Introduction
Aging is defined as a decline caused by accumulation of all sorts of damage, in particular, molecular damage. This statement seemed so obvious that it was not questioned. Yet several lines of evidence rule out molecular damage as a cause of aging [1-15]. Yes, of course, molecular damage accumulates over time. But this accumulation is not sufficient to cause organismal death. Eventually it would. But the organism does not live long enough, because another cause terminates life first [8]. This cause is aging, a continuation of developmental growth. Definitely, developmental growth is not driven by accumulation of molecular damage, although molecular damage accumulates. Similarly, aging is not driven by damage.
Growth is stimulated in part by mitogen- and nutrient-sensing (and other) signaling pathways such as mTOR [16-35]. Aging, “an aimless continuation of developmental program”, is driven by the same signaling pathways including mTOR [8, 14, 24]. Aging in turn causes damage: not molecular damage but non-random organ damage (stroke, infarction, renal failure and so on) and death [13]. Seemingly, one objection to this concept is that cancer is caused by molecular damage. And cancer is often a cause of death in mammals. So how may one claim that damage does not drive aging, if it is involved in cancer. Let us discuss this.
Damage in cancer
Damage causes activate oncogenes and de-activate tumor suppressors due to genetic mutations, epigenetic alterations and microRNAs dysregulation [36-57]. Even according to alternative theories, cancer is caused by damage too [58]. So damage is involved in cancer. There are some exceptions, mostly related to embryonic cells. Also, in theory, extra-genetic alterations such as stable activation of oncogenic pathways via positive feedback loops can contribute to malignant phenotype [59]. Finally, positive feedback loops could be established between cancer and normal cells [59-61]. But in general molecular damage is a key factor in cancer origin. In agreement, cancer is associated with genetic instability [59, 62-69].
Not decline but robustness
Due to genetic instability, cancer cells accumulate high levels of unrepaired damage, resulting in genomic mutations and epigenetic alterations as well as aneuploidy [36-49, 70-80]. Despite of accumulation of damage, cancer is neither decline nor ‘wear and tear’. Cancer cells are robust and aggressive. Cancer cells damage organs, thus killing organism. If cancer cells with all damage are so robust, then how possibly aging of normal cells could be “a decline due to accumulation of molecular damage”. In fact, it does not.
Immortality of cancer cells
Cancer is associated with cellular immortality [38, 81-88]. Not only cancer cells can become cell lines but also they can become free-living organisms [89-96]. Such free-living cancer cells spread from one animal to another. Thus, venereal sarcoma in dogs spread as unicellular mammalian organisms for several millennia, once originated from a single cancer cell [89-96]. Thus accumulation of damage is associated with cellular immortality.
Damage is not sufficient to cause cancer
However, molecular damage is not sufficient either to cause cancer or to hurt organism. This damage is multiplied billions of times via cell replication. Also, cells with random mutations undergo non-random selection (Figure 1).
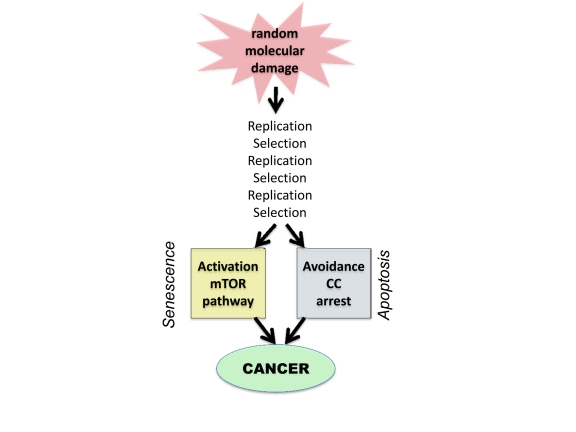
Figure 1. From random damage to cancer Random damage undergoes multiple rounds of replication and selection. Aging is one of selection forces that favors cells with oncogenic mutations. Cancer cell is characterized by (a) activation of growth-promoting pathways such as mTOR and (b) loss of cell cycle (CC) control. Isolated activation of mTOR favors senescence, whereas isolated cell cycle progression may trigger apoptosis.
Multiplication and selection
A 1 cm tumor contains 109 (1 billion) cells. Therefore, damage does not passively accumulate but is actively multiplied. Cells undergo clonal selection, analogous to Darwinian selection [70, 97-100]. Importantly, most mutations are so called “passenger” mutations that remain random and useless [72, 79, 80, 101]. But nevertheless they do not decrease cell vitality.
Selective microenvironment
Oncogenic mutations occur randomly. Cancer arises when cellular microenvironment favors oncogenic mutations, creating selective advantage to cells bearing oncogenic mutations. For example, carcinogens not only damage DNA but also cytostatic to normal cells, thus favoring selection of oncogenic mutations that render cells resistant to cytostatic/toxic carcinogens [102, 103]. This is especially apparent with non-damaging carcinogens such as phorbol esters [104]. Cancer therapy can select for additional oncogenic mutations (such as loss of p53), rendering cancer cells not only drug resistant but also increasingly oncogenic [102, 103, 105-108]. Inflammation and chronic infections also favor cancer [109-121]. And the aging microenvironment favors cancer [122-128].
Aging as selective force
Organismal aging is the most important risk factor in common cancers such as prostate, breast, colon, gastric, lung, pancreatic, skin, brain, thyroid (and so on) cancers as well as melanomas and certain leukemias. Calorie restriction [129-137] and rapamycin [138-141], which decelerate aging, also postpone cancer. Why does aging favors cancer? One explanation is that aging stromal cells secrete factors that promote growth of pre-cancer cells [122, 123, 142-144] and aging is associated with pro-inflammation that favors cancer growth [145-147]. The pro-inflammatory NF-kB pathway is involved in both DNA damage response (DDR), cancer and aging [60, 147-156].
One additional explanation is that chronic overactivation of mTOR renders normal cells irresponsive to growth factors [157]. (In fact, mTOR/S6K renders cells resistant to insulin and growth factors [158, 159]). Then, cancer cells, which are growth signal- independent, acquire selective advantage. In theory, by restoring responsiveness of normal cells to mitogenic signals, treatment with rapamycin can eliminate selective advantage for cancer cells. It was predicted that rapamycin can restore responsiveness of aging cells [157]. In fact, mTOR may cause exhaustion of the proliferative potential of stem cells and, in some studies, rapamycin improved the responsiveness of aging stem cells and immune cells [160-163]. As an example, activation of mTOR promoted leukemia-initiated cells, while depleting normal hematopoietic stem cell. Rapamycin not only depleted leukaemia-initiating cells but also restored normal stem cell function [160, 164]. Thus decreased proliferative potential of normal cells is associated with selective advantage to cancer cells.
Non-random activation of the PI3K/mTOR pathway
The PI3K/mTOR pathway is universally involved in cancer [37, 165-180]. It is activated by mutations in PI3K, Ras, Raf, non-receptor and growth factor receptor kinases and autocrine growth factors [165, 177, 181, 182]. Also, inactivation of tumor suppressors such as PTEN, AMPK, TSC2, LKB1, NF1 causes activation of this pathway [160, 169, 183-191]. In addition, the hypertrophic effect is often achieved via activation of downstream mTOR targets, translation factors [178]. Finally, p53, which is lost in cancer, is also a suppressor of the mTOR pathway [192-201]. Therefore, it can suppress conversion of cell cycle arrest to senescence [198-204]. In turn, the GF/PI3K/Akt/mTOR pathway drives cellular mass growth, hypersecrtory phenotype, HIF-1 expression, angiogenic phenotype, high levels of glycolysis and biosyntesis (metabolic switch) and apoptosis avoidance [16-35, 205-208]. In other words, it is involved in most of hallmarks of cancer [38, 88], with a notable exception of loss of cell cycle control. On the other hand, the mTOR pathway is involved in senescent phenotype. Therefore, the second alteration in cancer is deactivation of cell cycle checkpoints. Thus cancer cells can be viewed as cycling senescent cells.
Avoiding cell cycle arrest
In order to proliferate, cell with TOR-activating oncogenes must disable cell cycle control. Inactivation of tumor suppressors such p53, Rb, p16 and activation of c-myc, cyclins D and E, all disable cell cycle control, allowing “pro-senescent” cancer cell to proliferate [209-216]. Still, acute DNA damage, anticancer drugs and induction of p21 or p16 cause cell cycle arrest. Arrested cancer cells rapidly become senescent (geroconversion), revealing their pro-senescent phenotype.
Oncogenic transformation and gerogenic conversion
There are non-mutually exclusive ways to depict oncogenic transformation, as complementary activation/disabling of signaling pathways [88, 217-225]. Here to compare cancer with aging, I view oncogenic transformation as (a) activation of growth-promoting pathways such as mTOR and (b) loss of cell cycle control. Growth promoting pathways can drive either growth or aging, whereas avoidance of cell cycle arrest precludes aging (Fig. 1). In quiescent cells, activation of growth-promoting pathways (such as mTOR) converts quiescence into senescence, a process named gerogenic conversion or geroconversion [226, 227]. In proliferating cells, mTOR is fully activated. Induction of cell cycle arrest, without inhibition of mTOR causes gerogenic conversion too. When cell cycle is arrested, growth-promoting pathways drive hypertrophy and aging instead of growth. The difference between quiescence and senescence was recently discussed in detail [227]. Cellular hyper-functions and feedback signal resistance are manifestations of cellular senescence/aging that lead to age-related diseases [227]. These hallmarks result from excessive activation of signaling pathways not from accumulation of damage.
Why aging is not caused by accumulation of damage
To harbor the active mTOR pathway, cancer cells undergo multiple rounds of selection. In other words, numerous random mutations are selected for non-random activation of mTOR. In contrast it is resting non-dividing cells such as liver, muscle, fat, connective tissue, neurons that undergo aging (geroconversion) in the organism. Not only levels of molecular damage are low in normal cells, but also there is no amplification and selection. So random damage hardly can cause non-random activation of mTOR. Noteworthy, calorie restriction (CR) inhibits mTOR. Even short-term CR suppresses cellular senescence in the organism [228, 229].
Extragenetic activation of mTOR in aging
mTOR pathway is activated by growth factors, hormones, mitogens, pro-inflammatory cytokines and other secretory molecules and nutrients. Cells can overactivate each other, via positive feedback loops. For example in the liver and fat, hyper-active mTOR causes insulin-resistance, which in turn leads to activation mTOR in beta-cells, which produce insulin. Insulin further activates mTOR in the liver and fat.
DNA damage response (DDR) and aging
In proliferating cells, mTOR is fully activated. Acute DNA damage induces DDR and cell cycle arrest. If mTOR is still active, such cells undergo geroconversion. Rapamycin and other inhibitors of the mTOR pathway decelerate geroconversion [198, 200, 206, 226, 230-236]). This is how accelerated senescence is usually induced in proliferating cells (in cell culture). However, in quiescent cells with inactive mTOR, DNA damage does not induce sensecence, whereas activation of mTOR does [226, 237].
In oncogene-induced senescence (OIS), DDR causes cell cycle arrest, leading to senescence [238-245]. Noteworthy, most oncogenes that induce senescence (Ras, Raf, MEK, Akt and so on) activate the mTOR pathway. We can call them TOR-activating oncogenes or gerogenes [14], because they are involved in aging from cells to organisms [14, 246, 247]. Loss of PTEN also activates the mTOR pathway, causing senescence [243]. In OIS, oncogenes induce cell cycle arrest but not necessary DNA damage or even DDR [248, 243, 249]. Furthermore, atypical DDR can occur without DNA damage (pseudo-DDR) [231, 236, 250-256]. DDR pathways and the mTOR pathway are interconnected [257-260]. And it seems that pseudo-DDR and DDR are markers of cellular hyper-activation associated with senescence [145] and can be blocked by rapamycin [231].
Cancer prevention and therapy
Prevention of DNA damage can decrease cancer incidence. For example, non-smoking prevents smoking-induced cancer. Also, cancer can be prevented by decelerating the aging process by calorie restriction and rapamycin. Both calorie restriction and rapamycin delay cancer. Although rapalogs can directly affect cancer cells, rapalogs are only modestly effective as anti-cancer therapy [168, 261, 262], compared with their dramatic preventive effects. In any case, cancer can be prevented without decreasing levels of molecular damage. Furthermore, DNA damaging drugs are cornerstone of cancer therapy. And these drugs are also carcinogens, because anti-cancer and carcinogenic effects are two sides of the same coin [103].
Conclusion
Although molecular damage is typically necessary for cancer initiation, this damage limits life span not because of cellular decline but because of cellular robustness. Damage undergoes multiplication and selection. Aging by itself is a selective force that favors cancer probably because aging cells are signal resistant, thus providing selective advantage to cells that by-pass the need in mitogenic signals. In addition to non-random selection for oncogenic mutations, cancer cells accumulate even higher levels of random “passenger” mutations. Despite that cancer cells are robust. It must be expected that a lower rate of DNA damage in normal cells cannot cause cellular decline. Yes, molecular damage accumulates but is not a driving force for aging. Aging would occur in the absence of any molecular damage. On the other hand, yes, molecular damage is involved in something like cancer that can limit lifespan in mammals to some extend. Noteworthy, worms and flies do not die from cancer. Still they undergo PI3K/TOR-dependent aging [263-269].
As already discussed, if quasi-programmed TOR-driven aging would be eliminated, thus extending lifespan, then accumulation of molecular damage would become life-limiting [10]. In any case, in mammals, cellular aging (characterized by cellular overactivation, hyperfunction and secondary signal resistance) can cause diseases, which lead to organ damage. And cancer, an age-related disease, is not an exception: it kills not because cancer cells fail due to decline but because these cells damage organs. Perhaps, cancer is not the only one damage-related disease among aging-dependent conditions. But a subtle interference of molecular damage with TOR-driven aging will be a topic for another article, which will discuss the intricate relationship between non-random organ damage and random molecular damage.
Conflicts of Interest
The author of this manuscript has no conflict of interest to declare.
References
- 1. Doonan R, McElwee JJ, Matthijssens F, Walker GA, Houthoofd K, Back P, Matscheski A, Vanfleteren JR, Gems D. Against the oxidative damage theory of aging: superoxide dismutases protect against oxidative stress but have little or no effect on life span in Caenorhabditis elegans. Genes Dev. 2008; 22: 3236 -3241. [PubMed] .
- 2. Gems D and Doonan R. Antioxidant defense and aging in C. elegans: Is the oxidative damage theory of aging wrong? Cell Cyce. 2009; 8: 1681 -1687. .
- 3. Cabreiro F, Ackerman D, Doonan R, Araiz C, Back P, Papp D, Braeckman BP, Gems D. Increased life span from overexpression of superoxide dismutase in Caenorhabditis elegans is not caused by decreased oxidative damage. Free Radic Biol Med. 2011; 51: 1575 -1582. [PubMed] .
- 4. Lapointe J and Hekimi S. When a theory of aging ages badly. Cell Mol Life Sci. 2009; 67: 1 -8. [PubMed] .
- 5. Van Raamsdonk JM, Meng Y, Camp D, Yang W, Jia X, Benard C, Hekimi S. Decreased energy metabolism extends life span in Caenorhabditis elegans without reducing oxidative damage. Genetics. 2010; 185: 559 -571. [PubMed] .
- 6. Speakman JR and Selman C. The free-radical damage theory: Accumulating evidence against a simple link of oxidative stress to ageing and lifespan. Bioessays. 2011; 33: 255 -259. [PubMed] .
- 7. Ristow M and Schmeisser S. Extending life span by increasing oxidative stress. Free Radic Biol Med. 2011; 51: 327 -336. [PubMed] .
- 8. Blagosklonny MV. Aging and immortality: quasi-programmed senescence and its pharmacologic inhibition. Cell Cycle. 2006; 5: 2087 -2102. [PubMed] .
- 9. Blagosklonny MV. An anti-aging drug today: from senescence-promoting genes to anti-aging pill. Drug Disc Today. 2007; 12: 218 -224. .
- 10. Blagosklonny MV. Program-like aging and mitochondria: instead of random damage by free radicals. J Cell Biochem. 2007; 102: 1389 -1399. [PubMed] .
- 11. Blagosklonny MV. Paradoxes of aging. Cell Cycle. 2007; 6: 2997 -3003. [PubMed] .
- 12. Blagosklonny MV. Aging: ROS or TOR. Cell Cycle. 2008; 7: 3344 -3354. [PubMed] .
- 13. Blagosklonny MV. mTOR-driven aging: speeding car without brakes. Cell Cycle. 2009; 8: 4055 -4059. [PubMed] .
- 14. Blagosklonny MV. Revisiting the antagonistic pleiotropy theory of aging: TOR-driven program and quasi-program. Cell Cycle. 2010; 9: 3151 -3156. [PubMed] .
- 15. Blagosklonny MV. Why men age faster but reproduce longer than women: mTOR and evolutionary perspectives. Aging. 2010; 2: 265 -273. [PubMed] .
- 16. Hay N and Sonenberg N. Upstream and downstream of mTOR. Genes Dev. 2004; 18: 1926 -1945. [PubMed] .
- 17. Sarbassov dos D, Ali SM, Sabatini DM. Growing roles for the mTOR pathway. Curr Opin Cell Biol. 2005; 17: 596 -603. [PubMed] .
- 18. Inoki K, Corradetti MN, Guan KL. Dysregulation of the TSC-mTOR pathway in human disease. Nat Genet. 2005; 37: 19 -24. [PubMed] .
- 19. Wullschleger S, Loewith R, Hall MN. TOR signaling in growth and metabolism. Cell. 2006; 124: 471 -484. [PubMed] .
- 20. Tee AR and Blenis J. mTOR, translational control and human disease. Semin Cell Dev Biol. 2005; 16: 29 -37. [PubMed] .
- 21. Dann SG, Selvaraj A, Thomas G. mTOR Complex1-S6K1 signaling: at the crossroads of obesity, diabetes and cancer. Trends Mol Med. 2007; 13: 252 -259. [PubMed] .
- 22. Hands SL, Proud CG, Wyttenbach A. mTOR's role in ageing: protein synthesis or autophagy? Aging. 2009; 1: 586 -597. [PubMed] .
- 23. Glazer HP, Osipov RM, Clements RT, Sellke FW, Bianchi C. Hypercholesterolemia is associated with hyperactive cardiac mTORC1 and mTORC2 signaling. Cell Cycle. 2009; 8: 1738 -1746. [PubMed] .
- 24. Blagosklonny MV and Hall MN. Growth and aging: a common molecular mechanism. Aging. 2009; 1: 357 -362. [PubMed] .
- 25. Kapahi P, Chen D, Rogers AN, Katewa SD, Li PW, Thomas EL, Kockel L. With TOR, less is more: a key role for the conserved nutrient-sensing TOR pathway in aging. Cell Metab. 2010; 11: 453 -465. [PubMed] .
- 26. Boguta M. Control of RNA polymerases I and III by the TOR signaling pathway. Cell Cycle. 2009; 8: 4023 -4024. [PubMed] .
- 27. Jiang Y. mTOR goes to the nucleus. Cell Cycle. 2009; 9: 868 [PubMed] .
- 28. Dazert E and Hall MN. mTOR signaling in disease. Curr Opin Cell Biol. 2011; 23: 744 -55. [PubMed] .
- 29. Loewith R and Hall MN. Target of Rapamycin (TOR) in Nutrient Signaling and Growth Control. Genetics. 2011; 189: 1177 -1201. [PubMed] .
- 30. Zoncu R, Efeyan A, Sabatini DM. mTOR: from growth signal integration to cancer, diabetes and ageing. Nat Rev Mol Cell Biol. 2010; 12: 21 -35. [PubMed] .
- 31. Conn CS and Qian SB. mTOR signaling in protein homeostasis: less is more? Cell Cycle. 2011; 10: 1940 -1947. [PubMed] .
- 32. Ma XM and Blenis J. Molecular mechanisms of mTOR-mediated translational control. Nat Rev Mol Cell Biol. 2009; 10: 307 -318. [PubMed] .
- 33. Proud CG. mTOR-mediated regulation of translation factors by amino acids. Biochem Biophys Res Commun. 2004; 313: 429 -436. [PubMed] .
- 34. Hall MN. mTOR-what does it do? Transplant Proc. 2008; 40: S5 -8. [PubMed] .
- 35. Magnuson B, Ekim B, Fingar DC. Regulation and function of ribosomal protein S6 kinase (S6K) within mTOR signalling networks. Biochem J. 2012; 441: 1 -21. [PubMed] .
- 36. Vogelstein B and Kinzler KW. The multistep nature of cancer. Trends Genet. 1993; 9: 138 -141. [PubMed] .
- 37. Vogelstein B and Kinzler KW. Cancer genes and the pathways they control. Nat Med. 2004; 10: 789 -799. [PubMed] .
- 38. Hanahan D and Weinberg RA. The hallmarks of cancer. Cell. 2000; 100: 57 -70. [PubMed] .
- 39. Jackson SP and Bartek J. The DNA-damage response in human biology and disease. Nature. 2009; 461: 1071 -1078. [PubMed] .
- 40. Lukas J, Lukas C, Bartek J. More than just a focus: The chromatin response to DNA damage and its role in genome integrity maintenance. Nat Cell Biol. 2011; 13: 1161 -1169. [PubMed] .
- 41. Baylin SB and Herman JG. DNA hypermethylation in tumorigenesis: epigenetics joins genetics. Trends Genet. 2000; 16: 168 -174. [PubMed] .
- 42. Egger G, Liang G, Aparicio A, Jones PA. Epigenetics in human disease and prospects for epigenetic therapy. Nature. 2004; 429: 457 -463. [PubMed] .
- 43. McKenna ES and Roberts CW. Epigenetics and cancer without genomic instability. Cell Cycle. 2009; 8: 23 -26. [PubMed] .
- 44. Timp W, Levchenko A, Feinberg AP. A new link between epigenetic progenitor lesions in cancer and the dynamics of signal transduction. Cell Cycle. 2009; 8: 383 -390. [PubMed] .
- 45. Johnson SM, Grosshans H, Shingara J, Byrom M, Jarvis R, Cheng A, Labourier E, Reinert KL, Brown D, Slack FJ. RAS is regulated by the let-7 microRNA family. Cell. 2005; 120: 635 -647. [PubMed] .
- 46. Calin GA, Ferracin M, Cimmino A, Di Leva G, Shimizu M, Wojcik SE, Iorio MV, Visone R, Sever NI, Fabbri M, Iuliano R, Palumbo T, Pichiorri F, Roldo C, Garzon R, Sevignani C, et al. A MicroRNA signature associated with prognosis and progression in chronic lymphocytic leukemia. N Engl J Med. 2005; 353: 1793 -1801. [PubMed] .
- 47. Lujambio A and Esteller M. How epigenetics can explain human metastasis: a new role for microRNAs. Cell Cycle. 2009; 8: 377 -382. [PubMed] .
- 48. Burdach S, Plehm S, Unland R, Dirksen U, Borkhardt A, Staege MS, Muller-Tidow C, Richter GH. Epigenetic maintenance of stemness and malignancy in peripheral neuroectodermal tumors by EZH2. Cell Cycle. 2009; 8: 1991 -1996. [PubMed] .
- 49. Chen SS, Sherman MH, Hertlein E, Johnson AJ, Teitell MA, Byrd JC, Plass C. Epigenetic alterations in a murine model for chronic lymphocytic leukemia. Cell Cycle. 2009; 8: 3663 -3667. [PubMed] .
- 50. Esquela-Kerscher A and Slack FJ. Oncomirs - microRNAs with a role in cancer. Nat Rev Cancer. 2006; 6: 259 -269. [PubMed] .
- 51. Kundu ST, Nallur S, Paranjape T, Boeke M, Weidhaas JB, Slack FJ. KRAS alleles: The LCS6 3'UTR variant and KRAS coding sequence mutations in the NCI-60 panel. Cell Cycle. 2012; 11: .
- 52. Kasinski AL and Slack FJ. Epigenetics and genetics. MicroRNAs en route to the clinic: progress in validating and targeting microRNAs for cancer therapy. Nat Rev Cancer. 2011; 11: 849 -864. [PubMed] .
- 53. Croce CM. Causes and consequences of microRNA dysregulation in cancer. Nat Rev Genet. 2009; 10: 704 -714. [PubMed] .
- 54. Sozzi G, Pastorino U, Croce CM. MicroRNAs and lung cancer: from markers to targets. Cell Cycle. 2011; 10: 2045 -2046. [PubMed] .
- 55. Pekarsky Y and Croce CM. Is miR-29 an oncogene or tumor suppressor in CLL? Oncotarget. 2010; 1: 224 -227. [PubMed] .
- 56. Valastyan S and Weinberg RA. miR-31: a crucial overseer of tumor metastasis and other emerging roles. Cell Cycle. 2010; 9: 2124 -2129. [PubMed] .
- 57. Mavrakis KJ and Wendel HG. TargetScreen: an unbiased approach to identify functionally important microRNA targets. Cell Cycle. 2010; 9: 2080 -2084. [PubMed] .
- 58. Duesberg P and Li R. Multistep carcinogenesis: a chain reaction of aneuploidizations. Cell Cycle. 2003; 2: 202 -210. [PubMed] .
- 59. Blagosklonny MV. Molecular theory of cancer. Cancer Biol Ther. 2005; 4: 621 -627. [PubMed] .
- 60. Martinez-Outschoorn UE, Trimmer C, Lin Z, Whitaker-Menezes D, Chiavarina B, Zhou J, Wang C, Pavlides S, Martinez-Cantarin MP, Capozza F, Witkiewicz AK, Flomenberg N, Howell A, Pestell RG, Caro J, Lisanti MP, et al. Autophagy in cancer associated fibroblasts promotes tumor cell survival: Role of hypoxia, HIF1 induction and NFkappaB activation in the tumor stromal microenvironment. Cell Cycle. 2010; 9: 3515 -3533. [PubMed] .
- 61. Liu D, Martin V, Fueyo J, Lee OH, Xu J, Cortes-Santiago N, Alonso MM, Aldape K, Colman H, Gomez-Manzano C. Tie2/TEK modulates the interaction of glioma and brain tumor stem cells with endothelial cells and promotes an invasive phenotype. Oncotarget. 2010; 1: 700 -709. [PubMed] .
- 62. Lengauer C, Kinzler KW, Vogelstein B. Genetic instabilities in human cancers. Nature. 1998; 396: 643 -649. [PubMed] .
- 63. Komarova NL, Lengauer C, Vogelstein B, Nowak MA. Dynamics of genetic instability in sporadic and familial colorectal cancer. Cancer Biol Ther. 2002; 1: 685 -692. [PubMed] .
- 64. Li L, Borodyansky L, Yang Y. Genomic instability en route to and from cancer stem cells. Cell Cycle. 2009; 8: 1000 -1002. [PubMed] .
- 65. Gupta A, Yang Q, Pandita RK, Hunt CR, Xiang T, Misri S, Zeng S, Pagan J, Jeffery J, Puc J, Kumar R, Feng Z, Powell SN, Bhat A, Yaguchi T, Wadhwa R, et al. Cell cycle checkpoint defects contribute to genomic instability in PTEN deficient cells independent of DNA DSB repair. Cell Cycle. 2009; 8: 2198 -2210. [PubMed] .
- 66. Meyn RE. Linking PTEN with genomic instability and DNA repair. Cell Cycle. 2009; 8: 2322 -2323. [PubMed] .
- 67. Tuduri S, Crabbe L, Tourriere H, Coquelle A, Pasero P. Does interference between replication and transcription contribute to genomic instability in cancer cells? Cell Cycle. 2010; 9: 1886 -1892. [PubMed] .
- 68. Guirouilh-Barbat JK, Wilhelm T, Lopez BS. AKT1/BRCA1 in the control of homologous recombination and genetic stability: the missing link between hereditary and sporadic breast cancers. Oncotarget. 2010; 1: 691 -699. [PubMed] .
- 69. Ho CC, Hau PM, Marxer M, Poon RY. The requirement of p53 for maintaining chromosomal stability during tetraploidization. Oncotarget. 2010; 1: 583 -595. [PubMed] .
- 70. Beerenwinkel N, Antal T, Dingli D, Traulsen A, Kinzler KW, Velculescu VE, Vogelstein B, Nowak MA. Genetic progression and the waiting time to cancer. PLoS Comput Biol. 2007; 3: e225 [PubMed] .
- 71. Wood LD, Parsons DW, Jones S, Lin J, Sjoblom T, Leary RJ, Shen D, Boca SM, Barber T, Ptak J, Silliman N, Szabo S, Dezso Z, Ustyanksky V, Nikolskaya T, Nikolsky Y, et al. The genomic landscapes of human breast and colorectal cancers. Science. 2007; 318: 1108 -1113. [PubMed] .
- 72. Carter H, Chen S, Isik L, Tyekucheva S, Velculescu VE, Kinzler KW, Vogelstein B, Karchin R. Cancer-specific high-throughput annotation of somatic mutations: computational prediction of driver missense mutations. Cancer Res. 2009; 69: 6660 -6667. [PubMed] .
- 73. Zhao L and Vogt PK. Hot-spot mutations in p110alpha of phosphatidylinositol 3-kinase (pI3K): differential interactions with the regulatory subunit p85 and with RAS. Cell Cycle. 2010; 9: 596 -600. [PubMed] .
- 74. Martinez AC and van Wely KH. Are aneuploidy and chromosome breakage caused by a CINgle mechanism? Cell Cycle. 2010; 9: 2275 -2280. [PubMed] .
- 75. McClelland SE, Burrell RA, Swanton C. Chromosomal instability: a composite phenotype that influences sensitivity to chemotherapy. Cell Cycle. 2009; 8: 3262 -3266. [PubMed] .
- 76. Baker DJ and van Deursen JM. Chromosome missegregation causes colon cancer by APC loss of heterozygosity. Cell Cycle. 2010; 9: 1711 -1716. [PubMed] .
- 77. Chen Z, Feng J, Saldivar JS, Gu D, Bockholt A, Sommer SS. EGFR somatic doublets in lung cancer are frequent and generally arise from a pair of driver mutations uncommonly seen as singlet mutations: one-third of doublets occur at five pairs of amino acids. Oncogene. 2008; 27: 4336 -4343. [PubMed] .
- 78. Torkamani A and Schork NJ. Prediction of cancer driver mutations in protein kinases. Cancer Res. 2008; 68: 1675 -1682. [PubMed] .
- 79. Loriaux MM, Levine RL, Tyner JW, Frohling S, Scholl C, Stoffregen EP, Wernig G, Erickson H, Eide CA, Berger R, Bernard OA, Griffin JD, Stone RM, Lee B, Meyerson M, Heinrich MC, et al. High-throughput sequence analysis of the tyrosine kinome in acute myeloid leukemia. Blood. 2008; 111: 4788 -4796. [PubMed] .
- 80. Frohling S, Scholl C, Levine RL, Loriaux M, Boggon TJ, Bernard OA, Berger R, Dohner H, Dohner K, Ebert BL, Teckie S, Golub TR, Jiang J, Schittenhelm MM, Lee BH, Griffin JD, et al. Identification of driver and passenger mutations of FLT3 by high-throughput DNA sequence analysis and functional assessment of candidate alleles. Cancer Cell. 2007; 12: 501 -513. [PubMed] .
- 81. Counter CM, Avilion AA, LeFeuvre CE, Stewart NG, Greider CW, Harley CB, Bacchetti S. Telomere shortening associated with chromosome instability is arrested in immortal cells which express telomerase activity. Embo J. 1992; 11: 1921 -1929. [PubMed] .
- 82. Harley CB, Kim NW, Prowse KR, Weinrich SL, Hirsch KS, West MD, Bacchetti S, Hirte HW, Counter CM, Greider CW, et al. Telomerase, cell immortality, and cancer. Cold Spring Harb Symp Quant Biol. 1994; 59: 307 -315. [PubMed] .
- 83. Counter CM, Hahn WC, Wei W, Caddle SD, Beijersbergen RL, Lansdorp PM, Sedivy JM, Weinberg RA. Dissociation among in vitro telomerase activity, telomere maintenance, and cellular immortalization. Proc Natl Acad Sci U S A. 1998; 95: 14723 -14728. [PubMed] .
- 84. Hahn WC. Immortalization and transformation of human cells. Mol Cells. 2002; 13: 351 -361. [PubMed] .
- 85. Lundberg AS, Randell SH, Stewart SA, Elenbaas B, Hartwell KA, Brooks MW, Fleming MD, Olsen JC, Miller SW, Weinberg RA, Hahn WC. Immortalization and transformation of primary human airway epithelial cells by gene transfer. Oncogene. 2002; 21: 4577 -4586. [PubMed] .
- 86. Blagosklonny MV. Cell immortality and hallmarks of cancer. Cell Cycle. 2003; 2: 296 -299. [PubMed] .
- 87. Bazarov AV, Hines WC, Mukhopadhyay R, Beliveau A, Melodyev S, Zaslavsky Y, Yaswen P. Telomerase activation by c-Myc in human mammary epithelial cells requires additional genomic changes. Cell Cycle. 2009; 8: 3373 -3378. [PubMed] .
- 88. Hanahan D and Weinberg RA. Hallmarks of cancer: the next generation. Cell. 2011; 144: 646 -674. [PubMed] .
- 89. Cohen D. The canine transmissible venereal tumor: a unique result of tumor progression. Adv Cancer Res. 1985; 43: 75 -112. [PubMed] .
- 90. Liao KW, Hung SW, Hsiao YW, Bennett M, Chu RM. Canine transmissible venereal tumor cell depletion of B lymphocytes: molecule(s) specifically toxic for B cells. Vet Immunol Immunopathol. 2003; 92: 149 -162. [PubMed] .
- 91. Leroi AM, Koufopanou V, Burt A. Cancer selection. Nat Rev Cancer. 2003; 3: 226 -231. [PubMed] .
- 92. Pearse AM and Swift K. Allograft theory: transmission of devil facial-tumour disease. Nature. 2006; 439: 549 [PubMed] .
- 93. Siddle HV, Kreiss A, Eldridge MD, Noonan E, Clarke CJ, Pyecroft S, Woods GM, Belov K. Transmission of a fatal clonal tumor by biting occurs due to depleted MHC diversity in a threatened carnivorous marsupial. Proc Natl Acad Sci U S A. 2007; 104: 16221 -16226. [PubMed] .
- 94. McAloose D and Newton AL. Wildlife cancer: a conservation perspective. Nat Rev Cancer. 2009; 9: 517 -526. [PubMed] .
- 95. Murchison EP. Clonally transmissible cancers in dogs and Tasmanian devils. Oncogene. 2008; 27: Suppl 2 S19 -30. [PubMed] .
- 96. Murchison EP, Tovar C, Hsu A, Bender HS, Kheradpour P, Rebbeck CA, Obendorf D, Conlan C, Bahlo M, Blizzard CA, Pyecroft S, Kreiss A, Kellis M, Stark A, Harkins TT, Marshall Graves JA, et al. The Tasmanian devil transcriptome reveals Schwann cell origins of a clonally transmissible cancer. Science. 2010; 327: 84 -87. [PubMed] .
- 97. Schollnberger H, Beerenwinkel N, Hoogenveen R, Vineis P. Cell selection as driving force in lung and colon carcinogenesis. Cancer Res. 2010; 70: 6797 -6803. [PubMed] .
- 98. Cahill DP, Kinzler KW, Vogelstein B, Lengauer C. Genetic instability and darwinian selection in tumours. Trends Cell Biol. 1999; 9: M57 -60. [PubMed] .
- 99. Hempen PM, Zhang L, Bansal RK, Iacobuzio-Donahue CA, Murphy KM, Maitra A, Vogelstein B, Whitehead RH, Markowitz SD, Willson JK, Yeo CJ, Hruban RH, Kern SE. Evidence of selection for clones having genetic inactivation of the activin A type II receptor (ACVR2) gene in gastrointestinal cancers. Cancer Res. 2003; 63: 994 -999. [PubMed] .
- 100. Zhang Y, Italia MJ, Auger KR, Halsey WS, Van Horn SF, Sathe GM, Magid-Slav M, Brown JR, Holbrook JD. Molecular evolutionary analysis of cancer cell lines. Mol Cancer Ther. 2010; 9: 279 -291. [PubMed] .
- 101. Bozic I, Antal T, Ohtsuki H, Carter H, Kim D, Chen S, Karchin R, Kinzler KW, Vogelstein B, Nowak MA. Accumulation of driver and passenger mutations during tumor progression. Proc Natl Acad Sci U S A. 2010; 107: 18545 -18550. [PubMed] .
- 102. Blagosklonny MV. Oncogenic resistance to growth-limiting conditions. Nat Rev Cancer. 2002; 2: 221 -225. [PubMed] .
- 103. Blagosklonny MV. Carcinogenesis, cancer therapy and chemoprevention. Cell Death Differ. 2005; 12: 592 -602. [PubMed] .
- 104. Dotto GP, Parada LF, Weinberg RA. Specific growth response of ras-transformed embryo fibroblasts to tumour promoters. Nature. 1985; 318: 472 -475. [PubMed] .
- 105. Blagosklonny MV. Antiangiogenic therapy and tumor progression. Cancer Cell. 2004; 5: 13 -17. [PubMed] .
- 106. Blagosklonny MV. Why therapeutic response may not prolong the life of a cancer patient: selection for oncogenic resistance. Cell Cycle. 2005; 4: 1693 -1698. [PubMed] .
- 107. Fleenor CJ, Marusyk A, DeGregori J. Ionizing radiation and hematopoietic malignancies: altering the adaptive landscape. Cell Cycle. 2010; 9: 3005 -3011. [PubMed] .
- 108. Aziz MH, Shen H, Maki CG. Acquisition of p53 mutations in response to the non-genotoxic p53 activator Nutlin-3. Oncogene. 2011; 30: 4678 -4686. [PubMed] .
- 109. Uemura N, Okamoto S, Yamamoto S, Matsumura N, Yamaguchi S, Yamakido M, Taniyama K, Sasaki N, Schlemper RJ. Helicobacter pylori infection and the development of gastric cancer. N Engl J Med. 2001; 345: 784 -789. [PubMed] .
- 110. Coussens LM and Werb Z. Inflammation and cancer. Nature. 2002; 420: 860 -867. [PubMed] .
- 111. Karin M, Lawrence T, Nizet V. Innate immunity gone awry: linking microbial infections to chronic inflammation and cancer. Cell. 2006; 124: 823 -835. [PubMed] .
- 112. Lin WW and Karin M. A cytokine-mediated link between innate immunity, inflammation, and cancer. J Clin Invest. 2007; 117: 1175 -1183. [PubMed] .
- 113. Porta C, Subhra Kumar B, Larghi P, Rubino L, Mancino A, Sica A. Tumor promotion by tumor-associated macrophages. Adv Exp Med Biol. 2007; 604: 67 -86. [PubMed] .
- 114. Shan W and Liu J. Inflammation: a hidden path to breaking the spell of ovarian cancer. Cell Cycle. 2009; 8: 3107 -3111. [PubMed] .
- 115. Silvera D and Schneider RJ. Inflammatory breast cancer cells are constitutively adapted to hypoxia. Cell Cycle. 2009; 8: 3091 -3096. [PubMed] .
- 116. Gonda TA, Tu S, Wang TC. Chronic inflammation, the tumor microenvironment and carcinogenesis. Cell Cycle. 2009; 8: 2005 -2013. [PubMed] .
- 117. Wu Y and Zhou BP. Inflammation: a driving force speeds cancer metastasis. Cell Cycle. 2009; 8: 3267 -3273. [PubMed] .
- 118. Paradisi A and Mehlen P. Netrin-1, a missing link between chronic inflammation and tumor progression. Cell Cycle. 2010; 9: 1253 -1262. [PubMed] .
- 119. Barykova YA, Logunov DY, Shmarov MM, Vinarov AZ, Fiev DN, Vinarova NA, Rakovskaya IV, Baker PS, Shyshynova I, Stephenson AJ, Klein EA, Naroditsky BS, Gintsburg AL, Gudkov AV. Association of Mycoplasma hominis infection with prostate cancer. Oncotarget. 2011; 2: 289 -297. [PubMed] .
- 120. Lisanti MP, Martinez-Outschoorn UE, Pavlides S, Whitaker-Menezes D, Pestell RG, Howell A, Sotgia F. Accelerated aging in the tumor microenvironment: connecting aging, inflammation and cancer metabolism with personalized medicine. Cell Cycle. 2011; 10: 2059 -2063. [PubMed] .
- 121. Balliet RM, Capparelli C, Guido C, Pestell TG, Martinez-Outschoorn UE, Lin Z, Whitaker-Menezes D, Chiavarina B, Pestell RG, Howell A, Sotgia F, Lisanti MP. Mitochondrial oxidative stress in cancer-associated fibroblasts drives lactate production, promoting breast cancer tumor growth: Understanding the aging and cancer connection. Cell Cycle. 2011; 10: 4065 -4073. [PubMed] .
- 122. Krtolica A and Campisi J. Integrating epithelial cancer, aging stroma and cellular senescence. Adv Gerontol. 2003; 11: 109 -116. [PubMed] .
- 123. Campisi J. Senescent cells, tumor suppression, and organismal aging: good citizens, bad neighbors. Cell. 2005; 120: 513 -522. [PubMed] .
- 124. Vicente-Duenas C, Abollo-Jimenez F, Ruiz-Roca L, Alonso-Escudero E, Jimenez R, Cenador MB, Criado FJ, Cobaleda C, Sanchez-Garcia I. The age of the target cell affects B-cell leukaemia malignancy. Aging. 2010; 2: 908 -913. [PubMed] .
- 125. Henry CJ, Marusyk A, Zaberezhnyy V, Adane B, DeGregori J. Declining lymphoid progenitor fitness promotes aging-associated leukemogenesis. Proc Natl Acad Sci U S A. 2010; 107: 21713 -21718. [PubMed] .
- 126. Henry CJ, Marusyk A, DeGregori J. Aging-associated changes in hematopoiesis and leukemogenesis: what's the connection? Aging. 2011; 3: 643 -656. [PubMed] .
- 127. Lewis DA, Travers JB, Machado C, Somani AK, Spandau DF. Reversing the aging stromal phenotype prevents carcinoma initiation. Aging. 2011; 3: 407 -416. [PubMed] .
- 128. Campisi J, Andersen JK, Kapahi P, Melov S. Cellular senescence: A link between cancer and age-related degenerative disease? Semin Cancer Biol. 2011; 21: 354 -359. [PubMed] .
- 129. Hursting SD, Perkins SN, Phang JM. Calorie restriction delays spontaneous tumorigenesis in p53-knockout transgenic mice. Proc Natl Acad Sci U S A. 1994; 91: 7036 -7040. [PubMed] .
- 130. Berrigan D, Perkins SN, Haines DC, Hursting SD. Adult-onset calorie restriction and fasting delay spontaneous tumorigenesis in p53-deficient mice. Carcinogenesis. 2002; 23: 817 -822. [PubMed] .
- 131. Hursting SD, Smith SM, Lashinger LM, Harvey AE, Perkins SN. Calories and carcinogenesis: lessons learned from 30 years of calorie restriction research. Carcinogenesis. 2010; 31: 83 -89. [PubMed] .
- 132. Ingram DK, Zhu M, Mamczarz J, Zou S, Lane MA, Roth GS, deCabo R. Calorie restriction mimetics: an emerging research field. Aging Cell. 2006; 5: 97 -108. [PubMed] .
- 133. Ingram DK, Anson RM, de Cabo R, Mamczarz J, Zhu M, Mattison J, Lane MA, Roth GS. Development of calorie restriction mimetics as a prolongevity strategy. Ann N Y Acad Sci. 2004; 1019: 412 -423. [PubMed] .
- 134. Hursting SD, Lavigne JA, Berrigan D, Perkins SN, Barrett JC. Calorie restriction, aging, and cancer prevention: mechanisms of action and applicability to humans. Annu Rev Med. 2003; 54: 131 -152. [PubMed] .
- 135. Longo VD and Fontana L. Calorie restriction and cancer prevention: metabolic and molecular mechanisms. Trends Pharmacol Sci. 2010; 31: 89 -98. [PubMed] .
- 136. Fontana L, Partridge L, Longo VD. Extending healthy life span–from yeast to humans. Science. 2010; 328: 321 -326. [PubMed] .
- 137. Blagosklonny MV. Calorie restriction: Decelerating mTOR-driven aging from cells to organisms (including humans). Cell Cycle. 2010; 9: 683 -688. [PubMed] .
- 138. Blagosklonny MV. Prevention of cancer by inhibiting aging. Cancer Biol Ther. 2008; 7: 1520 -1524. [PubMed] .
- 139. Harrison DE, Strong R, Sharp ZD, Nelson JF, Astle CM, Flurkey K, Nadon NL, Wilkinson JE, Frenkel K, Carter CS, Pahor M, Javors MA, Fernandezr E, Miller RA. Rapamycin fed late in life extends lifespan in genetically heterogenous mice. Nature. 2009; 460: 392 -396. [PubMed] .
- 140. Anisimov VN, Zabezhinski MA, Popovich IG, Piskunova TS, Semenchenko AV, Tyndyk ML, Yurova MN, Antoch MP, Blagosklonny MV. Rapamycin extends maximal lifespan in cancer-prone mice. Am J Pathol. 2010; 176: 2092 -2097. [PubMed] .
- 141. Anisimov VN, Zabezhinski MA, Popovich IG, Piskunova TS, Semenchenko AV, Tyndyk ML, Yurova MN, Blagosklonny MV. Rapamycin increases lifespan and inhibits spontaneous tumorigenesis in inbred female mice. Cell Cycle. 2011; 10: .
- 142. Krtolica A, Parrinello S, Lockett S, Desprez PY, Campisi J. Senescent fibroblasts promote epithelial cell growth and tumorigenesis: a link between cancer and aging. Proc Natl Acad Sci U S A. 2001; 98: 12072 -12077. [PubMed] .
- 143. Parrinello S, Coppe JP, Krtolica A, Campisi J. Stromal-epithelial interactions in aging and cancer: senescent fibroblasts alter epithelial cell differentiation. J Cell Sci. 2005; 118: 485 -496. [PubMed] .
- 144. Coppe JP, Patil CK, Rodier F, Sun Y, Munoz DP, Goldstein J, Nelson PS, Desprez PY, Campisi J. Senescence-associated secretory phenotypes reveal cell-nonautonomous functions of oncogenic RAS and the p53 tumor suppressor. PLoS Biol. 2008; 6: 2853 -2868. [PubMed] .
- 145. Rodier F, Coppe JP, Patil CK, Hoeijmakers WA, Munoz DP, Raza SR, Freund A, Campeau E, Davalos AR, Campisi J. Persistent DNA damage signalling triggers senescence-associated inflammatory cytokine secretion. Nat Cell Biol. 2009; 11: 973 -979. [PubMed] .
- 146. Davalos AR, Coppe JP, Campisi J, Desprez PY. Senescent cells as a source of inflammatory factors for tumor progression. Cancer Metastasis Rev. 2011; 29: 273 -283. [PubMed] .
- 147. Gudkov AV, Gurova KV, Komarova EA. Inflammation and p53: A Tale of Two Stresses. Genes Cancer. 2011; 2: 503 -516. [PubMed] .
- 148. Karin M and Greten FR. NF-kappaB: linking inflammation and immunity to cancer development and progression. Nat Rev Immunol. 2005; 5: 749 -759. [PubMed] .
- 149. Adler AS, Kawahara TL, Segal E, Chang HY. Reversal of aging by NFkappaB blockade. Cell Cycle. 2008; 7: 556 -559. [PubMed] .
- 150. Chauncey SS, Boothman DA, Habib AA. The receptor interacting protein 1 mediates a link between NFkappaB and PI3-kinase signaling. Cell Cycle. 2009; 8: 2671 -2672. [PubMed] .
- 151. Barre B, Coqueret O, Perkins ND. Regulation of activity and function of the p52 NF-kappaB subunit following DNA damage. Cell Cycle. 2010; 9: 4795 -4804. [PubMed] .
- 152. Donato AJ, Pierce GL, Lesniewski LA, Seals DR. Role of NFkappaB in age-related vascular endothelial dysfunction in humans. Aging. 2009; 1: 678 -680. [PubMed] .
- 153. Demchenko YN and Kuehl WM. A critical role for the NFkB pathway in multiple myeloma. Oncotarget. 2010; 1: 59 -68. [PubMed] .
- 154. Ohanna M, Giuliano S, Bonet C, Imbert V, Hofman V, Zangari J, Bille K, Robert C, Bressac-de Paillerets B, Hofman P, Rocchi S, Peyron JF, Lacour JP, Ballotti R, Bertolotto C. Senescent cells develop a PARP-1 and nuclear factor-{kappa}B-associated secretome (PNAS). Genes Dev. 2011; 25: 1245 -1261. [PubMed] .
- 155. Vaughan S and Jat PS. Deciphering the role of nuclear factor-kappaB in cellular senescence. Aging. 2011; 3: 913 -919. [PubMed] .
- 156. Moskalev A and Shaposhnikov M. Pharmacological inhibition of NF-kappaB prolongs lifespan of Drosophila melanogaster. Aging. 2011; 3: 391 -394. [PubMed] .
- 157. Blagosklonny MV. Aging, stem cells, and mammalian target of rapamycin: a prospect of pharmacologic rejuvenation of aging stem cells. Rejuvenation Res. 2008; 11: 801 -808. [PubMed] .
- 158. Shah OJ, Wang Z, Hunter T. Inappropriate activation of the TSC/Rheb/mTOR/S6K cassette induces IRS1/2 depletion, insulin resistance, and cell survival deficiencies. Curr Biol. 2004; 14: 1650 -1656. [PubMed] .
- 159. Zhang H, Bajraszewski N, Wu E, Wang H, Moseman AP, Dabora SL, Griffin JD, Kwiatkowski DJ. PDGFRs are critical for PI3K/Akt activation and negatively regulated by mTOR. J Clin Invest. 2007; 117: 730 -738. [PubMed] .
- 160. Yilmaz OH, Valdez R, Theisen BK, Guo W, Ferguson DO, Wu H, Morrison SJ. Pten dependence distinguishes haematopoietic stem cells from leukaemia-initiating cells. Nature. 2006; 441: 475 -482. [PubMed] .
- 161. Chen C, Liu Y, Zheng P. The axis of mTOR-mitochondria-ROS and stemness of the hematopoietic stem cells. Cell Cycle. 2009; 8: 1158 -1160. [PubMed] .
- 162. Castilho RM, Squarize CH, Chodosh LA, Williams BO, Gutkind JS. mTOR mediates Wnt-induced epidermal stem cell exhaustion and aging. Cell Stem Cell. 2009; 5: 279 -289. [PubMed] .
- 163. Gan B and DePinho RA. mTORC1 signaling governs hematopoietic stem cell quiescence. Cell Cycle. 2009; 8: 1003 -1006. [PubMed] .
- 164. Lee JY, Nakada D, Yilmaz OH, Tothova Z, Joseph NM, Lim MS, Gilliland DG, Morrison SJ. mTOR activation induces tumor suppressors that inhibit leukemogenesis and deplete hematopoietic stem cells after Pten deletion. Cell Stem Cell. 2010; 7: 593 -605. [PubMed] .
- 165. Parsons DW, Wang TL, Samuels Y, Bardelli A, Cummins JM, DeLong L, Silliman N, Ptak J, Szabo S, Willson JK, Markowitz S, Kinzler KW, Vogelstein B, Lengauer C, Velculescu VE. Colorectal cancer: mutations in a signalling pathway. Nature. 2005; 436: 792 [PubMed] .
- 166. Shaw RJ and Cantley LC. Ras, PI(3)K and mTOR signalling controls tumour cell growth. Nature. 2006; 441: 424 -430. [PubMed] .
- 167. Janes MR and Fruman DA. Targeting TOR dependence in cancer. Oncotarget. 2010; 1: 69 -76. [PubMed] .
- 168. Markman B, Dienstmann R, Tabernero J. Targeting the PI3K/Akt/mTOR pathway–beyond rapalogs. Oncotarget. 2010; 1: 530 -543. [PubMed] .
- 169. Cully M, You H, Levine AJ, Mak TW. Beyond PTEN mutations: the PI3K pathway as an integrator of multiple inputs during tumorigenesis. Nat Rev Cancer. 2006; 6: 184 -192. [PubMed] .
- 170. Guertin DA and Sabatini DM. Defining the role of mTOR in cancer. Cancer Cell. 2007; 12: 9 -22. [PubMed] .
- 171. Courtney KD, Corcoran RB, Engelman JA. The PI3K pathway as drug target in human cancer. J Clin Oncol. 2010; 28: 1075 -1083. [PubMed] .
- 172. Schmidt-Kittler O, Zhu J, Yang J, Liu G, Hendricks W, Lengauer C, Gabelli SB, Kinzler KW, Vogelstein B, Huso DL, Zhou S. PI3Kalpha inhibitors that inhibit metastasis. Oncotarget. 2010; 1: 339 -348. [PubMed] .
- 173. Dbouk HA and Backer JM. A beta version of life: p110beta takes center stage. Oncotarget. 2010; 1: 729 -733. [PubMed] .
- 174. Martelli AM, Evangelisti C, Chiarini F, McCubrey JA. The phosphatidylinositol 3-kinase/Akt/mTOR signaling network as a therapeutic target in acute myelogenous leukemia patients. Oncotarget. 2010; 1: 89 -103. [PubMed] .
- 175. Nucera C, Lawler J, Hodin R, Parangi S. The BRAFV600E mutation: what is it really orchestrating in thyroid cancer? Oncotarget. 2010; 1: 751 -756. [PubMed] .
- 176. Zawel L. P3Kalpha: a driver of tumor metastasis? Oncotarget. 2010; 1: 315 -316. [PubMed] .
- 177. Zhang Z, Stiegler AL, Boggon TJ, Kobayashi S, Halmos B. EGFR-mutated lung cancer: a paradigm of molecular oncology. Oncotarget. 2010; 1: 497 -514. [PubMed] .
- 178. Shahbazian D, Parsyan A, Petroulakis E, Hershey J, Sonenberg N. eIF4B controls survival and proliferation and is regulated by proto-oncogenic signaling pathways. Cell Cycle. 2010; 9: 4106 -4109. [PubMed] .
- 179. Bhatia B, Nahle Z, Kenney AM. Double trouble: when sonic hedgehog signaling meets TSC inactivation. Cell Cycle. 2010; 9: 456 -459. [PubMed] .
- 180. Fujishita T, Aoki M, Taketo MM. The role of mTORC1 pathway in intestinal tumorigenesis. Cell Cycle. 2009; 8: 3684 -3687. [PubMed] .
- 181. Roux PP, Ballif BA, Anjum R, Gygi SP, Blenis J. Tumor-promoting phorbol esters and activated Ras inactivate the tuberous sclerosis tumor suppressor complex via p90 ribosomal S6 kinase. Proc Natl Acad Sci U S A. 2004; 101: 13489 -13494. [PubMed] .
- 182. De La OJ and Murtaugh LC. Notch and Kras in pancreatic cancer: at the crossroads of mutation, differentiation and signaling. Cell Cycle. 2009; 8: 1860 -1864. [PubMed] .
- 183. Li J, Yen C, Liaw D, Podsypanina K, Bose S, Wang SI, Puc J, Miliaresis C, Rodgers L, McCombie R, Bigner SH, Giovanella BC, Ittmann M, Tycko B, Hibshoosh H, Wigler MH, et al. PTEN, a putative protein tyrosine phosphatase gene mutated in human brain, breast, and prostate cancer. Science. 1997; 275: 1943 -1947. [PubMed] .
- 184. Di Cristofano A, Pesce B, Cordon-Cardo C, Pandolfi PP. Pten is essential for embryonic development and tumour suppression. Nat Genet. 1998; 19: 348 -355. [PubMed] .
- 185. Ma L, Chen Z, Erdjument-Bromage H, Tempst P, Pandolfi PP. Phosphorylation and functional inactivation of TSC2 by Erk implications for tuberous sclerosis and cancer pathogenesis. Cell. 2005; 121: 179 -193. [PubMed] .
- 186. Jones RG and Thompson CB. Tumor suppressors and cell metabolism: a recipe for cancer growth. Genes Dev. 2009; 23: 537 -548. [PubMed] .
- 187. Shackelford DB and Shaw RJ. The LKB1-AMPK pathway: metabolism and growth control in tumour suppression. Nat Rev Cancer. 2009; 9: 563 -575. [PubMed] .
- 188. Vazquez-Martin A, Oliveras-Ferraros C, Lopez-Bonet E, Menendez JA. AMPK: Evidence for an energy-sensing cytokinetic tumor suppressor. Cell Cycle. 2009; 8: 3679 -3683. [PubMed] .
- 189. Peng C, Chen Y, Li D, Li S. Role of Pten in leukemia stem cells. Oncotarget. 2010; 1: 156 -160. [PubMed] .
- 190. Green AS, Chapuis N, Lacombe C, Mayeux P, Bouscary D, Tamburini J. LKB1/AMPK/mTOR signaling pathway in hematological malignancies: from metabolism to cancer cell biology. Cell Cycle. 2011; 10: 2115 -2120. [PubMed] .
- 191. Johannessen CM, Reczek EE, James MF, Brems H, Legius E, Cichowski K. The NF1 tumor suppressor critically regulates TSC2 and mTOR. Proc Natl Acad Sci U S A. 2005; 102: 8573 -8578. [PubMed] .
- 192. Feng Z and Levine AJ. The regulation of energy metabolism and the IGF-1/mTOR pathways by the p53 protein. Trends Cell Biol. 2010; 20: 427 -434. [PubMed] .
- 193. Feng Z, Hu W, Rajagopal G, Levine AJ. The tumor suppressor p53: cancer and aging. Cell Cycle. 2008; 7: 842 -847. [PubMed] .
- 194. Feng Z, Hu W, de Stanchina E, Teresky AK, Jin S, Lowe S, Levine AJ. The regulation of AMPK beta1, TSC2, and PTEN expression by p53: stress, cell and tissue specificity, and the role of these gene products in modulating the IGF-1-AKT-mTOR pathways. Cancer Res. 2007; 67: 3043 -3053. [PubMed] .
- 195. Feng Z, Zhang H, Levine AJ, Jin S. The coordinate regulation of the p53 and mTOR pathways in cells. Proc Natl Acad Sci U S A. 2005; 102: 8204 -8209. [PubMed] .
- 196. Levine AJ, Feng Z, Mak TW, You H, Jin S. Coordination and communication between the p53 and IGF-1-AKT-TOR signal transduction pathways. Genes Dev. 2006; 20: 267 -275. [PubMed] .
- 197. Budanov AV and Karin M. p53 target genes sestrin1 and sestrin2 connect genotoxic stress and mTOR signaling. Cell. 2008; 134: 451 -460. [PubMed] .
- 198. Demidenko ZN, Korotchkina LG, Gudkov AV, Blagosklonny MV. Paradoxical suppression of cellular senescence by p53. Proc Natl Acad Sci U S A. 2010; 107: 9660 -9664. [PubMed] .
- 199. Korotchkina LG, Leontieva OV, Bukreeva EI, Demidenko ZN, Gudkov AV, Blagosklonny MV. The choice between p53-induced senescence and quiescence is determined in part by the mTOR pathway. Aging. 2010; 2: 344 -352. [PubMed] .
- 200. Leontieva O, Gudkov A, Blagosklonny M. Weak p53 permits senescence during cell cycle arrest. Cell Cycle. 2010; 9: 4323 -4327. [PubMed] .
- 201. Galluzzi L, Kepp O, Kroemer G. TP53 and MTOR crosstalk to regulate cellular senescence. Aging. 2010; 2: 535 -537. [PubMed] .
- 202. Long JS and Ryan KM. p53 and senescence: a little goes a long way. Cell Cycle. 2010; 9: 4050 -4051. .
- 203. Santoro R and Blandino G. p53: The pivot between cell cycle arrest and senescence. Cell Cycle. 2010; 9: 4262 -4263. [PubMed] .
- 204. Serrano M. Shifting senescence into quiescence by turning up p53. Cell Cycle. 2010; 9: 4256 -4257. [PubMed] .
- 205. DeBerardinis RJ, Lum JJ, Hatzivassiliou G, Thompson CB. The biology of cancer: metabolic reprogramming fuels cell growth and proliferation. Cell Metab. 2008; 7: 11 -20. [PubMed] .
- 206. Demidenko ZN and Blagosklonny MV. Quantifying pharmacologic suppression of cellular senescence: prevention of cellular hypertrophy versus preservation of proliferative potential. Aging. 2009; 1: 1008 -1016. [PubMed] .
- 207. Demidenko ZN and Blagosklonny MV. The purpose of the HIF-1/PHD feedback loop: to limit mTOR-induced HIF-1alpha. Cell Cycle. 2011; 10: 1557 -1562. [PubMed] .
- 208. Narita M, Young AR, Arakawa S, Samarajiwa SA, Nakashima T, Yoshida S, Hong S, Berry LS, Reichelt S, Ferreira M, Tavare S, Inoki K, Shimizu S. Spatial coupling of mTOR and autophagy augments secretory phenotypes. Science. 2011; 332: 966 -970. [PubMed] .
- 209. Medema RH, Herrera RE, Lam F, Weinberg RA. Growth suppression by p16ink4 requires functional retinoblastoma protein. Proc natl Acad Sci USA. 1995; 92: 62289 -66293. .
- 210. de Jonge HJ, Woolthuis CM, de Bont ES, Huls G. Paradoxical down-regulation of p16 mRNA with advancing age in acute myeloid leukemia. Aging (Albany NY). 2009; 1: 949 -953. [PubMed] .
- 211. Schmidt EV, Ravitz MJ, Chen L, Lynch M. Growth controls connect: interactions between c-myc and the tuberous sclerosis complex-mTOR pathway. Cell Cycle. 2009; 8: 1344 -1351. [PubMed] .
- 212. Cannell IG and Bushell M. Regulation of Myc by miR-34c: A mechanism to prevent genomic instability? Cell Cycle. 2010; 9: 2726 -2730. [PubMed] .
- 213. Haferkamp S, Tran SL, Becker TM, Scurr LL, Kefford RF, Rizos H. The relative contributions of the p53 and pRb pathways in oncogene-induced melanocyte senescence. Aging. 2009; 1: 542 -556. [PubMed] .
- 214. Noonan EJ, Place RF, Basak S, Pookot D, Li LC. miR-449a causes Rb-dependent cell cycle arrest and senescence in prostate cancer cells. Oncotarget. 2010; 1: 349 -358. [PubMed] .
- 215. Akakura S, Nochajski P, Gao L, Sotomayor P, Matsui S, Gelman IH. Rb-dependent cellular senescence, multinucleation and susceptibility to oncogenic transformation through PKC scaffolding by SSeCKS/AKAP12. Cell Cycle. 2010; 9: 4656 -4665. [PubMed] .
- 216. Ertel A, Dean JL, Rui H, Liu C, Witkiewicz AK, Knudsen KE, Knudsen ES. RB-pathway disruption in breast cancer: differential association with disease subtypes, disease-specific prognosis and therapeutic response. Cell Cycle. 2010; 9: 4153 -4163. [PubMed] .
- 217. Land H, Parada LF, Weinberg RA. Tumorigenic conversion of primary embryo fibroblasts requires at least two cooperating oncogenes. Nature. 1983; 304: 596 -602. [PubMed] .
- 218. Kauffmann-Zeh A, Rodriguez-Viciana P, Ulrich E, Gilbert C, Coffer P, Downward J, Evan G. Suppression of c-Myc-induced apoptosis by Ras signalling through PI(3)K and PKB. Nature. 1997; 385: 544 -548. [PubMed] .
- 219. Serrano M, Lin AW, McCurrach ME, Beach D, Lowe SW. Oncogenic ras provokes premature cell senescence associated with accumulation of p53 and p16INK4a. Cell. 1997; 88: 593 -602. [PubMed] .
- 220. Hueber AO and Evan GI. Traps to catch unwary oncogenes. Trends Genet. 1998; 14: 364 -367. [PubMed] .
- 221. Hahn WC, Counter CM, Lundberg AS, Beijersbergen RL, Brooks MW, Weinberg RA. Creation of human tumour cells with defined genetic elements. Nature. 1999; 400: 464 -468. [PubMed] .
- 222. Podsypanina K, Politi K, Beverly LJ, Varmus HE. Oncogene cooperation in tumor maintenance and tumor recurrence in mouse mammary tumors induced by Myc and mutant Kras. Proc Natl Acad Sci U S A. 2008; 105: 5242 -5247. [PubMed] .
- 223. Hoffman B and Liebermann DA. Apoptotic signaling by c-MYC. Oncogene. 2008; 27: 6462 -6472. [PubMed] .
- 224. Chawla R, Procknow JA, Tantravahi RV, Khurana JS, Litvin J, Reddy EP. Cooperativity of Cdk4R24C and Ras in melanoma development. Cell Cycle. 2010; 9: 3305 -3314. [PubMed] .
- 225. Debbas M and White E. Wild-type p53 mediates apoptosis by E1A, which is inhibited by E1B. Genes Dev. 1993; 7: 546 -554. [PubMed] .
- 226. Leontieva OV and Blagosklonny MV. DNA damaging agents and p53 do not cause senescence in quiescent cells, while consecutive re-activation of mTOR is associated with conversion to senescence. Aging (Albany NY). 2010; 2: 924 -935. [PubMed] .
- 227. Blagosklonny MV. Cell cycle arrest is not senescence. Aging. 2011; 3: 94 -101. [PubMed] .
- 228. Wang C, Maddick M, Miwa S, Jurk D, Czapiewski R, Saretzki G, Langie SA, Godschalk RW, Cameron K, von Zglinicki T. Adult-onset, short-term dietary restriction reduces cell senescence in mice. Aging. 2010; 2: 555 -566. [PubMed] .
- 229. Kirkland JL. Perspectives on cellular senescence and short term dietary restriction in adults. Aging. 2010; 2: 542 -544. [PubMed] .
- 230. Demidenko ZN and Blagosklonny MV. Growth stimulation leads to cellular senescence when the cell cycle is blocked. Cell Cycle. 2008; 7: 3355 -3361. [PubMed] .
- 231. Pospelova TV, Demidenk ZN, Bukreeva EI, Pospelov VA, Gudkov AV, Blagosklonny MV. Pseudo-DNA damage response in senescent cells. Cell Cycle. 2009; 8: 4112 -4118. [PubMed] .
- 232. Blagosklonny MV. Aging-suppressants: cellular senescence (hyperactivation) and its pharmacologic deceleration. Cell Cycle. 2009; 8: 1883 -1887. [PubMed] .
- 233. Demidenko ZN, Zubova SG, Bukreeva EI, Pospelov VA, Pospelova TV, Blagosklonny MV. Rapamycin decelerates cellular senescence. Cell Cycle. 2009; 8: 1888 -1895. [PubMed] .
- 234. Demidenko ZN, Shtutman M, Blagosklonny MV. Pharmacologic inhibition of MEK and PI-3K converges on the mTOR/S6 pathway to decelerate cellular senescence. Cell Cycle. 2009; 8: 1896 -1900. [PubMed] .
- 235. Demidenko ZN and Blagosklonny MV. At concentrations that inhibit mTOR, resveratrol suppresses cellular senescence. Cell Cycle. 2009; 8: 1901 -1904. [PubMed] .
- 236. Romanov VS, Abramova MV, Svetlikova SB, Bykova TV, Zubova SG, Aksenov ND, Fornace AJ Jr., Pospelova TV, Pospelov VA. p21(Waf1) is required for cellular senescence but not for cell cycle arrest induced by the HDAC inhibitor sodium butyrate. Cell Cycle. 2010; 9: 3945 -3955. [PubMed] .
- 237. Wesierska-Gadek J. mTOR and its link to the picture of Dorian Gray - re-activation of mTOR promotes aging. Aging. 2010; 2: 892 -893. [PubMed] .
- 238. Bartkova J, Rezaei N, Liontos M, Karakaidos P, Kletsas D, Issaeva N, Vassiliou LV, Kolettas E, Niforou K, Zoumpourlis VC, Takaoka M, Nakagawa H, Tort F, Fugger K, Johansson F, Sehested M, et al. Oncogene-induced senescence is part of the tumorigenesis barrier imposed by DNA damage checkpoints. Nature. 2006; 444: 633 -637. [PubMed] .
- 239. Di Micco R, Fumagalli M, Cicalese A, Piccinin S, Gasparini P, Luise C, Schurra C, Garre M, Nuciforo PG, Bensimon A, Maestro R, Pelicci PG, d'Adda di Fagagna F. Oncogene-induced senescence is a DNA damage response triggered by DNA hyper-replication. Nature. 2006; 444: 638 -642. [PubMed] .
- 240. Mallette FA, Gaumont-Leclerc MF, Ferbeyre G. The DNA damage signaling pathway is a critical mediator of oncogene-induced senescence. Genes Dev. 2007; 21: 43 -48. [PubMed] .
- 241. Bartek J, Lukas J, Bartkova J. DNA damage response as an anti-cancer barrier: damage threshold and the concept of ‘conditional haploinsufficiency’. Cell Cycle. 2007; 6: 2344 -2347. [PubMed] .
- 242. Halazonetis TD, Gorgoulis VG, Bartek J. An oncogene-induced DNA damage model for cancer development. Science. 2008; 319: 1352 -1355. [PubMed] .
- 243. Alimonti A, Nardella C, Chen Z, Clohessy JG, Carracedo A, Trotman LC, Cheng K, Varmeh S, Kozma SC, Thomas G, Rosivatz E, Woscholski R, Cognetti F, Scher HI, Pandolfi PP. A novel type of cellular senescence that can be enhanced in mouse models and human tumor xenografts to suppress prostate tumorigenesis. J Clin Invest. 2010; 120: 681 -693. [PubMed] .
- 244. Courtois-Cox S, Jones SL, Cichowski K. Many roads lead to oncogene-induced senescence. Oncogene. 2008; 27: 2801 -2809. [PubMed] .
- 245. Reddy JP, Peddibhotla S, Bu W, Zhao J, Haricharan S, Du YC, Podsypanina K, Rosen JM, Donehower LA, Li Y. Defining the ATM-mediated barrier to tumorigenesis in somatic mammary cells following ErbB2 activation. Proc Natl Acad Sci U S A. 2010; 107: 3728 -3733. [PubMed] .
- 246. Pinkston JM, Garigan D, Hansen M, Kenyon C. Mutations that increase the life span of C. elegans inhibit tumor growth. Science. 2006; 313: 971 -975. [PubMed] .
- 247. Longo VD, Lieber MR, Vijg J. Turning anti-ageing genes against cancer. Nat Rev Mol Cell Biol. 2008; 9: 903 -910. [PubMed] .
- 248. Toledo LI, Murga M, Gutierrez-Martinez P, Soria R, Fernandez-Capetillo O. ATR signaling can drive cells into senescence in the absence of DNA breaks. Genes Dev. 2008; 22: 297 -302. [PubMed] .
- 249. Efeyan A, Murga M, Martinez-Pastor B, Ortega-Molina A, Soria R, Collado M, Fernandez-Capetillo O, Serrano M. Limited role of murine ATM in oncogene-induced senescence and p53-dependent tumor suppression. PLoS One. 2009; 4: e5475 [PubMed] .
- 250. McManus KJ and Hendzel MJ. ATM-dependent DNA damage-independent mitotic phosphorylation of H2AX in normally growing mammalian cells. Mol Biol Cell. 2005; 16: 5013 -5025. [PubMed] .
- 251. Nelson G, Buhmann M, von Zglinicki T. DNA damage foci in mitosis are devoid of 53BP1. Cell Cycle. 2009; 8: 3379 -3383. [PubMed] .
- 252. Soutoglou E and Misteli T. Activation of the cellular DNA damage response in the absence of DNA lesions. Science. 2008; 320: 1507 -1510. [PubMed] .
- 253. Soutoglou E. DNA lesions and DNA damage response: even long lasting relationships need a “break”. Cell Cycle. 2008; 7: 3653 -3658. [PubMed] .
- 254. Bencokova Z, Kaufmann MR, Pires IM, Lecane PS, Giaccia AJ, Hammond EM. ATM activation and signaling under hypoxic conditions. Mol Cell Biol. 2009; 29: 526 -537. [PubMed] .
- 255. Pankotai T, Hoffbeck AS, Boumendil C, Soutoglou E. DNA damage response in the absence of DNA lesions continued. Cell Cycle. 2009; 8: 4025 -4026. [PubMed] .
- 256. Bouquet F, Ousset M, Biard D, Fallone F, Dauvillier S, Frit P, Salles B, Muller C. A DNA-dependent stress response involving DNA-PK occurs in hypoxic cells and contributes to cellular adaptation to hypoxia. J Cell Sci. 2011; 124: 1943 -1951. [PubMed] .
- 257. Alexander A and Walker CL. Differential localization of ATM is correlated with activation of distinct downstream signaling pathways. Cell Cycle. 2010; 9: 3685 -3686. [PubMed] .
- 258. Rodriguez-Jimenez FJ, Moreno-Manzano V, Mateos-Gregorio P, Royo I, Erceg S, Murguia JR, Sanchez-Puelles JM. FM19G11: A new modulator of HIF that links mTOR activation with the DNA damage checkpoint pathways. Cell Cycle. 2010; 9: 2803 -2813. [PubMed] .
- 259. Guo Z, Deshpande R, Paull TT. ATM activation in the presence of oxidative stress. Cell Cycle. 2010; 9: 4805 -4811. [PubMed] .
- 260. Cam H, Easton JB, High A, Houghton PJ. mTORC1 signaling under hypoxic conditions is controlled by ATM-dependent phosphorylation of HIF-1alpha. Mol Cell. 2010; 40: 509 -520. [PubMed] .
- 261. Shor B, Gibbons JJ, Abraham RT, Yu K. Targeting mTOR globally in cancer: thinking beyond rapamycin. Cell Cycle. 2009; 8: 3831 -3837. [PubMed] .
- 262. Benjamin D, Colombi M, Moroni C, Hall MN. Rapamycin passes the torch: a new generation of mTOR inhibitors. Nat Rev Drug Discov. 2011; 10: 868 -880. [PubMed] .
- 263. Morris JZ, Tissenbaum HA, Ruvkun G. A phosphatidylinositol-3-OH kinase family member regulating longevity and diapause in Caenorhabditis elegans. Nature. 1996; 382: 536 -539. [PubMed] .
- 264. Babar P, Adamson C, Walker GA, Walker DW, Lithgow GJ. P13-kinase inhibition induces dauer formation, thermotolerance and longevity in C. elegans. Neurobiol Aging. 1999; 20: 513 -519. [PubMed] .
- 265. Vellai T, Takacs-Vellai K, Zhang Y, Kovacs AL, Orosz L, Muller F. Genetics: influence of TOR kinase on lifespan in C. elegans. Nature. 2003; 426: 620 [PubMed] .
- 266. Hansen M, Taubert S, Crawford D, Libina N, Lee SJ, Kenyon C. Lifespan extension by conditions that inhibit translation in Caenorhabditis elegans. Aging Cell. 2007; 6: 95 -110. [PubMed] .
- 267. Kapahi P, Zid BM, Harper T, Koslover D, Sapin V, Benzer S. Regulation of lifespan in Drosophila by modulation of genes in the TOR signaling pathway. Curr Biol. 2004; 14: 885 -890. [PubMed] .
- 268. Edman U, Garcia AM, Busuttil RA, Sorensen D, Lundell M, Kapahi P, Vijg J. Lifespan extension by dietary restriction is not linked to protection against somatic DNA damage in Drosophila melanogaster. Aging Cell. 2009; 8: 331 -338. [PubMed] .
- 269. Katewa SD and Kapahi P. Role of TOR signaling in aging and related biological processes in Drosophila melanogaster. Exp Gerontol. 2011; 46: 382 -390. [PubMed] .



