identified 21 independent mutants that define seven eak genes, six of which have been cloned (Table 1) [33,34,35]. Strikingly, five of the six cloned eak genes are expressed specifically in the two endocrine XXX cells [34,35,36]; in contrast, eak-7 is expressed in the XXX cells as well as multiple other tissues [33]. The phenotypic similarity of all eak single mutants and the observation that no eak;eak double mutant combination tested to date exhibits phenotypic enhancement compared to eak single mutants indicate that the EAK proteins are components of a single pathway.
Table 1. Seven eak genes. The molecular identity of eak-1 is not known.
| Gene | Predicted product | Human ortholog | Expression pattern | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| eak-1 | ? | ? | ? | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| eak-2 | hsd-1 | Sdr42e1 | XXX cells | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| eak-3 | N-myristoylated protein | ? | XXX cells | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| eak-4 | N-myristoylated protein | ? | XXX cells | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| eak-5 | sdf-9 | ? | XXX cells | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| eak-6 | Protein tyrosine phosphatase | ? | XXX cells1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| eak-7 | N-myristoylated TLDc protein | KIAA1609 | XXX cells, neurons, intestine, other tissues | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| eak-6 is also expressed in the M1 pharyngeal motor neuron | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
EAK proteins and AKT-1 both control DAF-16/FoxO target gene expression. However, in contrast to AKT-1, which inhibits DAF-16/FoxO activity by promoting its translocation from the nucleus to the cytoplasm, EAK proteins inhibit nuclear DAF-16/FoxO activity without influencing its subcellular localization [33,34,35]. Collectively, the data indicate that EAK proteins define a new conserved endocrine pathway that acts in parallel to Akt/PKB to regulate the activity of nuclear DAF-16/FoxO. Two eak genes, eak-2/hsd-1 and eak-7, have putative mammalian orthologs based on comparative sequence analysis [33,34].
EAK-2/HSD-1
EAK-2 is allelic to the 3β-hydroxysteroid dehydro-genase family member HSD-1 [34,37]. HSD-1 is thought to participate with the cytochrome P450 DAF-9 in the biosynthesis of Δ4-dafachronic acid (Δ4-DA) [31,37]; the related steroid Δ7-DA is synthesized by the Rieske oxygenase family member DAF-36 and DAF-9 [31,38]. Both Δ4- and Δ7-DA are high-affinity ligands for the nuclear receptor DAF-12 [31].
The identification of hsd-1 mutations in the eak screen implicates steroid hormone signaling in the regulation of nuclear DAF-16/FoxO activity. In support of this, we have shown that daf-36 and daf-9mutations also enhance the akt-1 dauer-constitutive phenotype [34]. Furthermore, hsd-1 mutation synergizes with akt-1 mutation to increase DAF-16/FoxO target gene expression, and DAF-16/FoxO target gene expression in hsd-1;akt-1 double mutants requires not only DAF-16/FoxO but also DAF-12 [34]. Thus, DAF-12 may act in parallel to DAF-16/FoxO to coregulate a subset of DAF-16/FoxO target genes. hsd-1 is expressed exclusively in the two endocrine XXX cells, where daf-9 is also expressed [34,37,39]. Taken together, these findings suggest that DAs synthesized in the XXX cells act through DAF-12 and in parallel to AKT-1 to coregulate DAF-16/FoxO target gene expression. In contrast to akt-1 mutation, hsd-1 mutation does not promote nuclear translocation of a functional DAF-16::GFP fusion protein [34], indicating that HSD-1 controls DAF-16/FoxO activity without promoting DAF-16/FoxO translocation to the cytoplasm.
The role of HSD-1 in lifespan control is complex. In wild-type animals, HSD-1 does not have a major impact on adult lifespan. However, in animals with reduced IIS, HSD-1 is required for full lifespan extension [34]. This is reminiscent of the requirement for DAF-36 and DAF-9 in lifespan extension induced by germline ablation [29,40]. Surprisingly, in contrast to daf-36 and daf-9 mutations, which substantially shorten the lifespan of animals lacking a germline [29,40], hsd-1 mutation does not affect lifespan extension induced by germline ablation [34]. This may be a consequence of the relative potencies of Δ4- and Δ7-DA and/or the anatomical source of distinct DAs (XXX cells vs. hypodermis and somatic gonad). Alternatively, HSD-1 may participate in the biosynthesis of a different steroid hormone that is required for full lifespan extension in the context of reduced IIS but dispensable for lifespan extension induced by germline ablation.
EAK-7
In contrast to hsd-1 and other eak single mutants, eak-7 single mutants live approximately 25% longer than wild-type animals, and this lifespan extension requires DAF-16/FoxO as well as the DAF-16/FoxO cofactors HSF-1 and SMK-1 [33]. As is the case for other EAK proteins, EAK-7 inhibits nuclear DAF-16/FoxO activity without influencing its subcellular localization [33]. Notably, although eak-7 mutation strongly enhances the dauer-constitutive phenotype of akt-1 mutants, its enhancement of lifespan extension in akt-1 mutants is relatively modest [33]. This is likely a consequence of the influence of the germline on DAF-16/FoxO subcellular localization; since the germline promotes the cytoplasmic sequestration of DAF-16/FoxO [24], akt-1 mutation may not increase nuclear concentrations of DAF-16/FoxO to the extent that it does in early larval stages, before the germline has proliferated. In accordance with this, eak-7 mutation strongly enhances lifespan extension (by ~50-80%) in animals lacking a germline [33].
How EAK-7 controls nuclear DAF-16/FoxO activity is unclear. EAK-7 contains a consensus N-myristoylation (N-myr) motif and a TLDc (TBC- and LysM-domain-containing) domain [33], the function of which is poorly understood. Interestingly, a functional EAK-7::GFP fusion protein localizes to the plasma membrane and does not appear to translocate in adult animals [33]. Thus, EAK-7 likely regulates nuclear DAF-16/FoxO activity indirectly. eak-7 mutation increases steady-state DAF-16/FoxO protein levels in akt-1 mutants without altering daf-16/FoxO transcript levels, suggesting that EAK-7 may modulate DAF-16/FoxO synthesis or turnover [33]. A hypothetical model of how EAK-7 controls DAF-16/FoxO activity in concert with IIS and the germline is shown in Figure 1.
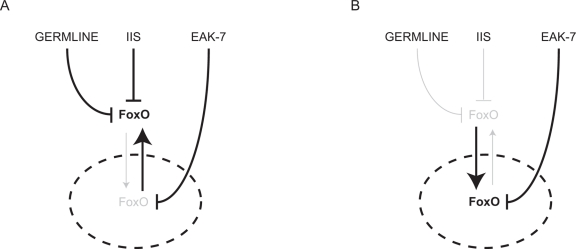
Figure 1. Hypothetical model of FoxO regulation by the germline, IIS, and EAK-7. The nucleus is denoted by the dashed ellipse. A. In the presence of an intact germline and IIS, most FoxO is sequestered in the cytoplasm, and EAK-7 inhibits the activity of the small fraction of cellular FoxO that resides in the nucleus. B. In animals with reduced IIS and/or germline signaling, EAK-7 has a greater influence on FoxO activity, since relative concentrations of nuclear FoxO are increased.
Potential role of the EAK pathway in diseases associated with aging
Although studies in mammalian cells have also implicated a regulatory input that controls the activity of FoxO TFs that are localized to the nucleus [41], the nature of this input is obscure. Our data suggest that the putative HSD-1 and EAK-7 mammalian orthologs Sdr42e1 and KIAA1609 may be components of this input. Recent work on mice harboring conditional knockout alleles of FoxO1 point to a potential role for FoxO TFs in the pathogenesis of Type 2 diabetes [17,18,19,20], cancer [14], and osteoporosis [16,42]; thus, it is conceivable that the EAK pathway may influence the pathogenesis of these aging-associated diseases.
The function of the putative mammalian HSD-1 ortholog Sdr42e1 is not known. Sdr42e1 mRNA is expressed in a limited number of mouse tissues, with strikingly high expression in liver [43], where FoxO1 plays an important role in metabolic homeostasis [19,20]. In contexts of insulin resistance, reduction of hepatic FoxO1 activity substantially ameliorates dysregulation of glucose metabolism in mice [19,20]. Since FoxO1 is nuclear in these circumstances [44], Sdr42e1 could play an important role in controlling hepatic FoxO1 activity and glucose homeostasis. The putative EAK-7 ortholog KIAA1609 is widely expressed in mouse tissues and is also present in mouse liver [43], indicating that KIAA1609 may also influence glucose metabolism in the context of insulin resistance. Both Sdr42e1 and KIAA1609 are expressed in mouse osteoblasts [43], where FoxO TFs promote the maintenance of normal bone mass and control metabolism nonautonomously through regulation of osteocalcin activity [16,42]; thus, EAK pathway activity in osteoblasts could influence the development of both osteoporosis and Type 2 diabetes. Intriguingly, KIAA1609 is highly expressed in mouse retinal pigment epithelium (RPE), where FoxO1 and FoxO3 are also expressed [43]. Although nothing is known about the function of FoxO TFs in RPE, the extreme levels of KIAA1609 expression in this tissue suggest that the EAK pathway could be important in regulating FoxO activity in RPE.
It is noteworthy that the genes encoding human Sdr42e1 and KIAA1609 lie within 2.5 megabases of each other at chromosome 16q23. Copy number variation in this genomic region could affect both genes coordinately, changing baseline EAK pathway activity (and, in turn, FoxO TF activity) accordingly.
Summary
The EAK pathway is a new conserved FoxO regulatory input that acts in parallel to Akt/PKB and the germline to control C. elegans lifespan. Continued investigation of the C. elegans EAK pathway promises to reveal new insights into mechanisms of FoxO regulation. In light of the potential role of FoxO TFs in mammalian aging and the pathogenesis of aging-associated diseases, EAK proteins could emerge as promising targets for the development of new drugs to treat Type 2 diabetes, cancer, and osteoporosis. Studies of Sdr42e1 and KIAA1609 function in mouse models of aging and disease will be especially revealing in this regard.
Acknowledgments
Our research is funded by the National Institute of Diabetes and Digestive and Kidney Diseases, the American Cancer Society, and the Sidney Kimmel Foundation for Cancer Research.
Conflicts of Interest
The authors of this manuscript have no conflict of interests to declare.
References
- 1. Kenyon C, Chang J, Gensch E, Rudner A, Tabtiang R. A C. elegans mutant that lives twice as long as wild type. Nature. 1993; 366: 461 -464. [PubMed] .
- 2. Hsin H and Kenyon C. Signals from the reproductive system regulate the lifespan of C. elegans. Nature. 1999; 399: 362 -366. [PubMed] .
- 3. Giannakou ME, Goss M, Junger MA, Hafen E, Leevers SJ, Partridge L. Long-lived Drosophila with overexpressed dFOXO in adult fat body. Science. 2004; 305: 361 [PubMed] .
- 4. Hwangbo DS, Gershman B, Tu MP, Palmer M, Tatar M. Drosophila dFOXO controls lifespan and regulates insulin signalling in brain and fat body. Nature. 2004; 429: 562 -566. [PubMed] .
- 5. Bluher M, Kahn BB, Kahn CR. Extended longevity in mice lacking the insulin receptor in adipose tissue. Science. 2003; 299: 572 -574. [PubMed] .
- 6. Holzenberger M, Dupont J, Ducos B, Leneuve P, Geloen A, Even PC, Cervera P, Le Bouc Y. IGF-1 receptor regulates lifespan and resistance to oxidative stress in mice. Nature. 2003; 421: 182 -187. [PubMed] .
- 7. Taguchi A, Wartschow LM, White MF. Brain IRS2 signaling coordinates life span and nutrient homeostasis. Science. 2007; 317: 369 -372. [PubMed] .
- 8. Anselmi CV, Malovini A, Roncarati R, Novelli V, Villa F, Condorelli G, Bellazzi R, Puca AA. Association of the FOXO3A locus with extreme longevity in a southern Italian centenarian study. Rejuvenation Res. 2009; 12: 95 -104. [PubMed] .
- 9. Flachsbart F, Caliebe A, Kleindorp R, Blanche H, von Eller-Eberstein H, Nikolaus S, Schreiber S, Nebel A. Association of FOXO3A variation with human longevity confirmed in German centenarians. Proc Natl Acad Sci U S A. 2009; 106: 2700 -2705. [PubMed] .
- 10. Willcox BJ, Donlon TA, He Q, Chen R, Grove JS, Yano K, Masaki KH, Willcox DC, Rodriguez B, Curb JD. FOXO3A genotype is strongly associated with human longevity. Proc Natl Acad Sci U S A. 2008; 105: 13987 -13992. [PubMed] .
- 11. Pawlikowska L, et al. Association of common genetic variation in the insulin/IGF1 signaling pathway with human longevity. Aging Cell. 2009; 8: 460 -472. [PubMed] .
- 12. Li Y, et al. Genetic association of FOXO1A and FOXO3A with longevity trait in Han Chinese populations. Hum Mol Genet. 2009; 18: 4897 -4904. [PubMed] .
- 13. Lunetta KL, D'Agostino RB Sr., Karasik D, Benjamin EJ, Guo CY, Govindaraju R, Kiel DP, Kelly-Hayes M, Massaro JM, Pencina MJ, Seshadri S, Murabito JM. Genetic correlates of longevity and selected age-related phenotypes: a genome-wide association study in the Framingham Study. BMC Med Genet. 2007; 8: Suppl 1 S13 [PubMed] .
- 14. Paik JH, et al. FoxOs are lineage-restricted redundant tumor suppressors and regulate endothelial cell homeostasis. Cell. 2007; 128: 309 -323. [PubMed] .
- 15. Ambrogini E, Almeida M, Martin-Millan M, Paik JH, Depinho RA, Han L, Goellner J, Weinstein RS, Jilka RL, O'Brien CA, Manolagas SC. FoxO-mediated defense against oxidative stress in osteoblasts is indispensable for skeletal homeostasis in mice. Cell Metab. 11: 136 -146. [PubMed] .
- 16. Rached MT, Kode A, Xu L, Yoshikawa Y, Paik JH, Depinho RA, Kousteni S. FoxO1 is a positive regulator of bone formation by favoring protein synthesis and resistance to oxidative stress in osteoblasts. Cell Metab. 2010; 11: 147 -160. [PubMed] .
- 17. Nakae J, Kitamura T, Kitamura Y, Biggs WH 3rd, Arden KC, Accili D. The forkhead transcription factor Foxo1 regulates adipocyte differentiation. Dev Cell. 2003; 4: 119 -129. [PubMed] .
- 18. Rached MT, Kode A, Silva BC, Jung DY, Gray S, Ong H, Paik JH, DePinho RA, Kim JK, Karsenty G, Kousteni S. FoxO1 expression in osteoblasts regulates glucose homeostasis through regulation of osteocalcin in mice. J Clin Invest. 2010; 120: 357 -368. [PubMed] .
- 19. Matsumoto M, Pocai A, Rossetti L, Depinho RA, Accili D. Impaired regulation of hepatic glucose production in mice lacking the forkhead transcription factor Foxo1 in liver. Cell Metab. 2007; 6: 208 -216. [PubMed] .
- 20. Dong XC, Copps KD, Guo S, Li Y, Kollipara R, DePinho RA, White MF. Inactivation of hepatic Foxo1 by insulin signaling is required for adaptive nutrient homeostasis and endocrine growth regulation. Cell Metab. 2008; 8: 65 -76. [PubMed] .
- 21. Greer EL and Brunet A. Different dietary restriction regimens extend lifespan by both independent and overlapping genetic pathways in C. elegans. Aging Cell. 2009; 8: 113 -127. [PubMed] .
- 22. Henderson ST and Johnson TE. daf-16 integrates developmental and environmental inputs to mediate aging in the nematode Caenorhabditis elegans. Curr Biol. 2001; 11: 1975 -1980. [PubMed] .
- 23. Lee RY, Hench J, Ruvkun G. Regulation of C. elegans DAF-16 and its human ortholog FKHRL1 by the daf-2 insulin-like signaling pathway. Curr Biol. 2001; 11: 1950 -1957. [PubMed] .
- 24. Lin K, Hsin H, Libina N, Kenyon C. Regulation of the Caenorhabditis elegans longevity protein DAF-16 by insulin/IGF-1 and germline signaling. Nat Genet. 2001; 28: 139 -145. [PubMed] .
- 25. Berdichevsky A, Viswanathan M, Horvitz HR, Guarente L. C. elegans SIR-2.1 interacts with 14-3-3 proteins to activate DAF-16 and extend life span. Cell. 2006; 125: 1165 -1177. [PubMed] .
- 26. Li J, Tewari M, Vidal M, Lee SS. The 14-3-3 protein FTT-2 regulates DAF-16 in Caenorhabditis elegans. Dev Biol. 2007; 301: 82 -91. [PubMed] .
- 27. Oh SW, Mukhopadhyay A, Svrzikapa N, Jiang F, Davis RJ, Tissenbaum HA. JNK regulates lifespan in Caenorhabditis elegans by modulating nuclear translocation of forkhead transcription factor/DAF-16. Proc Natl Acad Sci U S A. 2005; 102: 4494 -4499. [PubMed] .
- 28. Lehtinen MK, Yuan Z, Boag PR, Yang Y, Villen J, Becker EB, DiBacco S, de la Iglesia N, Gygi S, Blackwell TK, Bonni A. A conserved MST-FOXO signaling pathway mediates oxidative-stress responses and extends life span. Cell. 2006; 125: 987 -1001. [PubMed] .
- 29. Berman JR and Kenyon C. Germ-cell loss extends C. elegans life span through regulation of DAF-16 by kri-1 and lipophilic-hormone signaling. Cell. 2006; 124: 1055 -1068. [PubMed] .
- 30. Yamawaki TM, Berman JR, Suchanek-Kavipurapu M, McCormick M, Maria Gaglia M, Lee SJ, Kenyon C. The Somatic Reproductive Tissues of C. elegans Promote Longevity through Steroid Hormone Signaling. PLoS Biol. 2010; 8: e1000468 [PubMed] .
- 31. Motola DL, Cummins CL, Rottiers V, Sharma KK, Li T, Li Y, Suino-Powell K, Xu HE, Auchus RJ, Antebi A, Mangelsdorf DJ. Identification of ligands for DAF-12 that govern dauer formation and reproduction in C. elegans. Cell. 2006; 124: 1209 -1223. [PubMed] .
- 32. Hu PJ. Dauer. WormBook. 2007; 1 -19. [PubMed] .
- 33. Alam H, Williams TW, Dumas KJ, Guo C, Yoshina S, Mitani S, Hu PJ. EAK-7 Controls Development and Life Span by Regulating Nuclear DAF-16/FoxO Activity. Cell Metab. 2010; 12: 30 -41. [PubMed] .
- 34. Dumas KJ, Guo C, Wang X, Burkhart KB, Adams EJ, Alam H, Hu PJ. Functional divergence of dafachronic acid pathways in the control of C. elegans development and lifespan. Dev Biol. 2010; 340: 605 -612. [PubMed] .
- 35. Zhang Y, Xu J, Puscau C, Kim Y, Wang X, Alam H, Hu PJ. Caenorhabditis elegans EAK-3 inhibits dauer arrest via nonautonomous regulation of nuclear DAF-16/FoxO activity. Dev Biol. 2008; 315: 290 -302. [PubMed] .
- 36. Hu PJ, Xu J, Ruvkun G. Two membrane-associated tyrosine phosphatase homologs potentiate C. elegans AKT-1/PKB signaling. PLoS Genet. 2006; 2: e99 [PubMed] .
- 37. Patel DS, Fang LL, Svy DK, Ruvkun G, Li W. Genetic identification of HSD-1, a conserved steroidogenic enzyme that directs larval development in Caenorhabditis elegans. Development. 2008; 135: 2239 -2249. [PubMed] .
- 38. Rottiers V, Motola DL, Gerisch B, Cummins CL, Nishiwaki K, Mangelsdorf DJ, Antebi A. Hormonal control of C. elegans dauer formation and life span by a Rieske-like oxygenase. Dev Cell. 2006; 10: 473 -482. [PubMed] .
- 39. Ohkura K, Suzuki N, Ishihara T, Katsura I. SDF-9, a protein tyrosine phosphatase-like molecule, regulates the L3/dauer developmental decision through hormonal signaling in C. elegans. Development. 2003; 130: 3237 -3248. [PubMed] .
- 40. Gerisch B, Rottiers V, Li D, Motola DL, Cummins CL, Lehrach H, Mangelsdorf DJ, Antebi A. A bile acid-like steroid modulates Caenorhabditis elegans lifespan through nuclear receptor signaling. Proc Natl Acad Sci U S A. 2007; 104: 5014 -5019. [PubMed] .
- 41. Tsai WC, Bhattacharyya N, Han LY, Hanover JA, Rechler MM. Insulin inhibition of transcription stimulated by the forkhead protein Foxo1 is not solely due to nuclear exclusion. Endocrinology. 2003; 144: 5615 -5622. [PubMed] .
- 42. Ambrogini E, Almeida M, Martin-Millan M, Paik JH, Depinho RA, Han L, Goellner J, Weinstein RS, Jilka RL, O'Brien CA, Manolagas SC. FoxO-mediated defense against oxidative stress in osteoblasts is indispensable for skeletal homeostasis in mice. Cell Metab. 2010; 11: 136 -146. [PubMed] .
- 43. Su AI, Wiltshire T, Batalov S, Lapp H, Ching KA, Block D, Zhang J, Soden R, Hayakawa M, Kreiman G, Cooke MP, Walker JR, Hogenesch JB. A gene atlas of the mouse and human protein-encoding transcriptomes. Proc Natl Acad Sci U S A. 2004; 101: 6062 -6067. [PubMed] .
- 44. Aoyama H, Daitoku H, Fukamizu A. Nutrient control of phosphorylation and translocation of Foxo1 in C57BL/6 and db/db mice. Int J Mol Med. 2006; 18: 433 -439. [PubMed] .


