Introduction
Aging is associated with a progressive deterioration in the function of cells, tissues, and organs. This decline in function eventually leads to age-related declines in physiologic function, such as loss of muscle mass, declining blood flow, impaired immune recognition, decreased ability to utilize energy, diminished cognitive function, and reduced ability to respond to stress stimuli. These age-related declines culminate in the onset of age-associated diseases like sarcopenia, cardiovascular disease, cancer, obesity, diabetes, neurodegenerative diseases, and many other pathologies. Recently, Lopez-Otín et al [1] cataloged the molecular processes that decline with advancing age and underlie the phenotypes of aging. They include epigenetic changes, loss of telomere function, declining protein homeostasis, increased cellular senescence, depletion of the stem cell pool, and altered intercellular communication.
The phenotypic changes that characterize the aging process are governed by specific alterations in the pools of expressed proteins. Therefore, there is heightened interest in understanding the mechanisms that drive age-associated gene expression programs. These processes are mainly directed by proteins that bind DNA and RNA, as well as by a variety of noncoding (nc) RNA, both short ncRNAs (mainly microRNAs) and long (lncRNAs). Together, this vast and heterogeneous group of factors affects aging by controlling gene expression transcriptionally and post-transcriptionally in myriad different ways. The influence of transcription factors, RNA-binding proteins, and microRNAs in age-associated processes has been reviewed elsewhere [2-4]. Some lncRNAs have been studied for decades, but the broad range of expressed lncRNAs and their impact on protein expression programs has only come into view in recent years. LncRNAs modulate gene expression patterns at all levels: transcriptional, post-transcriptional, and post-translational [5-7]. Through their impact on the type and abundance of proteins, lncRNAs affect key cellular processes such as proliferation, differentiation, quiescence, senescence, the cellular response to stress and immune agents, and many others cellular functions relevant to the biology of aging. In this review, we describe and discuss the emerging impact of lncRNAs on these processes and their possible implications in senescence, aging and age-related pathologies.
LncRNAs. LncRNAs vary widely in size, genomic localization, and biogenesis. They can be expressed from intergenic regions (lincRNAs), from the opposite strand of mRNAs (antisense lncRNAs), from vestigial genes that lost their coding potential (pseudogene-encoded lncRNAs), from introns of annotated genes (long intronic ncRNAs), or from the promoter regions of coding mRNAs (promoter-associated lncRNAs); they can also be generated by the splicing machinery (circular RNAs) [8].
LncRNAs can be classified according to their molecular mechanism of action. Some nuclear lncRNAs can regulate gene expression epigenetically by recruiting chromatin-modification factors to activate or inactivate different loci [9, 10]. LncRNAs can also serve as transcriptional regulators by assembling transcriptional activators and repressors to modulate transcription initiation by RNA polymerase II [10, 11]. Other lncRNAs can function in nuclear compartmentalization and help maintain nuclear structures such as nuclear speckles, paraspeckles, and interchromatin granules [11]. In addition, lncRNAs can regulate gene expression post-transcriptionally by base-pairing with mRNAs to modulate their translation and/or stability [12-14] and by interfering with RNA-binding proteins to influence splicing and translation [14-16]. Competing endogenous RNAs (ceRNAs) and circular RNAs are stable lncRNAs that accumulate in large numbers and modulate gene expression in different ways, including as decoys or sponges for microRNAs [17, 18]. Finally, some lncRNAs have been shown to control protein turnover by facilitating ubiquitination [7] (Figure 1, Table 1).
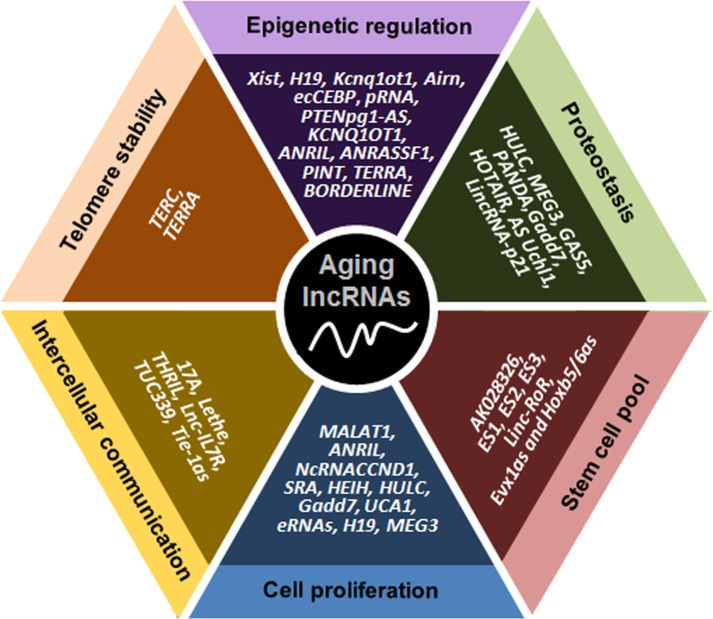
Figure 1. Schematic representation of lncRNAs affecting six major molecular traits of aging The lncRNAs indicated have been implicated in controlling telomere length, epigenetic gene expression, proteostasis, the pool of stem cells, cell proliferation and senescence, and communication among cells.
Table 1. LncRNAs affecting molecular traits of aging
| LncRNAs | Function in Aging Traits | Comments | Refs. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| LncRNAs controlling telomere function | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TERC | Promotion of telomere extension | Terc KO mice age prematurely | [20] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TERRA | Suppression of telomere extensionHeterochromatin | High TERRA triggers senescenceModulates telomeric structure | [23, 24][89] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LncRNAs controlling epigenetic changes | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Xist | DNA methylation | Declines with senescence | [34] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| H19 | DNA methylation Cell division cycle | Increases in some cancers Enhances proliferation | [39] [37-40] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Kcnq1ot1 | DNA methylation, cell division | Implicated in cancer and diabetes | [43] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Airn | DNA methylation | Controls IGF signaling pathway | [45] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ecCEBP | DNA methylation | Affects senescence TFs C/EBP | [49,50] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| pRNA | DNA methylation | Linked to reduced rRNA in AD | [55,56] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PAPAS | Histone methylation | Chromatin compaction at rDNA loci | [58] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PTENpg1-AS | DNA methylation | Modulates PTEN expression | [59] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TARID | DNA methylation | Triggers expression of TCF21 | [65] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| KCNQ1OT1 | Histone modifications | Blocks Kcnq1 (linked to age, CVD) | [73,74] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| NeST | DNA methylation | Increases IFN-γ upon infection | [76] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ANRIL | Histone modifications | Controls p16 levels (senescence, aging) | [77] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ANRASSF1 | Histone modifications | Recruits PRC2 to RASSF1A promoter | [79] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PINT | Histone modifications | Recruits PRC2, elicits p53 actions | [80-82] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BORDERLINE | Heterochromatin | Processed into brdrRNAs by Dicer | [90] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LncRNAs affecting proteostasis | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| HULC | Autophagy | Inhibits apoptosis, promotes autophagy | [97] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| MEG3 | Autophagy Growth arrest | Suppresses MDM2, upregulates p53 Blocks apoptosis | [99] [100] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 7SL | Autophagy Protein trafficking | Suppresses p53 Competes with HuR, linked to senescence | [14] [14] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GAS5 | Protein trafficking | Binds GR, linked to cognitive decline | [102] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PANDA | Protein trafficking | Inducible by p53 upon DNA damage | [104,105] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Gadd7 | Protein trafficking | Binds TDP-43, modulates Cdk6 levels | [107] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| HOTAIR | Protein abundance | Up in senescence, ubiquitination | [7] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| AS Uchl1 | Protein abundance | Induces senescence, upregulated in PD | [110,111] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LincRNA-p21 | Protein abundance | Represses translation of cancer proteins | [13] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LncRNAs modulating stem cell function | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| AK028326, AK141205 | Regulation of stem cell TFs | Control function of mouse ESC TF | [123] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ES1, ES2, ES3 | Regulation of stem cell TFs | Regulated by human ESC, iPSC factors | [129] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| linc-RoR | Regulation of stem cell TFs | Regulates hESCs reprograming | [130] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Evx1as, Hoxb5/6as | Histone methylation in stem cells | Associated with ES cell pluripotency | [133] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LncRNAs controlling proliferation and senescence | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| MALAT1 | Cell division cycle | Inhibits senescence, promotes division | [141] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ANRIL | Cell division cycle | Prevents expression of p15 and p16 | [144] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| NcRNACCND1 | Cell division cycle | Inhibits transcription of CCND1 gene | [119] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SRA | Cell division cycle | Inhibits senescence cdki p21, p27 | [146,147] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| HEIH | Cell division cycle | Inhibits senescence cdki p21, p16, p27 | [149] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| HULC | Cell division cycle | Inhibits senescence cdki p18 | [152] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Gadd7 | Cell division cycle | Binds TDP-43, lowers Cdk6 mRNA | [107] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| UCA1 | Cell division cycle | Inhibits senescence cdki p27 | [156] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| eRNAs | Cell division cycle | p53-regulated, affect senescence, aging | [161] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 7SL | Cell division cycle | Lowers p53 levels, inhibits senescence | [14] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LncRNAs governing intercellular communication | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 17A | Modulates inflammation | Upregulated in AD, linked to GPRs | [176] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Lethe | Modulates inflammation | Induced by TNFα, inhibits NF-κB | [178] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| THRIL | Modulates inflammation | Induced by TNFα, interacts with hnRNPL | [120] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Lnc-IL7R | Modulates inflammation | Regulates LPS-mediated inflammation | [180] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TUC339 | Transported in exosomes | Controls cell proliferation, tumor growth | [192] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Tie-1as | Transported in exosomes | Targets tie-1, affects endothelial junctions | [194] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Linc-RoR | Transported in exosomes | Contributes to chemoresistence in hepatocellular carcinoma cells | [195] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| The table lists potential age-associated lncRNAs (column 1), the cellular and molecular processes they regulate (column 2), and specifics of their expression and/or function (column 3). AD, Alzheimer's disease; cdki, cyclin-dependent kinase inhibitor; CVD, cardiovascular disease; GR, glucocorticoid receptor; PD, Parkinson's disease; TF, transcription factor. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
LncRNAs that modulate telomere length
Telomeres are structures that protect the ends of chromosomes against damage. Cellular senescence is generally associated with a gradual shortening of telomere length [19]. In order for DNA replication to take place on the telomeres, a specific polymerase, telomerase reverse transcriptase (TERT), is required to extend telomeres to preserve their lengths [1]. The length of telomeres is regulated by the telomerase ribonucleoprotein complex that contains the protein TERT and the lncRNA TERC (telomerase RNA component), as well as by the telomeric repeat-containing RNA lncRNA TERRA.
TERC. The essential RNA component of the telomerase enzyme complex, lncRNA TERC, has been directly implicated in the maintenance of telomere length and thus the prevention of premature senescence and aging. In support of this function, TERC-deficient mice displayed short telomeres, chromosomal instability, and premature aging [20]. TERC serves as a template for the synthesis of telomeric repeats and acts as a scaffold that brings the protein subunits of telomerase together with other accessory proteins associated with the complex. Additionally, TERC was shown to have a catalytic function in the process of adding telomere repeats [21].
TERRA. While TERC promotes and maintains telomere length, the lncRNA TERRA suppresses telomere elongation. TERRA transcripts are transcribed from telomeres by RNA polymerase II, an unexpected discovery, since telomeres were long believed to be transcriptionally silent, and have variable lengths ranging between 100 and >9000 nt in mammals [22]. The suppression of telomeric RNA elongation is linked to the presence of numerous copies of the telomere UUAGGG repeat in the TERRA transcript, spanning ~200 nt [23], which renders TERRA a high-affinity ligand (and hence a competitive inhibitor) for TERT [24].
Abnormal expression of TERRA may contribute to premature senescence and aging. For instance, mutation in the gene DNA methyltransferase 3B (DNMT3B) leads to hypomethylation of the telomeric region and elevated levels of TERRA. These changes result in the syndrome ICF (immunodeficiency, centromeric instability, and facial dysmorphism), in which fibroblasts exhibit premature senescence linked to the suppression of telomere elongation [25]. TERRA is also involved in the removal of 3′G overhangs of uncapped telomeres during DNA damage-induced senescence [23]. Although the loss of telomeres is believed to accelerate senescence, telomerase-deficient cells can avoid senescence by a unique mechanism that requires TERRA during telomere recombination [26]. Early in S phase, the levels of TERRA decline, rising again at the end of S phase and thereby displacing hnRNPA1 from the telomeres and recruiting the single-stranded DNA-binding protein POT1 (protection of telomeres 1). Thus, while TERRA suppresses telomere elongation, it can also protect telomere ends [27].
LncRNAs associated with epigenetic alterations in aging and senescence
Epigenetic changes modulate gene expression in a variety of processes that characterize age-associated pathology and physiology [28, 29]. In this section, we review the involvement of lncRNAs in epigenetic changes including DNA methylation, histone modification, and heterochromatin formation.
DNA methylation
A global decline in DNA methylation during aging and cellular senescence is well documented [30]. However, advancing age also leads to hypermethylation of several genes such as tumor suppressors and genes targeted by Polycomb group proteins in embryonic stem cells [31]. Several lncRNAs that contribute to the regulation of DNA methylation in the context of senescence and aging have been identified.
Xist. Responsible for imprinting and hence silencing of the X chromosome in females (to compensate for the dosage effect in males), Xist levels decline in senescent cells [32, 33, 34]. However, its specific function in senescence has not been described yet.
H19. The lncRNA H19 has been shown to play a role in embryonic development and growth [35]. It controls imprinting of a conserved cluster of genes that contains H19 itself and the insulin-like growth factor 2 (IGF2). This regulatory effect is mediated by the interaction of H19 with the methyl-CpG–binding domain protein 1 (MBD1) to form a ribonucleoprotein complex (H19-MBD1) that interacts with and recruits histone lysine methyltransferases and represses gene expression [36]. Both IGF2 and H19 are implicated in growth, proliferation, cell cycle, apoptosis, and aging [37-40]. Loss of imprinting of the IGF2-H19 locus during aging was observed in normal human prostate tissues leading to enhanced expression of IGF2, H19, and other genes in this locus. H19 levels were further elevated in old prostate tissues [41]. Loss of imprinting increases the levels of IGF2, which is associated with age-related diseases including cancer [42].
Kcnq1ot1. During mouse embryonic development, expression of the lncRNA Kcnq1ot1 leads to transcriptional gene silencing by the recruitment of DNA methyltransferases (DNMTs) to the paternal allele of the Kcnq locus. The promoter of Kcnq1ot1 is methylated at the maternal allele; the ensuing suppression of Kcnq1ot1 expression allows gene expression of genes on the Kcnq locus relevant to age-associated diseases like type 2 diabetes and cancer [43]. One of the genes within this locus is the cyclin-dependent kinase (cdk) inhibitor and tumor suppressor CDKN1C, encoding P57KIP2 [44]. Thus, Kcnq1ot1 can affect aging and senescence through its impact upon the cell cycle.
Airn. The lncRNA Airn is transcribed in antisense orientation to the maternally expressed Igf2r gene and controls Igf2r mRNA expression by silencing its transcription in cis. Interestingly, it is not the Airn lncRNA per se, but rather Airn transcription what drives methylation changes and Igf2r silencing [45]. Airn is likely involved in aging and senescence through its effect on IGF2R, since senescent cells show enhanced IGF2R expression compared to proliferating cells and IGF2R is implicated in longevity [46, 47]; however, a role for Airn in these age-related processes has not been studied directly.
ecCEBP. The lncRNA ecCEBP was also found to recruit DNMT1 in order to regulate local DNA methylation and silence the CEBP gene [48]. The encoded protein, CAAT enhancer-binding protein (C/EBP), influences cell cycle progression by interacting with and inhibiting kinases cdk2 and cdk4, triggering growth arrest [49]. The levels of several C/EBP family members, which are key regulators of adipogenesis, decline with age [50]. Recently, age-associated changes in C/EBP proteins were shown to cause severe liver injury and accelerated liver proliferation after treatment with CCl4 (carbon tetrachloride) [51], while C/EBPγ suppressed senescence and inflammatory gene expression by heterodimerizing with C/EBPβ [52]. Together, these findings indicate that the lncRNA ecCEBP regulates C/EBP expression and highlight its putative role in the age- and senescence-dependent changes in C/EBP abundance.
pRNA. The lncRNA pRNA regulates transcription of ribosomal RNA (rRNA) by interacting with DNA at the target site of the transcription factor TTF1; the resulting DNA-RNA triplex is specifically recognized by the DNA methyltransferase DNMT3b [53]. Accelerated rDNA methylation was observed in senescent Werner syndrome fibroblasts [54], although the possible involvement of pRNA in this process has not been examined. rRNA levels are selectively reduced with brain aging and this reduced ribosomal activity may contribute to Alzheimer disease (AD) [55, 56], although pRNA has not yet been implicated in the age-related changes in rRNA.
PAPAS. The heterogeneous population of lncRNAs termed PAPAS (promoter and pre-rRNA antisense) is derived from rDNA gene transcription in the antisense orientation [57]. During quiescence, PAPAS is upregulated to guide the H4K20 methyltrasnferase Suv4-20h2 to nucleolar chromatin, leading to increased trimethylation of H4K20 and chromatin compaction at rDNA loci [58].
PTENpg1-AS. The lncRNA PTENpg1-AS, which represses expression of the tumor-suppressor gene phosphatase and tensin homolog (PTEN), exists in two isoforms, α and β. PTENpg1-ASα represses PTEN expression by lowering its transcription via its interaction with DNMT3a and enhancer of Zeste on the PTEN promoter, while PTENpg1-ASβ regulates PTEN mRNA stability [59]. Given the role of PTEN in aging, senescence, and age-related diseases such as cancer, we anticipate a direct function for PTENpg1-AS in these processes [60-64].
TARID. The lncRNA TARID (TCF21 antisense RNA inducing demethylation) has the ability to trigger expression of the tumor suppressor protein TCF21 (transcritpion factor 21) by inducing promoter demethylation. During this process, TARID binds to GADD45A (Growth arrest and DNA-damage inducible alpha), a regulator of DNA demethylation, and recruits it to the TCF21 promoter. Interestingly, TARID is expressed in the antisense orientation to the TCF21 gene [65].
Histone modifications
The most common epigenetic alterations during aging and senescence include enhanced histone H4K16 acetylation, H4K20 trimethylation and H3K4 trimethylation, and decreased histone H3K9 methylation and H3K27 trimethylation [66, 67, 68, 69]. Importantly, disruption of histone modification influences longevity in nematodes, flies, and worms [70-72]. In this section, we examine lncRNAs implicated in histone modifications, particularly methylation, that play a role in processes such as cell cycle progression, inflammation, and senescence.
Kcnq1ot1. The levels of the potassium channel protein KvLQT1 (encoded by the gene KCNQ1) decline with aging and age-related cardiovascular disease [73, 74]. The 91-kb long lncRNA Kcnq1ot1 influences histone modifications by recruiting the histone methyl-transferases G9a and polycomb repressor complex (PRC2) to increase H3K9 and H3K27 trimethylation and repress transcription of the Kcnq1 locus [75].
NeST. The lncRNA NeST (nettoie Salmonella pas Theiler's) binds to the H3K4 methyltransferase component WDR5 (WD repeat domain 5) and recruits it to the IFNγ locus. Given that lncRNA NeST is involved in inflammation during microbial infection [76] and that IFNγ methylation increases with advancing age, NeST could contribute to the inflammatory response and infection in elderly.
ANRIL. The lncRNA ANRIL is transcribed from the same locus as INK4b/ARF/INK4a but in the opposite direction. ANRIL is involved in cell cycle regulation at least in part by recruiting CBX7 (Chromobox 7), a protein component of the of polycomb repressor complex 1 (PRC1), to the locus, increasing H3K27 methylation and thereby repressing INK4a transcription. Interestingly, CBX7 variants with point mutations that disrupt binding to RNA or methylated H3K27 repress genes in the INK4a locus, impairing cellular senescence [77].
ANRASSF1. The promoter of gene RASSF1A, which encodes the Ras association domain-containing protein 1, was found to be increasingly hypermethylated with advancing age [78]. The lncRNA ANRASSF1 forms an RNA/DNA hybrid at the RASSF1A transcriptional start site and recruits the chromatin-modifying PRC2 complex to the RASSF1A promoter. The PCR2 complex then selectively modifies histone H3K27 methylation, reducing RASSF1A transcription [79].
PINT. The p53-induced lncRNA PINT also interacts with PRC2 to regulate the expression of proteins in the TGF-β, MAPK and p53 pathways, which are associated with senescence, aging, and age-related diseases [2, 80-82].
Heterochromatin formation
Global disruption of heterochromatin is a hallmark of senescence and aging. In senescent cells, dramatic remodeling of the chromatin is associated with the formation of senescence-associated heterochromatin foci (SAHF) [83, 84]. The loss of heterochromatin with aging is believed to underlie various cellular processes associated with aging [85, 86]. Heterochromatin formation was found to promote longevity in Drosophila and the function of heterochromatin binding protein 1 (HP1a) correlates with life span in flies [87]. In addition, chromosomal stability is directly related to hetero-chromatin formation and maintenance. Epigenetic features including H3K9 trimethylation, H4K20 trimethylation, and HP1a binding are observed at constitutive heterochromatin in chromosomal DNA repeat regions such as subtelomeric and pericentromeric regions [88].
TERRA. Telomeric heterochromatin is modulated by TERRA through binding to several telomeric proteins: telomere repeat factors (TRF1, TRF2), origin recognition complex (ORC), HP1, and H3K9me3. Through these interactions TERRA helps to maintain telomeric structure and heterochromatin [89].
BORDERLINE. This lncRNA was found to prevent spreading of the HP1 protein SWI6 and H3K9 methylation beyond the pericentromeric region in yeast. Interestingly, BORDERLINE is processed into short RNAs (brdrRNAs) by Dicer, a type-III RNase implicated in aging and senescence [90-92]. Accordingly, Dicer might modulate BORDERLINE levels, function, and processing to brdrRNAs in age-relevant processes.
lncRNAs and proteostasis
Proteostasis (protein homeostasis) encompasses several biological processes that govern protein biogenesis, folding, trafficking, activity, interactions, degradation, and elimination. Disruption of proteostasis can lead to age-related diseases such as Alzheimer's, Parkinson's, and Huntington's diseases [93, 94]. Major proteolytic systems, including autophagy and the ubiquitin-proteasome pathway, decline with age, indicating that proteostasis is a common feature of aging [1]. In this section, we review the lncRNAs associated with proteostasis, including autophagy, and protein synthesis, trafficking, assembly, and degradation.
LncRNAs in autophagy
Changes in autophagy are another hallmark of aging [1]. Disruption of autophagy induces changes in mammalian tissue that resemble those associated with aging. Inhibition of autophagy also compromises the aging-inhibitory effects of interventions such as caloric restriction and sirtuin 1 activation [95]. Although their interdependence is not clearly understood, autophagy and senescence are integral processes of the cellular response to stress agents [96].
HULC, MEG3, and 7SL. These lncRNAs modulate autophagy in different ways. Overexpression of HULC in SGC7901 human gastric cancer cells promoted cell proliferation and invasion, and inhibited apoptosis by inducing autophagy [97]. In contrast, the levels of MEG3 and 7SL correlated inversely with the level of the autophagy marker LC3-II in vivo [14, 98, 99]. The underlying mechanisms through which these lncRNAs influence autophagy are not well understood, but they may involve p53, since 7SL can regulate p53 mRNA translation and MEG3 can regulate p53 directly via RNA-protein association or indirectly by lowering MDM2 expression levels [100, 101]. In sum, the aging- and senescence-associated process of autophagy is robustly affected by lncRNAs.
LncRNAs in protein trafficking
Some lncRNAs influence homeostasis by modulating the traffic of proteins and hence the abundance of a protein in a particular subcellular compartment. By recruiting transcription factors to the nucleus and specifically to certain DNA regions, lncRNAs can modulate transcription. Similarly, by recruiting certain RNA-binding proteins into ribonucleoprotein complexes, lncRNAs can elicit other gene regulatory functions. Although the complete spectrum of mechanisms through which lncRNAs affect protein trafficking and their impact on aging and senescence are largely unknown, a few examples are beginning to emerge.
GAS5. This lncRNA induces growth arrest by acting as a decoy for the transcription factor glucocorticoid receptor (GR) and thus inhibits GR-mediated gene expression [102]. Specifically, GAS5 binds GR in the cytoplasm and prevents its mobilization to the nucleus. Interestingly, the level of GR protein decreased in the nucleus but not the cytosol of aged Long-Evans rats with cognitive impairment [103], although it is not yet known if GAS5 is directly involved in the aberrant GR distribution in this animal model of aging.
PANDA. The lncRNA PANDA is induced by p53 upon DNA damage. It binds the transcription factor NF-YA and interferes with its transcriptional activity, lowering the expression of apoptotic genes. Interestingly, NF-YA interaction with p53 impacts upon cell cycle regulation and senescence [104, 105]. These findings indicate that PANDA could be involved in DNA damage-induced senescence through NF-YA and p53.
Gadd7. In Chinese hamster ovary (CHO) cells, Gadd7 was induced by DNA damage and oxidative stress and controlled the G1/S checkpoint and cell growth [106]. Gadd7 associated directly with TDP-43 and interfered with the binding of TAR DNA-binding protein (TDP-43) to Cdk6 mRNA, leading to Cdk6 mRNA destabilization, abnormal cell cycle progression, and possibly senescence [107, 108]. It is not known if Gadd7 interferes with other TDP-43 targets or if it influences other RNA-binding proteins involved in senescence or aging.
7SL. This highly abundant lncRNA interacts with the TP53 mRNA and suppresses p53 translation. The RNA-binding protein HuR can displace 7SL and enhances p53 translation. According to this competitive interaction, silencing 7SL increased HuR binding to TP53 mRNA and promoted p53 translation, in turn enhancing cell cycle arrest and senescence [14].
lncRNAs associated with protein synthesis and degradation
Protein synthesis and degradation are two critical processes that determine the steady-state abundance of proteins in cells. Protein synthesis is controlled by the rates of mRNA translation, while the ubiquitin proteasome pathway is the major driver for protein degradation. Some lncRNAs modulate protein levels indirectly by influencing the available pool of microRNAs and thereby affecting mRNA turnover and translation; for instance, lncRNAs such as linc-MD1 and lincRNA-RoR (below) can act as decoys for miRNAs that usually suppress the translation or stability of other mRNAs [17, 109]. However, some lncRNAs interact directly with mRNAs to enhance or suppress their translation or with proteins to modulate their half-lives:
AS Uchl1. AS Uchl1 enhances translation of UCHL1 (ubiquitin carboxy-terminal hydrolase L1) through an embedded SINE (short interspersed nuclear element) B2 repeat present in AS Uchl1 [110]. UCHL1 is involved in brain development and age-related neurodegenerative pathologies such as Parkinson's disease [111]. Overexpression of UCHL1 was found to induce senescence, likely due to increased production of p14ARF, p53, p27KIP1 and decreased MDM2 levels [112]. Accordingly, AS Uchl1 lncRNA may influence both senescence and neurodegeneration.
LncRNA-p21. Unlike AS Uchl1, the human lncRNA-p21 was found to interact with target CTNNB and JUNB mRNAs (encoding β-catenin and JunB, respectively) and suppressed their translation by recruiting translation repressors RCK and FMRP [13]. The exact role of lncRNA-p21 senescence and aging remains to be investigated, although β-catenin and JunB are known to influence cell proliferation and carcinogenesis [113-116].
HOTAIR. The levels of HOTAIR, a lncRNA upregulated in senescence, are reduced by the senescence-repressor HuR, an RNA-binding protein, via degradation of HOTAIR in a microRNA-dependent manner. HOTAIR was shown to serve as a scaffold to promote the ubiquitination and subsequent degradation of Ataxin-1 and Snurportin-1 [7]. These findings suggested that senescence-associated lncRNAs can function as platforms to facilitate protein ubiquitination and degradation to elicit cellular senescence.
LncRNAs and complex assembly
The assembly of protein complexes is widely required for the modulation of gene expression patterns. Several lncRNAs function in complexes with proteins, RNA, and DNA, as explained above. For instance, the lncRNA TERC is essential for the telomere complex formation that maintains telomere length [21, 117, 118], H19 interacts with MBD1 to form a ribonucleoprotein complex that recruits histone lysine methyltransferases to suppress gene expression [36], and ecCEBP forms a complex with DNMT1 to regulate DNA methylation and silencing of the CEBP gene [48]. The lncRNA pRNA interacts with DNA at a site which is specifically recognized by the DNMT3b to regulate rRNA transcription [53], PTENpg1-AS lncRNA interacts with DNMT3a forming a ribonucleoprotein complex to regulate PTEN transcription [59], and lncRNAs Kcnq1ot1, ANRIL, ANRASSF1, and PINT form complexes with histones or histone modifiers to regulate gene expression as explained above. Other examples include lncRNA-protein complexes NcRNACCND1-TLS, which are recruited to the promoter to suppress transcription of the CCND1 gene following DNA damage [119], and THRIL-hnRNPL, which is recruited to the tumor necrosis factor (TNF)α promoter site to regulate TNFα expression [120]. These complexes broadly regulate gene expression programs relevant to the control of cell cycle progression and inflammation, two processes central to senescence and aging.
LncRNAs and Stem cells
Stem cells are progenitor cells for the development of specific cell types in different tissues. The ability of stem cells to regenerate tissues (also known as ‘pluripotency’) declines with age. Like adult cells, stem cells are also exposed to stimuli that promote aging or senescence such as DNA damage and cell cycle inhibition. Disruption of stem cell gene expression programs is deleterious for various tissues and promotes age-related physiologic declines like immunosenescence (diminished production of adaptive immune cells) and reduction in muscle mass. They also underlie many pathologies, including cancer [1]. Pluripotency in human and mouse embryonic stem cells (ESCs) is tightly regulated and involves transcription factors (TFs) such as Oct4, Sox2 and Nanog along with several other coregulators [121]. These TFs transcribe genes that encode proteins involved in the maintenance of pluripotency and suppress the transcription of genes that encode proteins necessary for developing specific cell types [122]. Examples of lncRNAs that affect stem cell homeostasis are emerging, as discussed below.
Cross-regulation between lncRNAs and stem cell transcription factors
LncRNAs can regulate certain stem-cell transcription factors, which reciprocally can regulate the expression of lncRNAs. The abundance of some conserved lncRNAs is transcriptionally regulated by Oct4 and Nanog in mouse ESCs: AK028326 is activated by Oct4, while AK141205 is repressed by Nanog. Interestingly, knockdown or overexpression of these lncRNAs altered Oct4 and Nanog mRNA levels and subsequently cellular lineage-specific gene expression and pluripotency, suggesting a feedback regulatory loop [123]. In mouse ESCs, several lncRNAs involved in stem cell gene regulation were identified that bound chromatin-regulatory proteins and coordinated gene expression programs [124].
ES1, ES2, ES3. The levels of the transcription factor REST are elevated in ESCs and decline as ESCs convert to neuronal SCs [125]. REST is involved in neurodegeneration, neurogenesis, and neuronal pathologies like ischemia, epilepsy, and Huntington's disease [126, 127]. REST has been suggested to regulate the expression of lncRNAs which could be involved in neurodegeneration and cancer [128]. A more detailed study of human lncRNAs that promote pluripotency and neuronal differentiation indicated that lncRNAs ES1, ES2, and ES3 are exclusively expressed in human ESC or iPSC and also regulated by Nanog and/or Oct4 [129]. Interestingly, depletion of these lncRNAs was proposed to inhibit neurogenesis through binding to the epigenetic regulators REST and SUZ12 [129].
linc-RoR. Recently, another report indicated that the expression of 28 lncRNAs was higher in iPSCs than in ESCs, suggesting that they might be required for the development of iPSCs. One of these lncRNAs, linc-RoR, was found to regulate the reprograming of hESCs. Silencing of linc-RoR inhibited the formation of iPSC colonies while its overexpression favored reprograming, prompting the hypothesis that linc-RoR might act as an endogenous miRNA sponge to regulate Oct4, Nanog, and Sox2 in hESCs [130]. As linc-RoR levels decrease rapidly under differentiation conditions, it was proposed to be a self-renewal and pluripotency marker for hESCs [109, 131]. Linc-RoR was also found to lower the levels of the tumor suppressor and cell cycle regulator p53 under DNA damage conditions; similar to UCA1 (below), linc-RoR interacts with p-hnRNP I, which is required for p53 mRNA translation [132]. These findings indicate that linc-RoR is a potential stem cell regulator and modulator of cell growth and survival. The influence of linc-RoR on p53 expression connects linc-RoR to the control of cell cycle progression and cellular senescence.
Histone methylation regulates stem cell lncRNAs
Profiling of mouse ES cells differentiated into embryoid bodies identified 174 differentially expressed lncRNAs, many of which were associated with developmental genes (e.g., Dlx1, Dlx4, Gata6, Ecsit), including the pluripotency-associated lncRNAs Evx1as and Hoxb5/6as. They are derived from homeotic loci and associated with trimethylated H3K4 histones and the histone methyltransferase MLL1 [133]. Further studies suggested that lncRNA transcription in mouse ESCs is regulated by promoter methylation at CpG similar to protein-coding genes. For example, loss of the repressive H3K27me3 upon differentiation transcriptionally activated many lncRNAs, suggesting that ESC-specific lncRNAs are regulated via mechanisms similar to those that control protein-coding gene expression to influence stem cell differentiation and maintain pluripotency [134].
Cell cycle-associated lncRNAs
Environmental stresses, telomere dysfunction, oxidative stress, and DNA damage negatively influence normal cell cycle progression causing cells to withdraw from cell cycle, halt replication and enter senescence. Thus, as tissues and organs age, terminally arrested senescent cells accumulate [135-137]. Accordingly, DNA damage, a suggested primary cause of cellular senescence and organismal aging, increases with advancing age and elevates cell cycle inhibitors (e.g., p53 and p21) that promote senescence [138]. Recent insight into lncRNAs involved in cell cycle regulation and senescence were mentioned earlier in this review. Here, we discuss cell cycle-related lncRNAs that may influence senescence and organismal aging.
MALAT1. Several lines of evidence suggest that MALAT1 represses senescence. For example, depletion of MALAT1 in CaSki human cervical cancer cells induced G1 arrest and reduced cell growth, cell proliferation, and tumor size [139], and depletion of MALAT1 in the breast cancer cell line MB231 decreased cell proliferation and triggered G1 arrest [140]. Similarly, silencing MALAT1 in proliferating WI-38 cells enhanced senescence and induced G1/S arrest, two sets of effects that were linked to p53 actions [34, 141]. Together, these studies indicate that MALAT1 might be essential for cell proliferation. Unexpectedly, however MALAT1-knockout mice lacked an obvious phenotype or histological abnormalities, indicating that MALAT1 is not essential for development [142], although its impact may be revealed under specific pathological or environmental conditions.
ANRIL. As mentioned above, the lncRNA ANRIL is expressed from the p15/CDKN2B/CDKN2A/ARF gene cluster [143]. Downregulation of ANRIL significantly enhanced the levels of the cdk inhibitor (cdki) p15 and moderately increased the levels of cdki p16. Genome-wide analysis indicated that ANRIL levels decline in senescent WI-38 cells, and its silencing increased the abundance of cell cycle inhibitors and induced senescence [34, 144]. While these findings indicate that ANRIL is required for cell cycle progression and suppression of senescence, further studies are warranted to understand the roles of ANRIL more broadly in aging.
NcRNACCND1. The CCND1-associated lncRNA NcRNACCND1 modulates the expression of the cell cycle regulator cyclin D1 (CCND1), which is required for the activity of cdks, particularly Cdk2 and cdk4, to progress through the G1/S phase transition [145]. Upon exposure to DNA-damaging agents, the NcRNACCND1 binds the RNA-binding protein TLS to form a ribonucleoprotein complex that is recruited to the CCND1 promoter and inhibits transcription [119]. Since CCND1 is a major regulator of the cell cycle progression, NcRNACCND1 will likely be implicated in aging-relevant processes such as cancer.
SRA.SRA is a co-activator of steroid receptors and other transcription factors such as the myogenic regulatory transcription factor MyoD and the major regulator of adipogenesis PPARγ [146, 147]. Overexpression of SRA promotes the differentiation of mesenchymal precursor cells into adipocytes; conversely, SRA knockdown inhibits preadipocyte differentiation. SRA decreases the expression of cdkis p21 and p27 and it increases phosphorylation of Cdk1 [146, 147]. Since SRA is expressed in a wide range of tissues [148], similar regulatory mechanisms may occur in a variety of tissues that impact upon cell cycle progression, senescence, and adipogenesis.
HEIH. The lncRNA HEIH is highly expressed in human Hepatitis B virus-related hepatocellular carcinoma (HCC). It suppresses the expression of cdkis p16, p21, p27, and p57 and hence facilitates tumor cell growth [149]. Future studies are warranted to study the role of HEIH lncRNA in aging.
HULC. The lncRNA HULC is highly expressed in hepatocytes, HCC, and hepatic colorectal carcinomas [150, 151]. Hepatitis B virus X protein induces CREB-mediated HULC expression, which subsequently promotes hepatoma cell proliferation by lowering the expression of the cdki p18 [152, 153]. Thus, HULC may play a role in cell cycle progression and cellular senescence.
Gadd7. As mentioned above, Gadd7 was upregulated by DNA damage [106]. Gadd7 decreases cell growth by binding TDP-43 and preventing its interaction with Cdk6 mRNA, leading to Cdk6 mRNA decay [107]. As CDK6 affects cell cycle and senescence [108, 154], it is expected to influence mammalian aging.
UCA1.UCA1 is highly expressed in bladder transitional cell carcinoma [155]. Recently, UCA1 was found to promote breast cancer cell growth by lowering the expression of the tumor suppressor and cdki p27. Silencing UCA1 enhanced hnRNP I abundance in the cytoplasm, facilitating p27 mRNA translation and triggering G1 cell cycle arrest [156]. These findings suggest that UCA1 can affect cellular senescence and carcinogenesis by modulating p27 levels [157].
eRNAs. Enhancer ncRNAs (eRNAs) are short (50-1500 bp) RNAs transcribed from certain enhancer regions to influence transcription at distant sites (up to 1 Mbp away) [158-160]. Binding of p53 to enhancer regions (p53BERs) produced p53-dependent eRNAs that modulated p53 transcriptional activity and induced p53-dependent cell cycle arrest [161], linking eRNAs influence to senescence, aging, and carcinogenesis.
H19. As mentioned above, H19 is an epigenetic regulatory RNA that affects cell growth and proliferation. In breast cancer cells, H19 knockdown decreased cell growth and formation of colonies in culture [162]. Hypoxia elevated H19 abundance in HHC cells and H19 knockdown significantly reduced tumor growth after recovery following hypoxia. Additionally, H19 overexpression enhanced tumor growth in Hep3B cells [163]. These findings indicate that the high levels of H19 in cancer cells led to enhanced growth and proliferation, as well as to the avoidance of cellular senescence. Indeed, H19 downregulation lowered the levels of p57, which participates in cell division, differentiation, cell survival, cell proliferation and tumorigenesis [39, 164-167].
MEG3. The lncRNA MEG3 is repressed in many human cancer cell lines due to gene deletion or to DNA hypermethylation. Forced expression of MEG3 inhibited the growth of human cancer cell lines such as HeLa, MCF-7, and H4, suggesting that MEG3 may act as a tumor suppressor [168], while downregulation of MEG3 enhanced autophagy, increased cell proliferation and inhibited cell death [99]. A major mechanism through which MEG3 regulates cell proliferation is by lowering the levels MDM2, a repressor of p53 [169]. MEG3 expression also induces apoptosis and growth arrest in cervical cancer cells [170]. Global analysis on transcriptional networks and cellular senescence in human mammary fibroblasts suggested that the NR4A3 (nuclear receptor subfamily 4, group A, member3) is upregulated in senescent cells and affects cell growth via MEG3 [171], further linking MEG3 to cellular senescence and carcinogenesis.
7SL. Unlike MEG3, 7SL lncRNA is highly expressed in cancer cells [14, 172]. 7SL silencing was found to promote cell cycle arrest and senescence in HeLa cells. These effects were mediated, at least in part, through binding to and suppressing TP53 mRNA translation by competing with the RNA-binding protein HuR, as explained above [14].
LncRNAs and intercellular communications
Factors associated with aging in one tissue can be secreted, transported, and received by other tissues. These intercellular communication events involve the sharing of factors such as inflammatory cytokines and extracellular vesicles, which are associated with senescence, aging-associated declines, and diseases of advancing age [1, 173, 174].
Inflammation-associated lncRNAs
Senescent cells actively secrete pro-inflammatory cytokines, a phenomenon known as the senescence-associated secretory phenotype (SASP) [1, 175]. Given that senescent cells accumulate in older tissues, they are believed to be directly responsible for the low-grade pro-inflammatory state that characterizes aging. The emerging role of lncRNAs in inflammation is discussed here.
17A. The lncRNA 17A is upregulated in cerebral tissues derived from Alzheimer disease patients as well as in response to inflammatory stimuli such as IL-1α. Interestingly, 17A is encoded within the G-protein-coupled receptor 51 (GPR51) gene and 17A overexpression enhanced secretion of Aβ and regulated GABA B alternative splicing and signaling [176]. In light of the fact that GPR function is associated with age-associated declines [177], 17A provides an interesting link between GPRs and age-associated neurodegeneration.
Lethe. In mouse embryonic fibroblasts (MEFs), 54 lncRNAs were found to be regulated following treatment with TNFα, a pro-inflammatory cytokine associated with inflammation, aging, age-related diseases, and cellular senescence [178, 179]. Among these lncRNAs, Lethe was found to be particularly important for the pro-inflammatory state of aging tissues by providing an key negative feedback loop: binding of Lethe to the NF-κB subunit RelA inhibited DNA binding and reduced the production of inflammatory proteins [178].
THRIL. TNFα was also found to be induced by lncRNA THRIL (TNFα- and hnRNPL-related immunoregulatory lincRNA). This lncRNA interacts with hnRNP L, forming a ribonucleoprotein complex that binds the TNFα promoter and regulates its expression in THP1 macrophages [120]. Together, lncRNAs lethe and THRIL are involved in inflammation in a TNFα-dependent manner.
Lnc-IL7R. Recently, Lnc-IL7R was identified as a regulator of the lipopolysaccharide (LPS)-induced inflammatory response. Depletion of Lnc-IL7R reduces trimethylation of histone H3 at lysine 27 (H3K27me3) causing a reduction in the levels of inflammatory mediators including E-selectin, VCAM-1, IL-6 and IL-8 [180]. Given that pro-inflammatory cytokines are secreted by senescent cells and contribute to the aging phenotype [181, 182], Lnc-IL7R might be involved in sense-cence or aging by regulating these inflammatory factors.
Other lncRNAs. Toll-like receptors (TLRs) are vital in the response of macrophages to pathological stimuli. In normal aging, defects in TLR signaling enhanced inflammation [183]. Recently, inflammatory lncRNA signatures were characterized by RNA sequencing in healthy individuals treated with low-dose LPS, which activates TLR4 signaling. The study revealed tissue-specific regulation of distinct lncRNAs in the inflammatory physiology and pathology of the cardiovascular system [184]. However, the impact of these lncRNAs on age-related processes has not been investigated directly.
The TGF-β/Smad3 pathway is involved in inducing and resolving the inflammatory response [185]. High-throughput RNA analysis was used to identify Smad3-dependent lncRNAs related to renal inflammation. The analysis identified numerous lncRNAs altered in Smad3 knockout mice [186]. As this pathway is impaired with age and Smad3 regulates senescence phenotype [187, 188], this network of proteins and lncRNAs differentially expressed may play a role in aging-associated inflammation (‘inflammaging’).
LncRNAs and extracellular signaling through exosome
Like inflammatory cytokines, RNAs can serve as extracellular signaling molecules. RNAs can be transferred between cells through gap junctions or via extracellular vesicles such as exosomes and microvesicles [189]. Due to their presence in all body fluids, exosomes are believed to participate in many biological processes, including those that directly affect aging [173]. All RNAs, including mRNAs, dsRNAs, and miRNAs, can be transported among cells [190, 191]. The examples below highlight emerging functions of lncRNAs in intercellular communication in age-relevant processes, particularly inflammation and growth inhibition.
TUC339. Among the lncRNAs found in extracellular vesicles from hepatocellular carcinoma cells, the lncRNA TUC339 modulates cell cycle progression, tumor growth, and adhesion [192, 193]. Since TUC339 can be transported to distant tissues via extracellular vesicles, it can serve as a messenger to modulate the cell division cycle in distant tissues.
Tie-1as. Also involved in cell-cell communication, the lncRNA tie-1as interacts with and represses the mRNA encoding for tyrosine kinase-containing immunoglobulin and epidermal growth factor homology domain-1 (Tie-1), causing defective endothelial cell contact junctions [194]. Although endothelial function declines with age, the impact of Tie-1as on aging has not been examined directly.
Linc-ROR. This lncRNA is highly enriched in extracellular vesicles derived from hepatocellular carcinoma cells. TGFβ regulates the levels of Linc-ROR in the vesicles and incubation of vesicles with tumor cells reduces chemotherapy-induced cell death in recipient cells. Suppression of Linc-ROR leads to increased chemotherapy-induced apoptosis and cytotoxicity, suggesting that this lncRNA is a modulator of chemoresistence in hepatocellular carcinoma cells [195].
Other lncRNAs. Recently, several lncRNAs that are typically present in low abundance in cells were found to be selectively enriched in secreted exosomes, including MALAT1, HOTAIR, LincRNA-p21, GAS5, TUG1 and CCND1-ncRNA. Some of these lncRNAs are discussed above, as they influence cell cycle regulation, cellular senescence, and other processes. Treatment with the DNA damaging agent bleomycin elevated the concentration of LincRNA-p21 and ncRNA-CCND1 in exosomes [196]. Since damaged DNA accumulates with advancing age, the accumulation of these lncRNAs in exosomes may serve as a marker for aging in tissues.
Concluding remarks and perspectives
Aging traits are governed through changes in subsets of expressed proteins. LncRNAs can modulate protein expression patterns by controlling gene transcription, mRNA stability, and protein abundance. Through this influence, lncRNAs modulate key molecular events underlying the aging process, including those discussed here – the control of telomere length, epigenetic gene expression, proteostasis, stem cell function, intercellular communication, cell proliferation and cellular senescence.
The examples discussed in this review underscore the growing recognition that lncRNAs critically affect both the physiologic decline that occurs with aging and the pathologies associated with advancing age. Accordingly, there is mounting interest in the diagnostic, prognostic, and therapeutic value of lncRNAs. The facile detection of lncRNAs in body fluids (e.g. exosomes) and the ease of design of molecules to increase or decrease lncRNA levels make them particularly attractive clinical targets.
However, the potential usefulness of lncRNAs in aging dysfunction and disease cannot be fully realized at present. First, we must have a more comprehensive understanding of age-associated lncRNAs, their spatiotemporal pattern of expression, the molecules with which they interact (proteins, DNA, and RNA), and the impact of altering their abundance upon cell function. Second, we need to develop suitable animal models in which we can study age-associated lncRNAs, assess their function in tissues, organs, and systems, and evaluate how they influence the aging process. By advancing in these areas of knowledge, we expect to gain a deeper molecular understanding of aging and develop more effective interventions to ameliorate the losses of advancing age.
Acknowledgments
This work was supported in its entirety by the National Institute on Aging-Intramural Research Program of the National Institutes of Health.
Conflicts of Interest
The authors of this manuscript have no conflict of interests to declare.
References
- 1. López-Otin C, Blasco MA, Partridge L, Serrano M, Kroemer G. The hallmarks of aging. Cell. 2013; 153: 1194 -1217. [PubMed] .
- 2. Rufini A, Tucci P, Celardo I, Melino G. Senescence and aging: the critical roles of p53. Oncogene. 2013; 32: 5129 -5143. [PubMed] .
- 3. Wang WG. Regulatory RNA-binding proteins in senescence. Ageing Res Rev. 2012; 11: 485 -490. [PubMed] .
- 4. Wu G, Wang D, Huang Y, Han JD. The research progress of microRNAs in aging. Prog Biochem Biophys. 2014; 41: 273 -287. .
- 5. Yoon JH, Abdelmohsen K, Gorospe M. Posttranscriptional gene regulation by long noncoding RNA. J Mol Biol. 2013; 425: 3723 -3730. [PubMed] .
- 6. Singh DK and Prasanth KV. Functional insights into the role of nuclear-retained long noncoding RNAs in gene expression control in mammalian cells. Chromosome Res. 2013; 21: 695 -711. [PubMed] .
- 7. Yoon JH, Abdelmohsen K, Kim J, Yang XL, Martindale JL, Tominaga-Yamanaka K, White EJ, Orjalo AV, Rinn JL, Kreft SG, Wilson GM, Gorospe M. Scaffold function of long non-coding RNA HOTAIR in protein ubiquitination. Nat Commun. 2013; 4: .
- 8. Kung JT, Colognori D, Lee JT. Long noncoding RNAs:past, present, and future. Genetics. 2013; 193: 651 -669. [PubMed] .
- 9. Spitale RC, Tsai MC, Chang HY. RNA templating the epigenome Long noncoding RNAs as molecular scaffolds. Epigenetics US. 2011; 6: 539 -543. .
- 10. Lee JT. Epigenetic regulation by long noncoding RNAs. Science. 2012; 338: 1435 -1439. [PubMed] .
- 11. Bergmann JH and Spector DL. Long non-coding RNAs:modulators of nuclear structure and function. Curr Opin Cell Biol. 2014; 26: 10 -18. [PubMed] .
- 12. Gong CG and Maquat LE. lncRNAs transactivate STAU1-mediated mRNA decay by duplexing with 3 ' UTRs via Alu elements. Nature. 2011; 470: 284 -288. [PubMed] .
- 13. Yoon JH, Abdelmohsen K, Srikantan S, Yang XL, Martindale JL, De S, Huarte M, Zhan M, Becker KG, Gorospe M. LincRNA-p21 suppresses target mRNA translation. Mol Cell. 2012; 47: 648 -655. [PubMed] .
- 14. Abdelmohsen K, Panda AC, Kang MJ, Guo R, Kim J, Grammatikakis I, Yoon JH, Dudekula DB, Noh JH, Yang X, Martindale JL, Gorospe M. 7SL RNA represses p53 translation by competing with HuR. Nucleic Acids Res. 2014; 42: 10099 -10111. [PubMed] .
- 15. Mattick JS. Long noncoding RNAs in cell and developmental biology. Semin Cell Dev Biol. 2011; 22: 327 -327. [PubMed] .
- 16. Anko ML and Neugebauer KM. Long noncoding RNAs add another layer to pre-mRNA splicing regulation. Mol Cell. 2010; 39: 833 -834. [PubMed] .
- 17. Cesana M, Cacchiarelli D, Legnini I, Santini T, Sthandier O, Chinappi M, Tramontano A, Bozzoni I. A long noncoding RNA controls muscle differentiation by functioning as a competing endogenous RNA. Cell. 2011; 147: 358 -369. [PubMed] .
- 18. Tay Y, Rinn J, Polfi PP. The multilayered complexity of ceRNA crosstalk and competition. Nature. 2014; 505: 344 -352. [PubMed] .
- 19. Kuilman T, Michaloglou C, Mooi WJ, Peeper DS. The essence of senescence. Genes Dev. 2010; 24: 2463 -2479. [PubMed] .
- 20. Samper E, Flores JM, Blasco MA. Restoration of telomerase activity rescues chromosomal instability and premature aging in Terc−/− mice with short telomeres. EMBO Rep. 2001; 2: 800 -807. [PubMed] .
- 21. Collins K. Physiological assembly and activity of human telomerase complexes. Mech Ageing Dev. 2008; 129: 91 -98. [PubMed] .
- 22. Azzalin CM, Reichenbach P, Khoriauli L, Giulotto E, Lingner J. Telomeric repeat-containing RNA and RNA surveillance factors at mammalian chromosome ends. Science. 2007; 318: 798 -801. [PubMed] .
- 23. Porro A, Feuerhahn S, Lingner J. TERRA-reinforced association of LSD1 with MRE11 promotes processing of uncapped telomeres. Cell Rep. 2014; 6: 765 -776. [PubMed] .
- 24. Redon S, Reichenbach P, Lingner J. The non-coding RNA TERRA is a natural ligand and direct inhibitor of human telomerase. Nucleic Acids Res. 2010; 38: 5797 -5806. [PubMed] .
- 25. Deng Z, Campbell AE, Lieberman PM. TERRA, CpG methylation and telomere heterochromatin: lessons from ICF syndrome cells. Cell Cycle. 2010; 9: 69 -74. [PubMed] .
- 26. Yu TY, Kao YW, Lin JJ. Telomeric transcripts stimulate telomere recombination to suppress senescence in cells lacking telomerase. Proc Natl Acad Sci USA. 2014; 111: 3377 -3382. [PubMed] .
- 27. Flynn RL, Centore RC, O'Sullivan RJ, Rai R, Tse A, Songyang Z, Chang S, Karlseder J, Zou L. TERRA and hnRNPA1 orchestrate an RPA-to-POT1 switch on telomeric single-stranded DNA. Nature. 2011; 471: 532 -536. [PubMed] .
- 28. Munoz-Najar U and Sedivy JM. Epigenetic control of aging. Antiox. Redox Sig. 2011; 14: 241 -259. .
- 29. Pawlikowski JS, Adams PD, Nelson DM. Senescence at a glance. J Cell Sci. 2013; 126: 4061 -4067. [PubMed] .
- 30. Johnson AA, Akman K, Calimport SR, Wuttke D, Stolzing A, de Magalhaes JP. The role of DNA methylation in aging, rejuvenation, and age-related disease. Rejuv Res. 2012; 15: 483 -494. .
- 31. Maegawa S, Hinkal G, Kim HS, Shen L, Zhang L, Zhang J, Zhang N, Liang S, Donehower LA, Issa JP. Widespread and tissue specific age-related DNA methylation changes in mice. Genome Res. 2010; 20: 332 -340. [PubMed] .
- 32. Umlauf D, Fraser P, Nagano T. The role of long non-coding RNAs in chromatin structure and gene regulation:variations on a theme. Biol Chem. 2008; 389: 323 -331. [PubMed] .
- 33. Sado T and Brockdorff N. Advances in understanding chromosome silencing by the long non-coding RNA Xist. Philos Trans R Soc Lond B Biol Sci. 2013; 368: 20110325 [PubMed] .
- 34. Abdelmohsen K, Panda A, Kang MJ, Xu J, Selimyan R, Yoon JH, Martindale JL, De S, Wood WH 3rd, Becker KG, Gorospe M. Senescence-associated lncRNAs:senescence-associated long noncoding RNAs. Aging Cell. 2013; 12: 890 -900. [PubMed] .
- 35. Ratajczak MZ. Igf2-H19, an imprinted tandem gene, is an important regulator of embryonic development, a guardian of proliferation of adult pluripotent stem cells, a regulator of longevity, and a 'passkey' to cancerogenesis. Folia Histochem Cyto. 2012; 50: 171 -179. .
- 36. Monnier P, Martinet C, Pontis J, Stancheva I, Ait-Si-Ali S, Dolo L. H19 lncRNA controls gene expression of the Imprinted Gene Network by recruiting MBD1. Proc Natl Acad Sci USA. 2013; 110: 20693 -20698. [PubMed] .
- 37. Thorvaldsen JL, Duran KL, Bartolomei MS. Deletion of the H19 differentially methylated domain results in loss of imprinted expression of H19 and Igf2. Genes Dev. 1998; 12: 3693 -3702. [PubMed] .
- 38. Gabory A, Jammes H, Dolo L. The H19 locus:Role of an imprinted non-coding RNA in growth and development. Bioessays. 2010; 32: 473 -480. [PubMed] .
- 39. Matouk I, DeGroot N, Mezan S, Galun E, Hochberg A. The H19 non-coding RNA is essential for human tumor growth and may serve as an anti-tumor target. Hum Gene Ther. 2007; 18: 1034 -1034. .
- 40. Issa JPJ, Vertino PM, Boehm CD, Newsham IF, Baylin SB. Switch from monoallelic to biallelic human IGF2 promoter methylation during aging and carcinogenesis. Proc Natl Acad Sci USA. 1996; 93: 11757 -11762. [PubMed] .
- 41. Fu VX, Dobosy JR, Desotelle JA, Almassi N, Ewald JA, Srinivasan R, Berres M, Svaren J, Weindruch R, Jarrard DF. Aging and cancer-related loss of insulin-like growth factor 2 imprinting in the mouse and human prostate. Cancer Res. 2008; 68: 6797 -6802. [PubMed] .
- 42. Christofori G, Naik P, Hanahan D. A second signal supplied by insulin-like growth factor II in oncogene-induced tumorigenesis. Nature. 1994; 369: 414 -418. [PubMed] .
- 43. Travers ME, Mackay DJ, Dekker Nitert M, Morris AP, Lindgren CM, Berry A, Johnson PR, Hanley N, Groop LC, McCarthy MI, Gloyn AL. Insights into the molecular mechanism for type 2 diabetes susceptibility at the KCNQ1 locus from temporal changes in imprinting status in human islets. Diabetes. 2013; 62: 987 -992. [PubMed] .
- 44. Sazhenova EA and Lebedev IN. Epimutations of the KCNQ1OT1 imprinting center of chromosome 11 in early human embryo lethality. Genetika. 2008; 44: 1609 -1616. [PubMed] .
- 45. Latos PA, Pauler FM, Koerner MV, Senergin HB, Hudson QJ, Stocsits RR, Allhoff W, Stricker SH, Klement RM, Warczok KE, Aumayr K, Pasierbek P, Barlow DP. Airn transcriptional overlap, but not its lncRNA products, induces imprinted Igf2r silencing. Science. 2012; 338: 1469 -1472. [PubMed] .
- 46. Xu W, Zhuang ZX, Yang JP, Yang LQ, Xu YL, Zhang WJ, Zeng Y. The profile of IGF2R gene expression and H3 histone modifications in replicative cell senescence. J Sichuan Univ Med Sci. 2014; 45: 6 -9. .
- 47. Abdul Rahman A, Abdul Karim N, Abdul Hamid NA, Harun R, Ngah WZ. Senescence-related changes in gene expression of peripheral blood mononuclear cells from octo/nonagenarians compared to their offspring. Oxid Med Cell Longev. 2013; 9: 18912 .
- 48. Di Ruscio A, Ebralidze AK, Benoukraf T, Amabile G, Goff LA, Terragni J, Figueroa ME, De Figueiredo Pontes LL, Alberich-Jorda M, Zhang P, Wu M, D'Alò F, Melnick A, et al. DNMT1-interacting RNAs block gene-specific DNA methylation. Nature. 2013; 503: 371 -376. [PubMed] .
- 49. Wang HM, Iakova P, Wilde M, Welm A, Goode T, Roesler WJ, Timchenko NA. C/EBP alpha arrests cell proliferation through direct inhibition of cdk2 and cdk4. Mol Cell. 2001; 8: 817 -828. [PubMed] .
- 50. Karagiannides I, Tchkonia T, Dobson DE, Steppan CM, Cummins P, Chan G, Salvatori K, Hadzopoulou-Cladaras M, Kirkl JL. Altered expression of C/EBP family members results in decreased adipogenesis with aging. Am J Physiol. 2001; 280: R1772 -R1780. .
- 51. Hong IH, Lewis K, Iakova P, Jin JL, Sullivan E, Jawanmardi N, Timchenko L, Timchenko N. Age-associated change of C/EBP family proteins causes severe liver injury and acceleration of liver proliferation after CCl4 treatments. J Biol Chem. 2014; 289: 1106 -1118. [PubMed] .
- 52. Huggins CJ, Malik R, Lee S, Salotti J, Thomas S, Martin N, Quiñones OA, Alvord WG, Olanich ME, Keller JR, Johnson PF. C/EBP gamma suppresses senescence and inflammatory gene expression by heterodimerizing with C/EBP beta. Mol Cell Biol. 2013; 33: 3242 -3258. [PubMed] .
- 53. Schmitz KM, Mayer C, Postepska A, Grummt I. Interaction of noncoding RNA with the rDNA promoter mediates recruitment of DNMT3b and silencing of rRNA genes. Genes Dev. 2010; 24: 2264 -2269. [PubMed] .
- 54. Machwe A, Orren DK, Bohr VA. Accelerated methylation of ribosomal RNA genes during the cellular senescence of Werner syndrome fibroblasts. FASEB J. 2000; 14: 1715 -1724. [PubMed] .
- 55. Johnson R and Strehler BL. Loss of genes coding for ribosomal-RNA in aging brain cells. Nature. 1972; 240: 412 -414. [PubMed] .
- 56. Pietrzak M, Rempala G, Nelson PT, Zheng JJ, Hetman M. Epigenetic silencing of nucleolar rRNA genes in Alzheimer's disease. PLoS ONE. 2011; 6: e22585 [PubMed] .
- 57. Bierhoff H, Schmitz K, Maass F, Ye J, Grummt I. Noncoding transcripts in sense and antisense orientation regulate the epigenetic state of ribosomal RNA genes. Cold Spring Harbor Symp Quant Biol. 2010; 75: 357 -364. [PubMed] .
- 58. Bierhoff H, Dammert MA, Brocks D, Dambacher S, Schotta G, Grummt I. Quiescence-induced LncRNAs trigger H4K20 trimethylation and transcriptional silencing. Mol Cell. 2014; 54: 675 -682. [PubMed] .
- 59. Johnsson P, Ackley A, Vidarsdottir L, Lui WO, Corcoran M, Grander D, Morris KV. A pseudogene long-noncoding-RNA network regulates PTEN transcription and translation in human cells. Nat Struct Mol Biol. 2013; 20: 440 -446. [PubMed] .
- 60. Kitagishi Y and Matsuda S. Redox regulation of tumor suppressor PTEN in cancer and aging. Intl J Mol Med. 2013; 31: 511 -515. .
- 61. Ortega-Molina A and Serrano M. PTEN in cancer, metabolism, and aging. Trends Endoncrinol Metab. 2013; 24: 184 -189. .
- 62. Tarnawski AS, Pai R, Tanigawa T, Matysiak-Budnik T, Ahluwalia A. PTEN silencing reverses aging-related impairment of angiogenesis in microvascular endothelial cells. Biochem Biophys Res Commun. 2010; 394: 291 -296. [PubMed] .
- 63. Moon SH, Kim DK, Cha Y, Jeon I, Song J, Park KS. PI3K/Akt and Stat3 signaling regulated by PTEN control of the cancer stem cell population, proliferation and senescence in a glioblastoma cell line. Intl J Oncol. 2013; 42: 921 -928. .
- 64. Collado M. Exploring a 'pro-senescence' approach for prostate cancer therapy by targeting PTEN. Future Oncol. 2010; 6: 687 -689. [PubMed] .
- 65. Arab K, Park YJ, Lindroth AM, Schafer A, Oakes C, Weichenhan D, Lukanova A, Lundin E, Risch A, Meister M, Dienemann H, Dyckhoff G, Herold-Mende C, et al. Long noncoding RNA TARID directs demethylation and activation of the tumor suppressor TCF21 via GADD45A. Mol Cell. 2014; 55: 604 -614. [PubMed] .
- 66. D'Aquila P, Rose G, Bellizzi D, Passarino G. Epigenetics and aging. Maturitas. 2013; 74: 130 -136. [PubMed] .
- 67. Zhang W, Hu D, Ji W, Yang L, Yang J, Yuan J, Xuan A, Zou F, Zhuang Z. Histone modifications contribute to cellular replicative and hydrogen peroxide-induced premature senescence in human embryonic lung fibroblasts. Free Rad Res. 2014; 48: 550 -559. .
- 68. Han S and Brunet A. Histone methylation makes its mark on longevity. Trends Cell Biol. 2012; 22: 42 -49. [PubMed] .
- 69. Fraga MF and Esteller M. Epigenetics and aging:the targets and the marks. Trends Genet. 2007; 23: 413 -418. [PubMed] .
- 70. Jin C, Li J, Green CD, Yu X, Tang X, Han D, Xian B, Wang D, Huang X, Cao X, Yan Z, Hou L, Liu J, et al. Histone demethylase UTX-1 regulates C. elegans life span by targeting the insulin/IGF-1 signaling pathway. Cell Metab. 2011; 14: 161 -172. [PubMed] .
- 71. Greer EL, Maures TJ, Hauswirth AG, Green EM, Leeman DS, Maro GS, Han S, Banko MR, Gozani O. Members of the H3K4 trimethylation complex regulate lifespan in a germline-dependent manner in C. elegans. Nature. 2010; 466: 383 -U137. [PubMed] .
- 72. Siebold AP, Banerjee R, Tie F, Kiss DL, Moskowitz J, Harte PJ. Polycomb Repressive Complex 2 and Trithorax modulate Drosophila longevity and stress resistance. Proc Natl Acad Sci USA. 2010; 107: 169 -174. [PubMed] .
- 73. Moric-Janiszewska E, Glogowska-Ligus J, Paul-Samojedny M, Weglarz L, Markiewicz-Loskot G, Szydlowski L. Age-and sex-dependent mRNA expression of KCNQ1 and HERG in patients with long QT syndrome type 1 and 2. Arch Med Sci AMS. 2011; 7: 941 -947. .
- 74. Korostowski L, Sedlak N, Engel N. The Kcnq1ot1 long non-coding RNA affects chromatin conformation and expression of Kcnq1, but does not regulate its imprinting in the developing heart. PLoS Genet. 2012; 8: e1002956 [PubMed] .
- 75. Pandey RR, Mondal T, Mohammad F, Enroth S, Redrup L, Komorowski J, Nagano T, Mancini-Dinardo D, Kanduri C, et al. Kcnq1ot1 antisense noncoding RNA mediates lineage-specific transcriptional silencing through chromatin-level regulation. Mol Cell. 2008; 32: 232 -246. [PubMed] .
- 76. Gomez JA, Wapinski OL, Yang YW, Bureau JF, Gopinath S, Monack DM, Chang HY, Brahic M, Kirkegaard K. The NeST long ncRNA controls microbial susceptibility and epigenetic activation of the interferon-gamma locus. Cell. 2013; 152: 743 -754. [PubMed] .
- 77. Yap KL, Li S, Munoz-Cabello AM, Raguz S, Zeng L, Mujtaba S, Gil J, Walsh MJ, Zhou MM. Molecular interplay of the noncoding RNA ANRIL and methylated histone H3 lysine 27 by polycomb CBX7 in transcriptional silencing of INK4a. Mol Cell. 2010; 38: 662 -674. [PubMed] .
- 78. Kim DH, Kim JS, Ji YI, Shim YM, Kim H, Han J, Park J. Hypermethylation of RASSF1A promoter is associated with the age at starting smoking and a poor prognosis in primary non-small cell lung cancer. Cancer Res. 2003; 63: 3743 -3746. [PubMed] .
- 79. Beckedorff FC, Ayupe AC, Crocci-Souza R, Amaral MS, Nakaya HI, Soltys DT, Menck CF, Reis EM, Verjovski-Almeida S. The Intronic long noncoding RNA ANRASSF1 recruits PRC2 to the RASSF1A promoter, reducing the expression of RASSF1A and increasing cell proliferation. PLoS Genet. 2013; 9: 8 e1003705 [PubMed] .
- 80. Marin-Bejar O, Marchese FP, Athie A, Sanchez Y, Gonzalez J, Segura V, Huang L, Moreno I, Navarro A, Monzó M, García-Foncillas J, Rinn JL, Guo S, et al. Pint lincRNA connects the p53 pathway with epigenetic silencing by the Polycomb repressive complex 2. Genome Biol. 2013; 14: 9 r104 [PubMed] .
- 81. Krieglstein K, Miyazono K, ten Dijke P, Unsicker K. TGF-beta in aging and disease. Cell Tissue Res. 2012; 347: 5 -9. [PubMed] .
- 82. Williamson D, Gallagher P, Harber M, Hollon C, Trappe S. Mitogen-activated protein kinase (MAPK) pathway activation:effects of age and acute exercise on human skeletal muscle. J Phsyiol. 2003; 547: 977 -987. .
- 83. Narita M, Narita M, Krizhanovsky V, Nunez S, Chicas A, Hearn SA, Myers MP, Lowe SW. A novel role for high-mobility group a proteins in cellular senescence and heterochromatin formation. Cell. 2006; 126: 503 -514. [PubMed] .
- 84. Zhang R, Poustovoitov MV, Ye X, Santos HA, Chen W, Daganzo SM, Erzberger JP, Serebriiskii IG, Canutescu AA, Dunbrack RL, Pehrson JR, Berger JM, Kaufman PD, Adams PD. Formation of MacroH2A-containing senescence-associated heterochromatin foci and senescence driven by ASF1a and HIRA. Dev Cell. 2005; 8: 19 -30. [PubMed] .
- 85. Tsurumi A and Li WX. Global heterochromatin loss: a unifying theory of aging? Epigenetics. 2012; 7: 680 -688. [PubMed] .
- 86. Oberdoerffer P and Sinclair DA. The role of nuclear architecture in genomic instability and ageing. Nat Rev Mol Cell Biol. 2007; 8: 692 -702. [PubMed] .
- 87. Larson K, Yan SJ, Tsurumi A, Liu J, Zhou J, Gaur K, Guo D, Eickbush TH, Li WX. Heterochromatin formation promotes longevity and represses ribosomal RNA synthesis. PLoS Genet. 2012; 8: e1002473 [PubMed] .
- 88. Schotta G, Lachner M, Sarma K, Ebert A, Sengupta R, Reuter G, Reinberg D, Jenuwein T. A silencing pathway to induce H3-K9 and H4-K20 trimethylation at constitutive heterochromatin. Genes Dev. 2004; 18: 1251 -1262. [PubMed] .
- 89. Deng Z, Norseen J, Wiedmer A, Riethman H, Lieberman PM. TERRA RNA binding to TRF2 facilitates heterochromatin formation and ORC recruitment at telomeres. Mol Cell. 2009; 35: 403 -413. [PubMed] .
- 90. Keller C, Kulasegaran-Shylini R, Shimada Y, Hotz HR, Buhler M. Noncoding RNAs prevent spreading of a repressive histone mark. Nat Struct Mol Biol. 2013; 20: 994 -1000. [PubMed] .
- 91. Anderson RM. A Role for Dicer in Aging and Stress Survival. Cell Metab. 2012; 16: 285 -286. [PubMed] .
- 92. Srikantan S, Marasa BS, Becker KG, Gorospe M, Abdelmohsen K. Paradoxical microRNAs Individual gene repressors, global translation enhancers. Cell Cycle. 2011; 10: 751 -759. [PubMed] .
- 93. Powers ET, Morimoto RI, Dillin A, Kelly JW, Balch WE. Biological and chemical approaches to diseases of proteostasis deficiency. Annual Rev Biochem. 2009; 78: 959 -991. [PubMed] .
- 94. Balch WE, Morimoto RI, Dillin A, Kelly JW. Adapting proteostasis for disease intervention. Science. 2008; 319: 916 -919. [PubMed] .
- 95. Rubinsztein DC, Marino G, Kroemer G. Autophagy and aging. Cell. 2011; 146: 682 -695. [PubMed] .
- 96. Gewirtz DA. Autophagy and senescence:a partnership in search of definition. Autophagy. 2013; 9: 808 -812. [PubMed] .
- 97. Zhao Y, Guo Q, Chen J, Hu J, Wang S, Sun Y. Role of long non-coding RNA HULC in cell proliferation, apoptosis and tumor metastasis of gastric cancer:a clinical and in vitro investigation. Oncology Rep. 2014; 31: 358 -364. .
- 98. Zhang Q, Su M, Lu G, Wang J. The complexity of bladder cancer:long noncoding RNAs are on the stage. Mol Cancer. 2013; 12: 101 [PubMed] .
- 99. Ying L, Huang Y, Chen H, Wang Y, Xia L, Chen Y, Liu Y, Qiu F. Downregulated MEG3 activates autophagy and increases cell proliferation in bladder cancer. Mol Biosystems. 2013; 9: 407 -411. .
- 100. Baldassarre A and Masotti A. Long Non-Coding RNAs and p53 Regulation. Intl J Mol Sci. 2012; 13: 16708 -16717. .
- 101. Tasdemir E, Maiuri MC, Morselli E, Criollo A, D'Amelio M, Djavaheri-Mergny M, Cecconi F, Tavernarakis N, Kroemer G. A dual role of p53 in the control of autophagy. Autophagy. 2008; 4: 810 -814. [PubMed] .
- 102. Mourtada-Maarabouni M, Pickard MR, Hedge VL, Farzaneh F, Williams GT. GAS5, a non-protein-coding RNA, controls apoptosis and is downregulated in breast cancer. Oncogene. 2009; 28: 195 -208. [PubMed] .
- 103. Lee SY, Hwang YK, Yun HS, Han JS. Decreased levels of nuclear glucocorticoid receptor protein in the hippocampus of aged Long-Evans rats with cognitive impairment. Brain Res. 2012; 1478: 48 -54. [PubMed] .
- 104. Di Agostino S, Strano S, Emiliozzi V, Zerbini V, Mottolese F, Sacchi A, Blandino G, Piaggio G. Gain of function of mutant p53: The mutant p53/NF-Y protein complex reveals an aberrant transcriptional mechanism of cell cycle regulation. Cancer Cell. 2006; 10: 191 -202. [PubMed] .
- 105. Matuoka K and Chen KY. Possible role of subunit A of nuclear factor Y (NF-YA) in normal human diploid fibroblasts during senescence. Biogerontol. 2000; 1: 261 -271. .
- 106. Hollander MC, Alamo I, Fornace AJ Jr. A novel DNA damage-inducible transcript, gadd7, inhibits cell growth, but lacks a protein product. Nucleic Acids Res. 1996; 24: 1589 -1593. [PubMed] .
- 107. Liu X, Li D, Zhang W, Guo M, Zhan Q. Long non-coding RNA gadd7 interacts with TDP-43 and regulates Cdk6 mRNA decay. EMBO J. 2012; 31: 4415 -4427. [PubMed] .
- 108. Rader J, Russell MR, Hart LS, Nakazawa MS, Belcastro LT, Martinez D, Li Y, Carpenter EL, Attiyeh EF, Diskin SJ, Kim S, Parasuraman S, Caponigro G, et al. Dual CDK4/CDK6 inhibition induces cell-cycle arrest and senescence in neuroblastoma. Clin Cancer Res. 2013; 19: 6173 -6182. [PubMed] .
- 109. Wang Y, Xu Z, Jiang J, Xu C, Kang J, Xiao L, Guo X, Liu H. Endogenous miRNA sponge lincRNA-RoR regulates Oct4, Nanog, and Sox2 in human embryonic stem cell self-renewal. Dev Cell. 2013; 25: 69 -80. [PubMed] .
- 110. Carrieri C, Cimatti L, Biagioli M, Beugnet A, Zucchelli S, Fedele S, Pesce E, Ferrer I, Collavin L, Santoro C, Forrest AR, Carninci P, Biffo S, et al. Long non-coding antisense RNA controls Uchl1 translation through an embedded SINEB2 repeat. Nature. 2012; 491: 454 -457. [PubMed] .
- 111. Maraganore DM, Lesnick TG, Elbaz A, Chartier-Harlin MC, Gasser T, Kruger R, Hattori N, Mellick GD, Quattrone A, Satoh J, Toda T, Wang J, Ioannidis JP, de Andrade M, et al. UCHL1 is a Parkinson's disease susceptibility gene. Annals Neurol. 2004; 55: 512 -521. .
- 112. Ummanni R, Jost E, Braig M, Lohmann F, Mundt F, Barett C, Schlomm T, Sauter G, Senff T, Bokemeyer C, Sültmann H, Meyer-Schwesinger C, Brümmendorf TH, et al. Ubiquitin carboxyl-terminal hydrolase 1 (UCHL1) is a potential tumour suppressor in prostate cancer and is frequently silenced by promoter methylation. Mol Cancer. 2011; 1476-4598-10-129 .
- 113. Konishi N, Shimada K, Nakamura M, Ishida E, Ota I, Tanaka N, Fujimoto K. Function of junB in transient amplifying cell senescence and progression of human prostate cancer. Clin Cancer Res. 2008; 14: 4408 -4416. [PubMed] .
- 114. Marchand A, Atassi F, Gaaya A, Leprince P, Le Feuvre C, Soubrier F, Lompré AM, Nadaud S. The Wnt/beta-catenin pathway is activated during advanced arterial aging in humans. Aging Cell. 2011; 10: 220 -232. [PubMed] .
- 115. Ye XF, Zerlanko B, Kennedy A, Banumathy G, Zhang RG, Adams PD. Downregulation of Wnt signaling is a trigger for formation of facultative heterochromatin and onset of cell senescence in primary human cells. Mol Cell. 2007; 27: 183 -196. [PubMed] .
- 116. Xu M, Yu Q, Subrahmanyam R, Difilippantonio MJ, Ried T, Sen JM. beta-catenin expression results in p53-independent DNA damage and oncogene-induced senescence in prelymphomagenic thymocytes in vivo. Mol Cell Biol. 2008; 28: 1713 -1723. [PubMed] .
- 117. Lustig AJ. Telomerase, RNA:a flexible RNA scaffold for telomerase biosynthesis. Curr Biol. 2004; 14: R565 -567. [PubMed] .
- 118. Greider CW and Blackburn EH. A telomeric sequence in the RNA of Tetrahymena telomerase required for telomere repeat synthesis. Nature. 1989; 337: 331 -337. [PubMed] .
- 119. Kurokawa R. Promoter-associated long noncoding RNAs repress transcription through a RNA binding protein TLS. Adv Exp Med Biol. 2011; 722: 196 -208. [PubMed] .
- 120. Li Z, Chao TC, Chang KY, Lin N, Patil VS, Shimizu C, Head SR, Burns JC, Rana TM. The long noncoding RNA THRIL regulates TNFalpha expression through its interaction with hnRNPL. Proc Natl Acad Sci USA. 2014; 111: 1002 -1007. [PubMed] .
- 121. Li YQ. Master stem cell transcription factors and signaling regulation. Cellular Reprog. 2010; 12: 3 -13. .
- 122. Boyer LA, Plath K, Zeitlinger J, Brambrink T, Medeiros LA, Lee TI, Levine SS, Wernig M, Tajonar A, Ray MK, Bell GW, Otte AP, Vidal M, et al. Polycomb complexes repress developmental regulators in murine embryonic stem cells. Nature. 2006; 441: 349 -353. [PubMed] .
- 123. Sheik Mohamed J, Gaughwin PM, Lim B, Robson P, Lipovich L. Conserved long noncoding RNAs transcriptionally regulated by Oct4 and Nanog modulate pluripotency in mouse embryonic stem cells. RNA. 2010; 16: 324 -337. [PubMed] .
- 124. Guttman M, Donaghey J, Carey BW, Garber M, Grenier JK, Munson G, Young G, Lucas AB, Ach R, Bruhn L, Yang X, Amit I, Meissner A, et al. lincRNAs act in the circuitry controlling pluripotency and differentiation. Nature. 2011; 477: 295 -300. [PubMed] .
- 125. Ballas N, Grunseich C, Lu DD, Speh JC, Mel G. REST and its corepressors mediate plasticity of neuronal gene chromatin throughout neurogenesis. Cell. 2005; 121: 645 -657. [PubMed] .
- 126. Cai L, Bian M, Liu M, Sheng Z, Suo H, Wang Z, Huang F, Fei J. Ethanol-induced neurodegeneration in NRSF/REST neuronal conditional knockout mice. Neurosci. 2011; 181: 196 -205. .
- 127. Whalley K. Neurodegeneration:Ageing neurons need REST. Nat Rev Neurosci. 2014; 15: 279 [PubMed] .
- 128. Johnson R, Teh CH, Jia H, Vanisri RR, Pandey T, Lu ZH, Buckley NJ, Stanton LW, Lipovich L. Regulation of neural macroRNAs by the transcriptional repressor REST. RNA. 2009; 15: 85 -96. [PubMed] .
- 129. Ng SY, Johnson R, Stanton LW. Human long non-coding RNAs promote pluripotency and neuronal differentiation by association with chromatin modifiers and transcription factors. EMBO J. 2012; 31: 522 -533. [PubMed] .
- 130. Loewer S, Cabili MN, Guttman M, Loh YH, Thomas K, Park IH, Garber M, Curran M, Onder T, Agarwal S, Manos PD, Datta S, Lander ES, et al. Large intergenic non-coding RNA-RoR modulates reprogramming of human induced pluripotent stem cells. Nat Genet. 2010; 42: 1113 -1117. [PubMed] .
- 131. Cheng EC and Lin H. Repressing the repressor:a lincRNA as a MicroRNA sponge in embryonic stem cell self-renewal. Dev Cell. 2013; 25: 1 -2. [PubMed] .
- 132. Zhang A, Zhou N, Huang J, Liu Q, Fukuda K, Ma D, Lu Z, Bai C, Watabe K, Mo YY. The human long non-coding RNA-RoR is a p53 repressor in response to DNA damage. Cell Res. 2013; 23: 340 -350. [PubMed] .
- 133. Dinger ME, Amaral PP, Mercer TR, Pang KC, Bruce SJ, Gardiner BB, Askarian-Amiri ME, Ru K, Soldà G, Simons C, Sunkin SM, Crowe ML, Grimmond SM, Perkins AC, et al. Long noncoding RNAs in mouse embryonic stem cell pluripotency and differentiation. Genome Res. 2008; 18: 1433 -1445. [PubMed] .
- 134. Wu SC, Kallin EM, Zhang Y. Role of H3K27 methylation in the regulation of lncRNA expression. Cell Res. 2010; 20: 1109 -1116. [PubMed] .
- 135. van Deursen JM. The role of senescent cells in ageing. Nature. 2014; 509: 439 -446. [PubMed] .
- 136. Mao Z, Ke Z, Gorbunova V, Seluanov A. Replicatively senescent cells are arrested in G1 and G2 phases. Aging. 2012; 4: 431 -435. [PubMed] .
- 137. Zender L and Rudolph KL. Keeping your senescent cells under control. Aging. 2009; 1: 438 -441. [PubMed] .
- 138. Chen JH, Hales CN, Ozanne SE. DNA damage, cellular senescence and organismal ageing:causal or correlative? Nucleic Acids Res. 2007; 35: 7417 -7428. [PubMed] .
- 139. Guo F, Li Y, Liu Y, Wang J, Li Y, Li G. Inhibition of metastasis-associated lung adenocarcinoma transcript 1 in CaSki human cervical cancer cells suppresses cell proliferation and invasion. Acta Biochim Biophys Sinica. 2010; 42: 224 -229. .
- 140. Zhao Z, Chen C, Liu Y, Wu C. 17beta-Estradiol treatment inhibits breast cell proliferation, migration and invasion by decreasing MALAT-1 RNA level. Bioch Biphys Res Commun. 2014; 445: 388 -93. .
- 141. Tripathi V, Shen Z, Chakraborty A, Giri S, Freier SM, Wu X, Wu X, Zhang Y, Gorospe M, Prasanth SG, Lal A, Prasanth KV. Long noncoding RNA MALAT1 controls cell cycle progression by regulating the expression of oncogenic transcription factor B-MYB. PLoS Genet. 2013; 9: e1003368 [PubMed] .
- 142. Eissmann M, Gutschner T, Hammerle M, Gunther S, Caudron-Herger M, Gross M, Schirmacher P, Rippe K, Braun T, Zörnig M, Diederichs S. Loss of the abundant nuclear non-coding RNA MALAT1 is compatible with life and development. RNA Biol. 2012; 9: 1076 -1087. [PubMed] .
- 143. Pasmant E, Laurendeau I, Heron D, Vidaud M, Vidaud D, Bieche I. Characterization of a germ-line deletion, including the entire INK4/ARF locus, in a melanoma-neural system tumor family:Identification of ANRIL, an antisense noncoding RNA whose expression coclusters with ARF. Cancer Res. 2007; 67: 3963 -3969. [PubMed] .
- 144. Kotake Y, Nakagawa T, Kitagawa K, Suzuki S, Liu N, Kitagawa M, Xiong Y. Long non-coding RNA ANRIL is required for the PRC2 recruitment to and silencing of p15(INK4B) tumor suppressor gene. Oncogene. 2011; 30: 1956 -1962. [PubMed] .
- 145. Wang X, Arai S, Song X, Reichart D, Du K, Pascual G, Tempst P, Rosenfeld MG, Glass CK, Kurokawa R. Induced ncRNAs allosterically modify RNA-binding proteins in cis to inhibit transcription. Nature. 2008; 454: 126 -130. [PubMed] .
- 146. Hube F, Velasco G, Rollin J, Furling D, Francastel C. Steroid receptor RNA activator protein binds to and counteracts SRA RNA-mediated activation of MyoD and muscle differentiation. Nucleic Acids Res. 2011; 39: 513 -525. [PubMed] .
- 147. Xu B, Gerin I, Miao H, Vu-Phan D, Johnson CN, Xu R, Chen XW, Cawthorn WP, MacDougald OA, Koenig RJ. Multiple roles for the non-coding RNA SRA in regulation of adipogenesis and insulin sensitivity. PLoS ONE. 2010; 5: 12 e14199 [PubMed] .
- 148. Lanz RB, Chua SS, Barron N, Soder BM, DeMayo F, O'Malley BW. Steroid receptor RNA activator stimulates proliferation as well as apoptosis in vivo. Mol Cell Biol. 2003; 23: 7163 -7176. [PubMed] .
- 149. Yang F, Zhang L, Huo XS, Yuan JH, Xu D, Yuan SX, Zhu N, Zhou WP, Yang GS, Wang YZ, Shang JL, Gao CF, Zhang FR, et al. Long noncoding RNA high expression in hepatocellular carcinoma facilitates tumor growth through enhancer of zeste homolog 2 in humans. Hepatol. 2011; 54: 1679 -1689. .
- 150. Matouk IJ, Abbasi I, Hochberg A, Galun E, Dweik H, Akkawi M. Highly upregulated in liver cancer noncoding RNA is overexpressed in hepatic colorectal metastasis. Eur J Gastroen Hepat. 2009; 21: 688 -692. .
- 151. Panzitt K, Tschernatsch MMO, Guelly C, Moustafa T, Stradner M, Strohmaier HM, Buck CR, Denk H, Schroeder R, Trauner M, Zatloukal K. Characterization of HULC, a novel gene with striking up-regulation in hepatocellular carcinoma, as noncoding RNA. Gastroenterol. 2007; 132: 330 -342. .
- 152. Du Y, Kong G, You X, Zhang S, Zhang T, Gao Y, Ye L, Zhang X. Elevation of highly up-regulated in liver cancer (HULC) by hepatitis B virus X protein promotes hepatoma cell proliferation via down-regulating p18. J Biol Chem. 2012; 287: 26302 -26311. [PubMed] .
- 153. Bai F, Pei XH, Pandolfi PP, Xiong Y. p18 Ink4c and Pten constrain a positive regulatory loop between cell growth and cell cycle control. Mol Cell Biol. 2006; 26: 4564 -4576. [PubMed] .
- 154. Ruas M, Gregory F, Jones R, Poolman R, Starborg M, Rowe J, Brookes S, Peters G. CDK4 and CDK6 delay senescence by kinase-dependent and p16INK4a-independent mechanisms. Mol Cell Biol. 2007; 27: 4273 -4282. [PubMed] .
- 155. Wang XS, Zhang Z, Wang HC, Cai JL, Xu QW, Li MQ, Chen YC, Qian XP, Lu TJ, Yu LZ, Zhang Y, Xin DQ, Na YQ, et al. Rapid identification of UCA1 as a very sensitive and specific unique marker for human bladder carcinoma. Clin Cancer Res. 2006; 12: 4851 -4858. [PubMed] .
- 156. Huang J, Zhou N, Watabe K, Lu Z, Wu F, Xu M, Mo YY. Long non-coding RNA UCA1 promotes breast tumor growth by suppression of p27 (Kip1). Cell Death Dis. 2014; 5: e1008 [PubMed] .
- 157. Park SH, Lim JS, Jang KL. All-trans retinoic acid induces cellular senescence via upregulation of p16, p21, and p27. Cancer Lett. 2011; 310: 232 -239. [PubMed] .
- 158. Banerji J, Rusconi S, Schaffner W. Expression of a Beta-Globin Gene Is Enhanced by Remote Sv40 DNA-Sequences. Cell. 1981; 27: 299 -308. [PubMed] .
- 159. Geyer PK, Green MM, Gorces VG. Tissue-specific transcriptional enhancers may act in trans on the gene located in the homologous chromosome: the molecular basis of transvection in Drosophila. EMBO J. 1990; 9: 2247 -2256. [PubMed] .
- 160. Sakabe NJ, Savic D, Nobrega MA. Transcriptional enhancers in development and disease. Genome Biol. 2012; 13: 238 [PubMed] .
- 161. Melo CA, Drost J, Wijchers PJ, van de Werken H, de Wit E, Oude Vrielink JA, Elkon R, Melo SA, Léveillé N, Kalluri R, de Laat W, Agami R. eRNAs are required for p53-dependent enhancer activity and gene transcription. Mol Cell. 2013; 49: 524 -535. [PubMed] .
- 162. Barsyte-Lovejoy D, Lau SK, BoutroS PC, Khosravi F, Jurisica I, Andrulis IL, Tsao MS, Penn LZ. The c-Myc oncogene directly induces the H19 noncoding RNA by allele-specific binding to potentiate tumorigenesis. Cancer Res. 2006; 66: 5330 -5337. [PubMed] .
- 163. Matouk IJ, DeGroot N, Mezan S, Ayesh S, Abu-Iail R, Hochberg A, Galun E. The H19 Non-Coding RNA Is Essential for Human Tumor Growth. PLoS ONE. 2007; 2: 9 e845 [PubMed] .
- 164. Worster DT, Schmelzle T, Solimini NL, Lightcap ES, Millard B, Mills GB, Brugge JS, Albeck JG. Akt and ERK control the proliferative response of mammary epithelial cells to the growth factors IGF-1 and EGF through the cell cycle inhibitor p57Kip2. Sci Signal. 2012; 5: ra19 [PubMed] .
- 165. Joaquin M, Gubern A, Gonzalez-Nunez D, Josue Ruiz E, Ferreiro I, de Nadal E, Nebreda AR, Posas F. The p57 CDKi integrates stress signals into cell-cycle progression to promote cell survival upon stress. EMBO J. 2012; 31: 2952 -2964. [PubMed] .
- 166. Rosenberg E, Demopoulos RI, Zeleniuch-Jacquotte A, Yee H, Sorich J, Speyer JL, Newcomb EW. Expression of cell cycle regulators p57(KIP2), cyclin D1, and cyclin E in epithelial ovarian tumors and survival. Hum Pathol. 2001; 32: 808 -813. [PubMed] .
- 167. Li G, Domenico J, Lucas JJ, Gelf EW. Identification of multiple cell cycle regulatory functions of p57Kip2 in human T lymphocytes. J Immunol. 2004; 173: 2383 -2391. [PubMed] .
- 168. Zhang X, Zhou Y, Mehta KR, Danila DC, Scolavino S, Johnson SR, Klibanski A. A pituitary-derived MEG3 isoform functions as a growth suppressor in tumor cells. J Clin Endocrinol Metab. 2003; 88: 5119 -5126. [PubMed] .
- 169. Zhou Y, Zhang X, Klibanski A. MEG3 noncoding RNA:a tumor suppressor. J Mol Endocrinol. 2012; 48: R45 -53. [PubMed] .
- 170. Qin R, Chen Z, Ding Y, Hao J, Hu J, Guo F. Long non-coding RNA MEG3 inhibits the proliferation of cervical carcinoma cells through the induction of cell cycle arrest and apoptosis. Neoplasma. 2013; 60: 486 -492. [PubMed] .
- 171. Hardy K, Mansfield L, Mackay A, Benvenuti S, Ismail S, Arora P, O'Hare MJ, Jat PS. Transcriptional networks and cellular senescence in human mammary fibroblasts. Mol Biol Cell. 2005; 16: 943 -953. [PubMed] .
- 172. Chen W, Bocker W, Brosius J, Tiedge H. Expression of neu-ral BC200 RNA in human tumours. J Pathol. 1997; 183: 345 -351. [PubMed] .
- 173. Xu D and Tahara H. The role of exosomes and microRNAs in senescence and aging. Adv Drug Deliv Rev. 2013; 65: 368 -375. [PubMed] .
- 174. Salminen A, Kaarniranta K, Kauppinen A. Inflammaging:disturbed interplay between autophagy and inflammasomes. Aging. 2012; 4: 166 -175. [PubMed] .
- 175. Coppe JP, Patil CK, Rodier F, Sun Y, Munoz DP, Goldstein J, Nelson PS, Desprez PY, Campisi J. Senescence-associated secretory phenotypes reveal cell-nonautonomous functions of oncogenic RAS and the p53 tumor suppressor. PLoS Biology. 2008; 6: 2853 -2868. [PubMed] .
- 176. Massone S, Vassallo I, Fiorino G, Castelnuovo M, Barbieri F, Borghi R, Tabaton M, Robello M, Gatta E, Russo C, Florio T, Dieci G, Cancedda R, et al. 17A, a novel non-coding RNA, regulates GABA B alternative splicing and signaling in response to inflammatory stimuli and in Alzheimer disease. Neurobiol Dis. 2011; 41: 308 -317. [PubMed] .
- 177. Alemany R, Perona JS, Sanchez-Dominguez JM, Montero E, Canizares J, Bressani R, Escribá PV, Ruiz-Gutierrez V. G protein-coupled receptor systems and their lipid environment in health disorders during aging. Biochim Biophys Acta. 2007; 1768: 964 -975. [PubMed] .
- 178. Rapicavoli NA, Qu K, Zhang J, Mikhail M, Laberge RM, Chang HY. A mammalian pseudogene lncRNA at the interface of inflammation and anti-inflammatory therapeutics. eLife. 2013; 2: e00762 [PubMed] .
- 179. Ratliff M, Alter S, Frasca D, Blomberg BB, Riley RL. In senescence, age-associated B cells secrete TNFalpha and inhibit survival of B-cell precursors. Aging Cell. 2013; 12: 303 -311. [PubMed] .
- 180. Cui H, Xie N, Tan Z, Banerjee S, Thannickal VJ, Abraham E, Liu G. The human long noncoding RNA, lnc-IL7R, regulates inflammatory response. Eur J Immunol. 2014; 44: 2085 -95. [PubMed] .
- 181. Freund A, Orjalo AV, Desprez PY, Campisi J. Inflammatory networks during cellular senescence:causes and consequences. Trends Mol Med. 2010; 16: 238 -246. [PubMed] .
- 182. Jenny NS. Inflammation in aging:cause, effect, or both? Discovery medicine. 2012; 13: 451 -460. [PubMed] .
- 183. Dunston CR and Griffiths HR. The effect of ageing on macrophage Toll-like receptor-mediated responses in the fight against pathogens. Clin Exp Immunol. 2010; 161: 407 -416. [PubMed] .
- 184. Liu Y, Ferguson JF, Xue C, Ballantyne RL, Silverman IM, Gosai SJ, Serfecz J, Morley MP, Gregory BD, Li M, Reilly MP. Tissue-Specific RNA-Seq in Human Evoked Inflammation Identifies Blood and Adipose LincRNA Signatures of Cardiometabolic Diseases. Arterioscler Thromb Vasc Biol. 2014; 34: 902 -912. [PubMed] .
- 185. Graf K UM. Is Smad3 the key to inflammation and fibrosis in hypertensive heart disease? Hypertension. 2010; 55: 1088 -1089. [PubMed] .
- 186. Zhou Q, Chung AC, Huang XR, Dong Y, Yu X, Lan HY. Identification of novel long noncoding RNAs associated with TGF-beta/Smad3-mediated renal inflammation and fibrosis by RNA sequencing. Amer J Pathol. 2014; 184: 409 -417. [PubMed] .
- 187. Tichauer JE, Flores B, Soler B, Eugenin-von Bernhardi L, Ramirez G, von Bernhardi R. Age-dependent changes on TGFbeta1 Smad3 pathway modify the pattern of microglial cell activation. Brain Behav Immun. 2014; 37: 187 -196. [PubMed] .
- 188. Vijayachandra K, Lee J, Glick AB. Smad3 regulates senescence and malignant conversion in a mouse multistage skin carcinogenesis model. Cancer Res. 2003; 63: 3447 -3452. [PubMed] .
- 189. Dinger ME, Mercer TR, Mattick JS. RNAs as extracellular signaling molecules. J Mol Endocrinol. 2008; 40: 151 -159. [PubMed] .
- 190. Roberts CT and Kurre P. Vesicle Trafficking and RNA Transfer Add Complexity and Connectivity to Cell-Cell Communication. Cancer Res. 2013; 73: 3200 -3205. [PubMed] .
- 191. Valadi H, Ekstrom K, Bossios A, Sjostrand M, Lee JJ, Lotvall JO. Exosome-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells. Nat Cell Biol. 2007; 9: 654 -U672. [PubMed] .
- 192. Kogure T, Yan IK, Lin WL, Patel T. Extracellular Vesicle-Mediated Transfer of a Novel Long Noncoding RNA TUC339: A Mechanism of Intercellular Signaling in Human Hepatocellular Cancer. Genes Cancer. 2013; 4: 261 -272. [PubMed] .
- 193. Braconi C, Valeri N, Kogure T, Gasparini P, Huang N, Nuovo GJ, Terracciano L, Croce CM, Patel T. Expression and functional role of a transcribed noncoding RNA with an ultraconserved element in hepatocellular carcinoma. Proc Natl Acad Sci USA. 2011; 108: 786 -791. [PubMed] .
- 194. Li K, Blum Y, Verma A, Liu Z, Pramanik K, Leigh NR, Chun CZ, Samant GV, Zhao B, Garnaas MK, Horswill MA, Stanhope SA, North PE, Miao RQ, et al. A noncoding antisense RNA in tie-1 locus regulates tie-1 function in vivo. Blood. 2010; 115: 133 -139. [PubMed] .
- 195. Takahashi K, Yan IK, Kogure T, Haga H, Patel T. Extracellular vesicle-mediated transfer of long non-coding RNA ROR modulates chemosensitivity in human hepatocellular cancer. FEBS Open Bio. 2014; 4: 458 -467. .
- 196. Gezer U, Ozgur E, Cetinkaya M, Isin M, Dalay N. Long non-coding RNAs with low expression levels in cells are enriched in secreted exosomes. Cell Biol Intl. 2014; 38: 1076 -1079. .


