Introduction
Acute kidney injury (AKI) is a severe complication in critically ill cirrhotic patients, which occurs in up to 50% of the patients admitted with cirrhosis [1]. The most important causes of AKI are related with the development of severe complications of cirrhosis, such as spontaneous bacterial peritonitis [2], hepatorenal syndrome [3], variceal bleeding [4] and the majority are common reasons for admission to an intensive care unit (ICU). As universally accepted, AKI is a strong predictor for mortality in patients with critically ill cirrhosis [5].
In recent years, two separate bodies developed and published two consensus definitions for AKI: the Acute Dialysis Quality Initiative group for the Risk, Injury, Failure, Loss of Renal Function and End-Stage Renal Disease (RIFLE) criteria; and the Acute Kidney Injury Network (AKIN) group for the AKIN criteria [6,7]. In 2012, the Kidney Disease Improving Global Outcomes (KDIGO) criteria, were created based on the RIFLE and AKIN classifications for prediction of hospital mortality [8].The KDIGO criteria have been validated by many investigations for patients with AKI [9-11]. Until now, there are many established liver-specific and general ICU prognostic models. The Chronic liver Failure-Sequential Organ Failure Assessment (CLIF-SOFA) score is an excellent prognostic evaluation tool derived from the widely used sequential organ failure assessment score (SOFA) score for intensive care unit patients, aiming for a better reflection of the impact of organ failures in the context of cirrhosis [12]. The higher the CLIF-SOFA score, the higher the mortality rate in patients with acute-on-chronic liver failure [13]. Models for End-Stage Liver Disease (MELD) are also widely utilized for evaluating the severity of critically ill cirrhosis [14]. The Simplified acute physiology score (SAPS II) and SOFA score are widely used scoring systems used to assess the prognosis at ICU [15]. However, there is a lack of a specific prognostic score focusing on critically ill cirrhotic patients with AKI.
In this study, the main object was therefore, to develop a new score for critically ill cirrhotic patients with AKI in order to improve the predicting accuracy of the CLIF-SOFA score for hospital mortality. In addition, the study compared the performance of the novel score with CLIF-SOFA, MELD, SAPS II, SOFA and KDIGO.
Results
Baseline characteristics of acute kidney injury in critically ill cirrhotic patients
From June 2001 to October 2012, 527 critically cirrhosis ill patients with AKI met our criteria and were included in our study. The mean age of these patients was 57 years; 362 of the patients were male (68.7%). The in-hospital mortality rates were observed: for 30 days, 45.7%; for 90 days, 56.5%; for 270 days, 63.4% and for 365 days, 64.3%. Table 1 lists the patient demographic data, clinical characteristics laboratory parameters and clinical scores of both survivors and non-survivors. This study revealed that the demographic data were almost similar and the most frequent ethnic group was Caucasians in two groups. Compared with patients in the survival group, non-survivors were slightly older and had significantly higher temperatures, systolic blood pressure (SBP), diastolic blood pressure (DBP), mean arterial pressure (MAP), glucose, potassium, blood urea nitrogen (BUN), partial pressure of oxygen (PaO2), creatinine, lactate, bilirubin and urine output. Moreover, the clinical scores were also significantly different between survival and non-survival group.
Table 1. Characteristics of critically Ill cirrhosis patients with acute kidney disease on the first day of admission, stratified by survival.
| Variable | Survivors (n=188) | Non-survivors (n=339) | P-value | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Demographic parameters | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age, year | 56.1 ± 10.4 | 58.8 ± 12.2 | 0.010 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sex, male no. (%) | 128 (68.1%) | 234 (69.0%) | NS (0.824) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Height, cm | 172.2 ± 9.8 | 171.7 ± 9.9 | NS (0.619) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Weight, kg | 82.3 ± 20.5 | 84.4 ± 21.2 | NS (0.291) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Survival time | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Death time after admission | 365.0 ± 0.0 | 37.23 ± 60.1 | < 0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Ethnicity | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| White no. (%) | 143 (76.1%) | 227 (67.0%) | 0.015 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| African black no. (%) | 14 (7.4%) | 24 (7.4%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Other no. (%) | 31 (16.5%) | 88 (26.0%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Clinical parameters | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Heart rate, n. (%) | 89.9 ± 19.6 | 90.8 ± 19.8 | NS (0.584) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Respiratory rate, | 40.5 ± 33.3 | 40.3 ± 32.9 | NS (0.971) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Temperature, °C | 36.6 ± 0.8 | 36.3 ± 1.1 | 0.003 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SBP, mmHg | 117.0 ± 22.1 | 110.9 ± 21.8 | 0.002 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| DBP, mmHg | 62.6 ± 16.4 | 57.7 ± 14.8 | < 0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| MAP, mmHg | 80.8 ± 16.8 | 75.4 ± 15.1 | < 0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Vasopressin used, n. (%) | 82 (43.6%) | 221 (65.2%) | < 0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Laboratory parameters | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Glucose, mg/dL | 142.3 ± 75.7 | 127.7 ± 57.7 | 0.013 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| White blood cell, 109/L | 11.3 ± 6.7 | 12.0 ± 7.8 | NS (0.282) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Platelet,109/L | 134.9 ± 105.1 | 130.5 ± 92.2 | NS (0.618) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sodium, mEq/L | 134.4 ± 7.3 | 134.3 ± 7.1 | NS (0.801) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Potassium, mEq/L | 4.2 ± 0.9 | 4.4 ± 1.0 | 0.012 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BUN, mg/dL | 39.8 ± 28.0 | 47.1 ± 29.9 | 0.006 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PO2, mmHg | 166.9 ± 123.5 | 127.7 ± 103.8 | < 0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PCO2, mmHg | 37.7 ± 9.9 | 37.4 ± 11.4 | NS (0.754) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| FIO2 | 56.8 ± 33.4 | 66.4 ± 32.1 | 0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Bicarbonate, mEq/L | 21.1 ± 5.1 | 20.7 ± 5.6 | NS (0.339) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Creatinine, mg/dL | 2.0 ± 1.4 | 2.3 ± 1.8 | 0.045 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Creatinine(24h), mg/dL | 1.8 ± 1.2 | 2.4 ± 1.8 | < 0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Lactate, mg/dL | 2.9 ± 2.2 | 3.9 ± 3.3 | 0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| INR | 1.9 ± 0.8 | 2.4 ± 3.6 | 0.053 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Bilirubin, mg/dL | 6.5 ± 9.3 | 9.9 ± 11.2 | < 0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Urine output, ml | 1837.8 ± 2167.5 | 997.2 ± 1583.1 | < 0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Clinical scores | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CLIF-SOFA | 9.7 ± 3.4 | 11.8 ± 3.6 | < 0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| MELD | 21.9 ± 9.3 | 26.3 ± 10.9 | < 0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SAPSII | 43.2 ± 15.0 | 51.3 ± 14.2 | < 0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SOFA | 8.3 ± 3.5 | 10.1 ± 3.6 | < 0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| KDIGO | 2.0 ± 0.9 | 2.5 ± 0.8 | < 0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| AKI-CLIF-SOFA | 1.7 ± 1.1 | 2.6 ± 1.1 | < 0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| NOTE: CLIF-SOFA: chronic liver failure - sequential organ failure assessment score; DBP: diastolic blood pressure; INR: international normalized ratio; MELD: model for end-stage liver disease; AKI-CLIF-SOFA: acute kidney disease-chronic liver failure - sequential organ failure assessment score; SBP: systolic blood pressure; MAP: mean arterial pressure; KDIGO: kidney disease improving global outcomes; SAPSII: simplified acute physiology score; BUN: blood urea nitrogen; PaO2: partial pressure of oxygen; PCO2: partial pressure of carbon dioxide; FIO2: fraction of inspiration O2; N: not significance | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Development and construction of AKI-CLIF-SOFA score
To identify predictors of mortality of critically ill cirrhotic patients with AKI, the AKI-CLIF-SOFA scorings system was developed. Therefore, univariate and multivariate analyses for the study end-points were used to compute clinical and laboratory characteristics at patients’ enrollment (Table 2). Multivariate analysis demonstrated that age (HR 1.02, 95%CI 1.01-1.04), bilirubin (HR 1.03, 95%CI 1.02-1.04), 24h creatinine (HR 1.15, 95%CI 1.08-1.22), lactate (HR 1.10, 95%CI 1.06-1.13), vasopressin used (HR 1.68, 95%CI 1.32-2.13) were identified as independent risk factors for the mortality of these patients. Finally, these five parameters were included in novel scoring system: AKI-CLIF-SOFA. In addition, five optimal cutoff points were selected to distinguish two categorical variables that were directly associated with an increased mortality risk (Table 3). Subgroup analysis demonstrated that patients with age ≥ 64.5y, bilirubin ≥ 5.2 mg/dl, 24h creatinine ≥ 1.45 mg/dl, lactate ≥ 2.55 mg/dl and vasopressin used had a poorer survival probability (Figure 1).
Table 2. Univariate and multivariate analysis of the association between mortality and clinical and laboratory characteristics in patients.
| Univariate analysis | Multivariate analysis | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Variables | HR | 95%CI | P | HR | 95%CI | P | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Age | 1.01 | 1.00-1.02 | 0.008 | 1.02 | 1.01-1.04 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Bilirubin | 1.02 | 1.01-1.03 | <0.001 | 1.03 | 1.02-1.04 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Creatinine (24h) | 1.19 | 1.12-1.26 | <0.001 | 1.15 | 1.08-1.22 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Lactate | 1.10 | 1.06-1.34 | <0.001 | 1.10 | 1.06-1.13 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Vasopressin used* | 1.90 | 1.52-2.38 | <0.001 | 1.68 | 1.32-2.13 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Note: CI: confidence interval; HR: hazard ratio | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| *Dichotomous values | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Table 3. Variables of acute kidney disease-chronic liver failure - sequential organ failure assessment score.
| Variables | 0 | 1 |
| Creatinine (24h) | < 1.45 mg/dL | ≥ 1.45 mg/dL |
| Bilirubin | < 5.20 mg/dL | ≥ 5.20 mg/dL |
| Age | < 64.5 y | ≥ 64.5 y |
| Lactate | < 2.55 mg/dL | ≥ 2.55 mg/dL |
| Vasopressin used | no | yes |
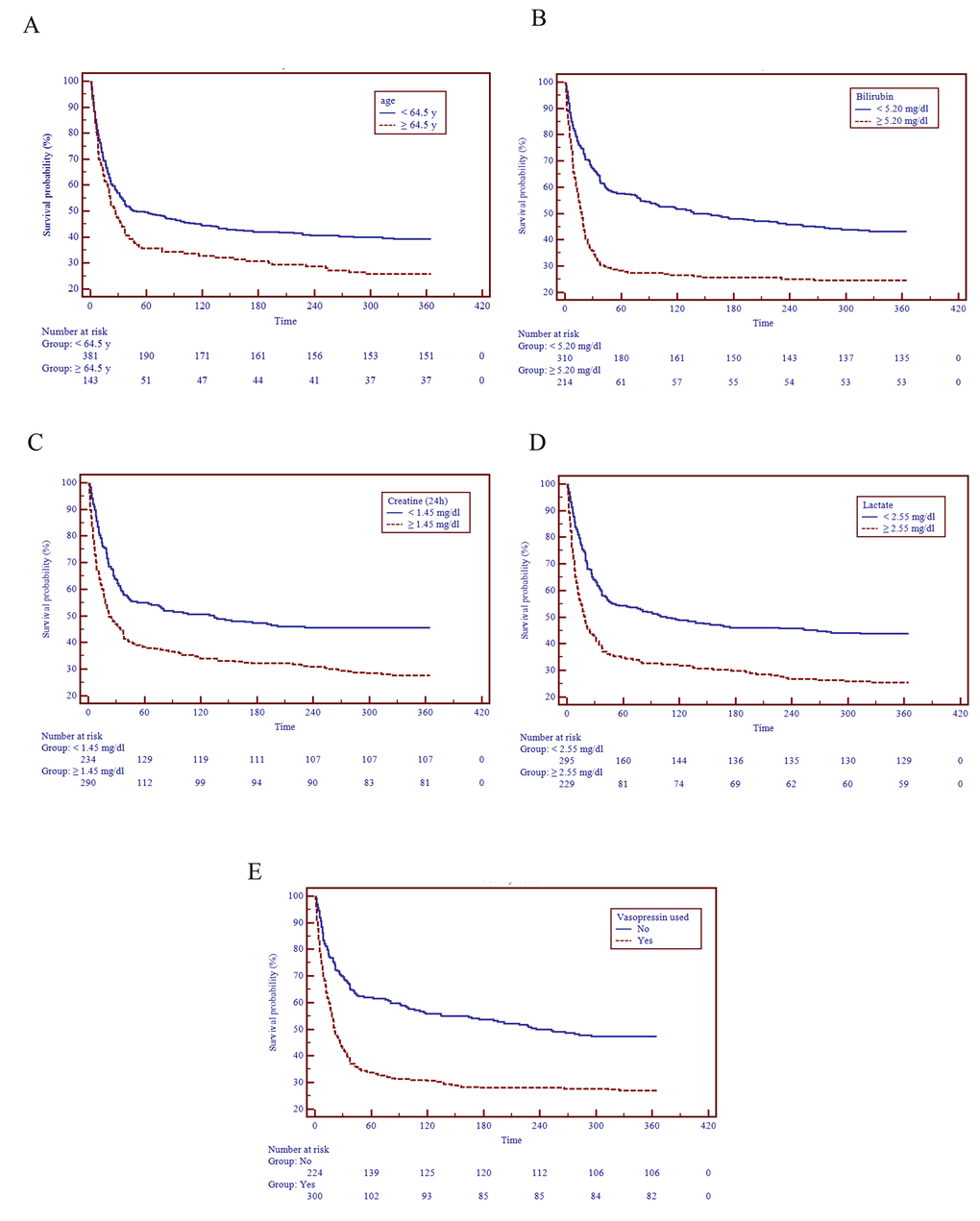
Figure 1. Survival distributions of different risk levels of the AKI-CLIF-SOFA scoring system.
After applying the AKI-CLIF-SOFA score for enrolled subjects, the minimum and maximum values were 0 and 5. In our study, the mean score of non-survivors and survivors were 2.5 ± 0.8, 2.0 ± 0.9, respectively. Moreover, the distribution of the novel score was showed in the Figure 2. Figure 3 demonstrated that a progressive and significant increase in the mortality rate was observed which correlated with the increasing AKI-CLIF-SOFA score.
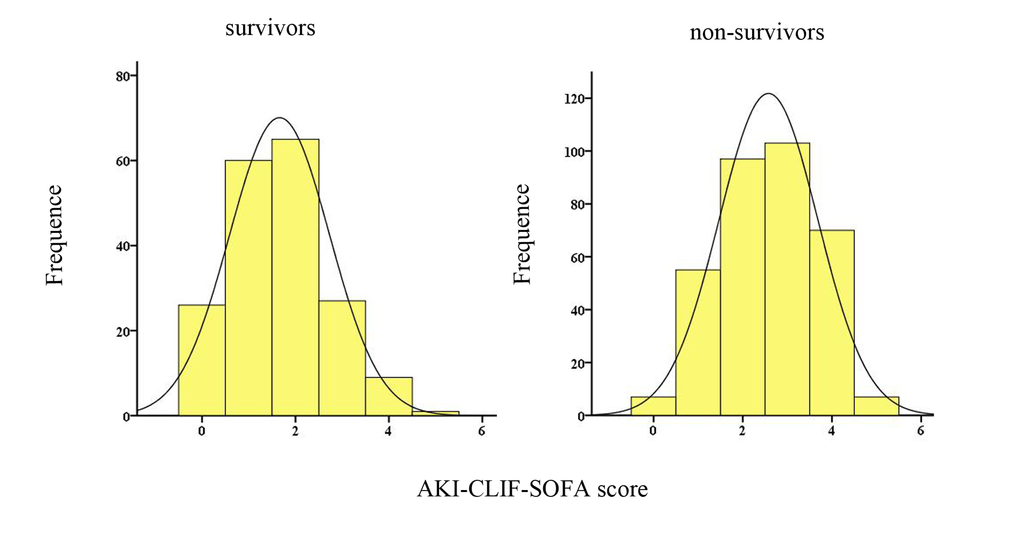
Figure 2. Distribution of the AKI-CLIF-SOFA score among survivors and non-survivors.
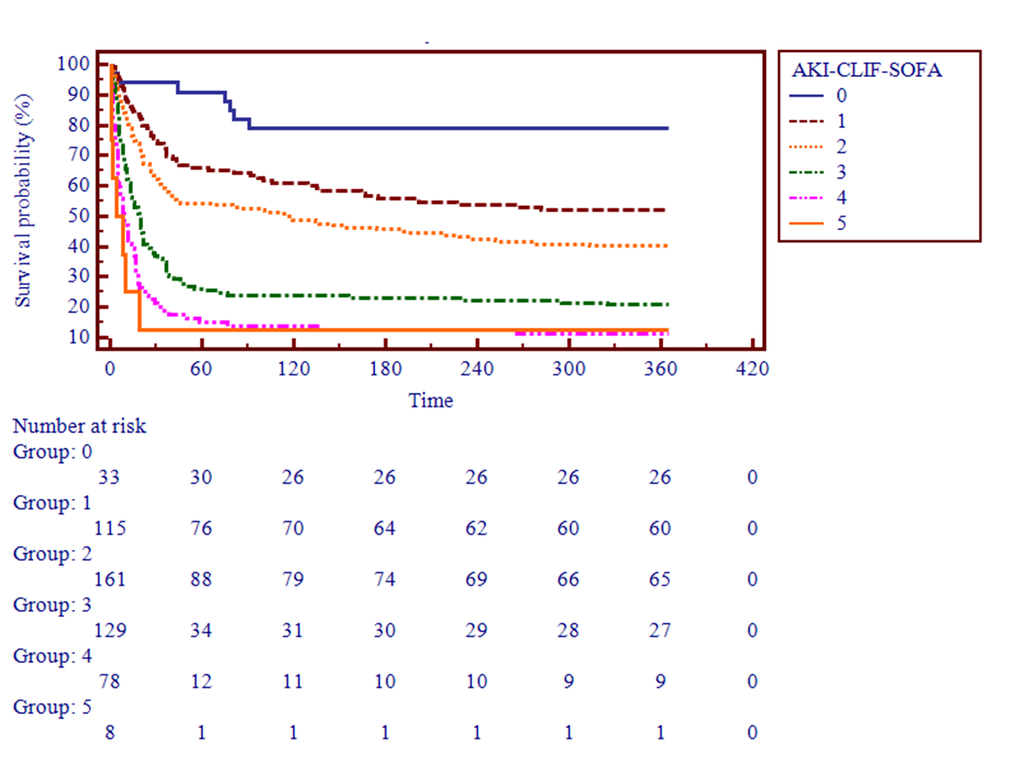
Figure 3. Survival probability based on the AKI-CLIF-SOFA score.
The performance of AKI-CLIF-SOFA to predict the mortality was presented in Table 4 and Figure 4. The the area under the receiver operating characteristic curve (auROC) of new scoring system were 0.74 (95% CI: 0.70-0.78) for 30 days, 0.74 ((95% CI: 0.70-0.78) for 90 days, 0.72 (95% CI: 0.68-0.75) for 270 days and 0.72 (95% CI: 0.68-0.76) for 365 days analysis. Moreover, we used an optimal cutoff point of 2 for the 365-days mortality according to best Youden index. The associated sensitivities and the specificities were 53.1% and 80.32% respectively.
Table 4. Performance of different prognostic models in predicting 365-day mortality using the optimal cut-off point.
| Progostic model | auROC | P-value (v.s. AKI-CLIF-SOFA) | Cut-off | Youden Index | Sensitivity (%) | Specificity (%) | PLR | NLR | PPV | NPV |
| AKI-CLIF-SOFA | 0.72 | - | 2 | 33.42 | 53.10 | 80.32 | 2.70 | 0.58 | 82.9 | 48.7 |
| CLIF-SOFA | 0.66 | 0.015 | 12 | 28.40 | 48.08 | 80.32 | 2.44 | 0.65 | 81.5 | 46.2 |
| MELD | 0.62 | < 0.001 | 22 | 21.99 | 64.01 | 57.98 | 1.52 | 0.62 | 73.3 | 47.2 |
| KDIGO | 0.63 | 0.004 | 2 | 23.93 | 67.55 | 56.38 | 1.55 | 0.58 | 73.6 | 49.1 |
| SAPS II | 0.65 | 0.014 | 34 | 24.79 | 89.68 | 35.11 | 1.38 | 0.29 | 71.4 | 65.3 |
| SOFA | 0.64 | 0.004 | 11 | 21.54 | 39.09 | 82.45 | 2.25 | 0.73 | 80.2 | 43.1 |
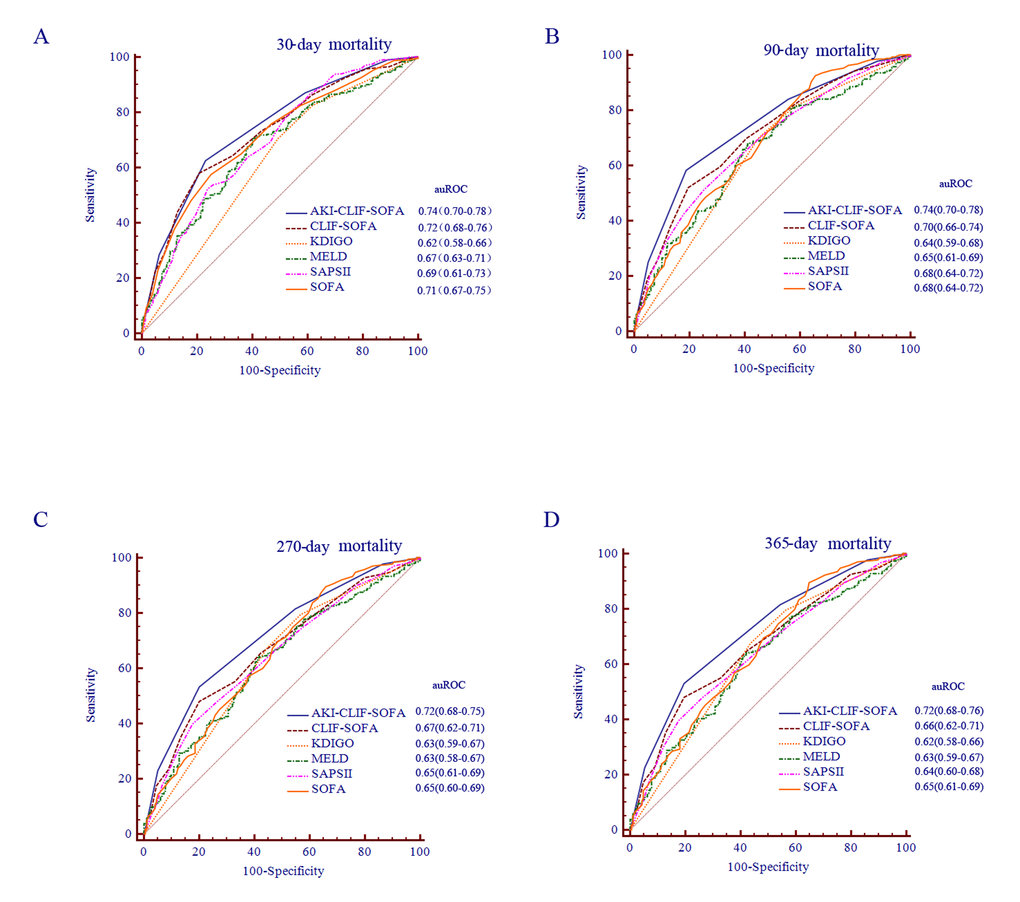
Figure 4. ROC analysis of the prognostic efficiency of AKI-CLFI-SOFA score and other models at different time periods.
Comparison of discrimination for predicting in-hospital mortality according to AKI-CLIF-SOFA, CLIF-SOFA, SOFA, MELD, SAPS II and KDIGO scores
The ability to predict mortality of different scores for critically ill cirrhotic patients with AKI was illustrated by the auROC for the different scores: CLIF-SOFA 0.66, MELD 0.62, SAPS II 0.65, KDIGO 0.63 and SOFA 0.64. The optimal cutoff point according to best Youden index for each score, and corresponding sensitivity, specificity, PPV, NPV, LR+ and LR- are shown in Table 4. Furthermore, the analysis carried out by comparing the auROCs corresponding to AKI-CLIF-SOFA, CLIF-SOFA, MELD, SAPS II, KDIGO and SOFA for 30-days, 90-days, 270-days and 365-days mortality all confirmed the superiority of AKI-CLIF-SOFA and the improvement in predictive ability with respect to the other scores.
Discussion
To our knowledge, many studies investigated the KDIGO criterionin critically ill patients [16-19]. Moreover, the International Club of Ascites (ICA) proposed and modified an adaptation of the KDIGO criteria to define AKI in patients with cirrhosis [20-22]. Nevertheless, only one study had generated a novel prognostic scoring system for renal-specific scores from critically ill patients with cirrhosis and this have not been widely endorsed [23]. The new score (MBRS score: MAP + bilirubin + respiratory failure + sepsis) was derived from 111 cirrhotic patients with acute renal failure. However, in this study, the definition of acute kidney failure was based on the RIFLE classification and the modification of diet in renal disease (MDRD) formula was applied to estimate baseline serum creatinine (SCr) concentrations. As we know, the MDRD formula is inaccurate in the estimation of estimation of glomerular filtration rate (GFR) in cirrhotic patients. In addition, the new score guaranteed a strait forward use and provided prognostic information using variables that are easily available for clinicians who first encountered with the patients on the day of admission to the hospital.
The study, which included 527 critically ill cirrhotic patients with AKI, demonstrated that an increasing trend of hospital mortality with progression of the new AKI-CLIF-SOFA score. The score performed better than the established and commonly used acute physiology, renal-specific and liver-specific scores in our cohort.
In the current AKI-CLIF-SOFA score, parameters consist of 24h creatinine, bilirubin, age, lactate and vasopressin used (0 or 1 for each variable, range 0-5 points). It was important to note that bilirubin and creatinine played an important role in predicting mortality as was also observed in the CLIF-SOFA score. As is generally accepted, creatinine is the most practical biomarker of renal function in patients. Furthermore, after adjusting for other parameters, the prognosis of creatinine at 24 hours after admission was superior to creatinine on admission in analysis. Elevated bilirubin concentration has been shown to be associated with mortality of patients with liver-disease. Age, lactate, and vasopressin used added discriminative power as organ function predictors, explaining the higher accuracy of AKI-CLIF-SOFA with respect to other renal scores. Vasopressin used is also an important indicator of systemic derangements related to circulatory failure [24]. High lactate levels are also considered essential related with aggravating events, such as sepsis, respiratory, or cardiac failure [25,26].
To compare the performance of this score against the current gold standards, KDIGO, CLIF-SOFA, MELD, SOFA and SAPSII score, we used the auROC analysis. In several recent studies, CLIF-SOFA demonstrated a favorable performance and proved to be a strong predictor for mortality in patients with cirrhosis [27-29]. Nevertheless, the CLIF-SOFA score had an inappropriate discriminatory power for predicting in-hospital mortality in these special patients (auROC = 0.66). The auROC analysis clearly showed that AKI-CLIF-SOFA was significantly more accurate in predicting 30-days, 90-days, 270-days and 365-days mortality than other scores. Therefore, the new scoring system provided a better discriminative ability than other liver-specific and kidney-specific scores. Moreover, the Kaplan-Meier survival curve showed that higher AKI-CLIF-SOFA score groups were associated with a higher risk for hospital mortality. However, validation of our model should be undertaken to confirm its clinical utility.
A few potential limitations need consideration. Firstly, because our study population was exclusively included from a single center, a potential selection bias might exist which limits the generalization of our findings. Secondly, sequential measurement of these scoring systems may reflect the dynamic aspects of clinical diseases, thus providing superior information on mortality risk. Thirdly, multi-center large-scale studies at more than 365-days follow-up are needed to further verify its prognosis of our new scoring system. Fourth, in order to use this scoring system as a model, predictive analytics request a validation of the model in order the model is suited for other patients then those included in this manuscript. Therefore cross-validation or bootstrap validation could be useful.
In conclusion, this is the largest study to evaluate prognostic scoring system for critically ill cirrhotic patients with AKI. The AKI-CLIF-SOFA scoring system has the best discriminatory power for predicting in-hospital mortality in study cohorts and may be an optimal scoring system for critically ill cirrhosis with AKI. Further research is needed to clarify the validity of AKI-CLIF-SOFA score.
Materials and Methods
The database
The Multi-parameter Intelligent Monitoring in Intensive Care III version 3.0 (MIMIC-III v3.0) database is a large, single-center database comprising information relating to patients admitted to critical care units [30, 31]. The database included general information (patient demographics, hospital admission and discharge dates), vital signs, medication, laboratory tests, fluid balance and notes and reports. The establishment of the database was approved by the Institutional Review Boards of the Beth Israel Deaconess Medical Center (Boston, MA) and the Massachusetts Institute of Technology (Cambrige, MA, USA). Currently, the database consisted of more than 40,000 ICU patients admitted to Beth Israel Deaconess Medical Center from June 2001 to October 2012. Our permission to access the database was approved after competition of the NIH web based training course named “Protecting Human Research Participants” (Our certification number: 1605699).
In this study, we included 527 consecutive patients with cirrhosis admitted to ICU, complicated with AKI and were followed-up for 365-days. Reasons for exclusion were: pediatric patients (age 18 years or below), patients admitted to the hospital for < 24h, patients with previous end-stage renal disease and received regular RRT, and patients with a history of liver transplantation.
Definition
Liver cirrhosis was defined when at least two of the following criteria were satisfied: 1) ultrasonographic evidence of a small-sized liver with and without splenomegaly/ascites; 2) hypoalbuminemia (serum albumin < 35 g/L); 3) aminotransferase to platelet ratio (× 109/L) × 100 > 2. Alcoholic cirrhosis of the liver was considered with a daily alcohol consumption of more than 80 g/day for at least five years.
The occurrence of AKI was determined based on the KDIGO classification. The definition is that SCr changes ≥ 1.5* baseline to have occurred within the prior 7 days or a 0.3 mg/d increase in SCr must occur within a 48 hours period or Urine output < 0.5 ml/kg/h * 6 hours.
1) Stage 1: Increase in SCr ≥ 1.5* baseline or of 0.3 mg/dl or Urine output < 0.5 ml/kg/h * 6 hours
2) Stage 2: Increase in SCr ≥ 2.0* baseline or Urine output < 0.5 ml/kg/h * 12 hours
3) Stage 3: Increase in SCr ≥ 3.0* baseline or increase in serum creatinine to ≥ 4.0 mg/dl or initiation of RRT or Urine output < 0.5 ml/kg/h * 12 hours
For patients without an available SCr value prior to hospitalization, we followed the recommendations of ICA and used the first SCr value measured during hospitalization as the baseline SCr [32]. Urine output was observed for the first 24h after ICU admission and was corrected for body weight.
Data collection
Our investigators extracted demographic parameters, survival time, clinical parameters and laboratory parameters. The clinical parameters, which included heart rate, respiration, temperature, SBP, DBP, and MAP, were derived by ICU nurses from the hospital's on-line information systems. The laboratory parameters from routine tests on admission, including glucose, white blood cell, platelet, sodium, potassium, BUN, PaO2, partial pressure of carbon dioxide (PCO2), fraction of inspiration O2 (FIO2), bicarbonate, lactate, international normalized ratio (INR) and bilirubin were organized into a relational database. Additionally, the urine output was measured for the first 24h after ICU admission and recorded at least 6h. SCr was measured when clinically needed, at least once in 24 hours. The other data included age, sex, height, weight, ethnicity, vasopressin used, renal replacement therapy (RRT) used and survival time. Mortality data were censored after hospital discharge and were obtained by Social Security Death Records from the United States government. For all patients the CLIF-SOFA, MELD, SAPS II, KDIGO and SOFA were calculated. The start date was the date of patient’s admission and the primary end points were defined at 30-days, 90-days, 270-days and 365-days for all-cause mortality.
Data extraction was performed using Oracle SQL Developer version 3.0 (Oracle Corporation, Redwood Shores, CA). Because this study is retrospective, no ethical approval was required for these analyses of non-patient identifiable and anonymous data.
Statistical analysis
Data were presented as mean and standard derivations for continuous and normally distributed variables, or frequencies (percentage) for categorical variables. The Kolmogorov–Smirnov test was calculated for assessing the distribution of the variables. For comparisons, the Student’s t -test and the Mann-Whitney test was used for continuous baseline characteristics of the each group for continuous variables with or without normal distribution, respectively. The Chi-square test was performed for categorical variables. Cox regression was used for univariate and multivariate analysis. Survival curves were constructed based on Kaplan-Meier estimates and comparisons were performed using the log-rank test.
Discrimination was examined using auROC. All the patients were enrolled for a comparison of the discriminative value of the CLIF-SOFA, MELD, SAPS II, KDIGO and SOFA scores, as well as for the AKI-CLIF-SOFA score for predicting mortality risks at 30-days, 90-days, 270-days and 365-days. The optimal cut-off point was identified based on the maximal Youden index (sensitivity + specificity − 1). In addition, the corresponding sensitivity, specificity, positive likelihood ratio (PLR), negative likelihood ratio (NLR), positive predictive value (PPV), negative predictive value (NPV) were calculated according to the auROC results. Statistical analyses were performed using SPSS version 18.0 software (IBM, Armonk, NY), MedCalc version 12.7 (MedCalc Software, Ostend, Belgium).
Author Contributions
Sun DQ, Zheng CF and Zheng MH designed the study and prepared figures, Liu WY collected data, Mao Z, Shi KQ and Wang XD did the statistical analyses, Sun DQ, Wang JD, Poucke SV and Zheng MH reviewed the results, interpreted data, and wrote the manuscript. All authors have made an intellectual contribution to the manuscript and approved the submission.
Conflicts of Interest
The authors have no conflict of interests to declare.
Funding
This work was supported by grants from the National Natural Science Foundation of China (81500665), Scientific Research Foundation of Wenzhou (Y20160223), High Level Creative Talents from Department of Public Health in Zhejiang Province, and Project of New Century 551 Talent Nurturing in Wenzhou. No writing assistance was obtained in this manuscript.
References
- 1. Moreau R, Lebrec D. Diagnosis and treatment of acute renal failure in patients with cirrhosis. Best Pract Res Clin Gastroenterol. 2007; 21:111–23. https://doi.org/10.1016/j.bpg.2006.10.004 [PubMed]
- 2. Fernández J, Navasa M, Planas R, Montoliu S, Monfort D, Soriano G, Vila C, Pardo A, Quintero E, Vargas V, Such J, Ginès P, Arroyo V. Primary prophylaxis of spontaneous bacterial peritonitis delays hepatorenal syndrome and improves survival in cirrhosis. Gastroenterology. 2007; 133:818–24. https://doi.org/10.1053/j.gastro.2007.06.065 [PubMed]
- 3. Ginès P, Guevara M, Arroyo V, Rodés J. Hepatorenal syndrome. Lancet. 2003; 362:1819–27. https://doi.org/10.1016/S0140-6736(03)14903-3 [PubMed]
- 4. Cárdenas A, Ginès P, Uriz J, Bessa X, Salmerón JM, Mas A, Ortega R, Calahorra B, De Las Heras D, Bosch J, Arroyo V, Rodés J. Renal failure after upper gastrointestinal bleeding in cirrhosis: incidence, clinical course, predictive factors, and short-term prognosis. Hepatology. 2001; 34:671–76. https://doi.org/10.1053/jhep.2001.27830 [PubMed]
- 5. Belcher JM, Garcia-Tsao G, Sanyal AJ, Bhogal H, Lim JK, Ansari N, Coca SG, Parikh CR, and TRIBE-AKI Consortium. Association of AKI with mortality and complications in hospitalized patients with cirrhosis. Hepatology. 2013; 57:753–62. https://doi.org/10.1002/hep.25735 [PubMed]
- 6. Bellomo R, Ronco C, Kellum JA, Mehta RL, Palevsky P, and Acute Dialysis Quality Initiative workgroup. Acute renal failure - definition, outcome measures, animal models, fluid therapy and information technology needs: the Second International Consensus Conference of the Acute Dialysis Quality Initiative (ADQI) Group. Crit Care. 2004; 8:R204–12. https://doi.org/10.1186/cc2872 [PubMed]
- 7. Mehta RL, Kellum JA, Shah SV, Molitoris BA, Ronco C, Warnock DG, Levin A, and Acute Kidney Injury Network. Acute Kidney Injury Network: report of an initiative to improve outcomes in acute kidney injury. Crit Care. 2007; 11:R31. https://doi.org/10.1186/cc5713 [PubMed]
- 8. Palevsky PM, Liu KD, Brophy PD, Chawla LS, Parikh CR, Thakar CV, Tolwani AJ, Waikar SS, Weisbord SD. KDOQI US commentary on the 2012 KDIGO clinical practice guideline for acute kidney injury. Am J Kidney Dis. 2013; 61:649–72. https://doi.org/10.1053/j.ajkd.2013.02.349 [PubMed]
- 9. Fujii T, Uchino S, Takinami M, Bellomo R. Validation of the Kidney Disease Improving Global Outcomes criteria for AKI and comparison of three criteria in hospitalized patients. Clin J Am Soc Nephrol. 2014; 9:848–54. https://doi.org/10.2215/CJN.09530913 [PubMed]
- 10. Zeng X, McMahon GM, Brunelli SM, Bates DW, Waikar SS. Incidence, outcomes, and comparisons across definitions of AKI in hospitalized individuals. Clin J Am Soc Nephrol. 2014; 9:12–20. https://doi.org/10.2215/CJN.02730313 [PubMed]
- 11. Machado MN, Nakazone MA, Maia LN. Acute kidney injury based on KDIGO (Kidney Disease Improving Global Outcomes) criteria in patients with elevated baseline serum creatinine undergoing cardiac surgery. Rev Bras Cir Cardiovasc. 2014; 29:299–307. [PubMed]
- 12. Pan HC, Jenq CC, Tsai MH, Fan PC, Chang CH, Chang MY, Tian YC, Hung CC, Fang JT, Yang CW, Chen YC. Scoring systems for 6-month mortality in critically ill cirrhotic patients: a prospective analysis of chronic liver failure - sequential organ failure assessment score (CLIF-SOFA). Aliment Pharmacol Ther. 2014; 40:1056–65. https://doi.org/10.1111/apt.12953 [PubMed]
- 13. McPhail MJ, Shawcross DL, Abeles RD, Chang A, Patel V, Lee GH, Abdulla M, Sizer E, Willars C, Auzinger G, Bernal W, Wendon JA. Increased Survival for Patients With Cirrhosis and Organ Failure in Liver Intensive Care and Validation of the Chronic Liver Failure-Sequential Organ Failure Scoring System. Clin Gastroenterol Hepatol. 2015; 13:1353–1360.e8. https://doi.org/10.1016/j.cgh.2014.08.041 [PubMed]
- 14. Biggins SW, Kim WR, Terrault NA, Saab S, Balan V, Schiano T, Benson J, Therneau T, Kremers W, Wiesner R, Kamath P, Klintmalm G. Evidence-based incorporation of serum sodium concentration into MELD. Gastroenterology. 2006; 130:1652–60. https://doi.org/10.1053/j.gastro.2006.02.010 [PubMed]
- 15. Le Gall JR, Lemeshow S, Saulnier F. A new Simplified Acute Physiology Score (SAPS II) based on a European/North American multicenter study. JAMA. 1993; 270:2957–63. https://doi.org/10.1001/jama.1993.03510240069035 [PubMed]
- 16. Izawa J, Uchino S, Takinami M. A detailed evaluation of the new acute kidney injury criteria by KDIGO in critically ill patients. J Anesth. 2016; 30:215–22. https://doi.org/10.1007/s00540-015-2109-6 [PubMed]
- 17. Sutherland SM, Byrnes JJ, Kothari M, Longhurst CA, Dutta S, Garcia P, Goldstein SL. AKI in hospitalized children: comparing the pRIFLE, AKIN, and KDIGO definitions. Clin J Am Soc Nephrol. 2015; 10:554–61. https://doi.org/10.2215/CJN.01900214 [PubMed]
- 18. Thomas ME, Blaine C, Dawnay A, Devonald MA, Ftouh S, Laing C, Latchem S, Lewington A, Milford DV, Ostermann M. The definition of acute kidney injury and its use in practice. Kidney Int. 2015; 87:62–73. https://doi.org/10.1038/ki.2014.328 [PubMed]
- 19. Pan HC, Chien YS, Jenq CC, Tsai MH, Fan PC, Chang CH, Chang MY, Tian YC, Fang JT, Yang CW, Chen YC. Acute Kidney Injury Classification for Critically Ill Cirrhotic Patients: A Comparison of the KDIGO, AKIN, and RIFLE Classifications. Sci Rep. 2016; 6:23022. https://doi.org/10.1038/srep23022 [PubMed]
- 20. Nadim MK, Durand F, Kellum JA, Levitsky J, O’Leary JG, Karvellas CJ, Bajaj JS, Davenport A, Jalan R, Angeli P, Caldwell SH, Fernández J, Francoz C, et al. Management of the critically ill patient with cirrhosis: A multidisciplinary perspective. J Hepatol. 2016; 64:717–35. https://doi.org/10.1016/j.jhep.2015.10.019 [PubMed]
- 21. Nadim MK, Kellum JA, Davenport A, Wong F, Davis C, Pannu N, Tolwani A, Bellomo R, Genyk YS, and ADQI Workgroup. Hepatorenal syndrome: the 8th International Consensus Conference of the Acute Dialysis Quality Initiative (ADQI) Group. Crit Care. 2012; 16:R23. https://doi.org/10.1186/cc11188 [PubMed]
- 22. Piano S, Rosi S, Maresio G, Fasolato S, Cavallin M, Romano A, Morando F, Gola E, Frigo AC, Gatta A, Angeli P. Evaluation of the Acute Kidney Injury Network criteria in hospitalized patients with cirrhosis and ascites. J Hepatol. 2013; 59:482–89. https://doi.org/10.1016/j.jhep.2013.03.039 [PubMed]
- 23. Fang JT, Tsai MH, Tian YC, Jenq CC, Lin CY, Chen YC, Lien JM, Chen PC, Yang CW. Outcome predictors and new score of critically ill cirrhotic patients with acute renal failure. Nephrol Dial Transplant. 2008; 23:1961–69. https://doi.org/10.1093/ndt/gfm914 [PubMed]
- 24. Aggarwal A, Ong JP, Younossi ZM, Nelson DR, Hoffman-Hogg L, Arroliga AC. Predictors of mortality and resource utilization in cirrhotic patients admitted to the medical ICU. Chest. 2001; 119:1489–97. https://doi.org/10.1378/chest.119.5.1489 [PubMed]
- 25. Zhang Z, Xu X. Lactate clearance is a useful biomarker for the prediction of all-cause mortality in critically ill patients: a systematic review and meta-analysis*. Crit Care Med. 2014; 42:2118–25. https://doi.org/10.1097/CCM.0000000000000405 [PubMed]
- 26. Okorie ON, Dellinger P. Lactate: biomarker and potential therapeutic target. Crit Care Clin. 2011; 27:299–326. https://doi.org/10.1016/j.ccc.2010.12.013 [PubMed]
- 27. Bao Q, Wang B, Yu L, Weng H, Ge J, Li L. A modified prognostic score for critically ill patients with cirrhosis: an observational study. J Gastroenterol Hepatol. 2016; 31:450–58. https://doi.org/10.1111/jgh.13076 [PubMed]
- 28. Silva PE, Fayad L, Lazzarotto C, Ronsoni MF, Bazzo ML, Colombo BS, Dantas-Correa EB, Narciso-Schiavon JL, Schiavon LL. Single-centre validation of the EASL-CLIF consortium definition of acute-on-chronic liver failure and CLIF-SOFA for prediction of mortality in cirrhosis. Liver Int. 2015; 35:1516–23. https://doi.org/10.1111/liv.12597 [PubMed]
- 29. Rodríguez E, Elia C, Solà E, Barreto R, Graupera I, Andrealli A, Pereira G, Poca M, Sánchez J, Guevara M, Soriano G, Alessandria C, Fernández J, et al. Terlipressin and albumin for type-1 hepatorenal syndrome associated with sepsis. J Hepatol. 2014; 60:955–61. https://doi.org/10.1016/j.jhep.2013.12.032 [PubMed]
- 30. Johnson AE, Pollard TJ, Shen L, Lehman LW, Feng M, Ghassemi M, Moody B, Szolovits P, Celi LA, Mark RG. MIMIC-III, a freely accessible critical care database. Sci Data. 2016; 3:160035. https://doi.org/10.1038/sdata.2016.35 [PubMed]
- 31. Francoz C, Prié D, Abdelrazek W, Moreau R, Mandot A, Belghiti J, Valla D, Durand F. Inaccuracies of creatinine and creatinine-based equations in candidates for liver transplantation with low creatinine: impact on the model for end-stage liver disease score. Liver Transpl. 2010; 16:1169–77. https://doi.org/10.1002/lt.22128 [PubMed]
- 32. Angeli P, Ginès P, Wong F, Bernardi M, Boyer TD, Gerbes A, Moreau R, Jalan R, Sarin SK, Piano S, Moore K, Lee SS, Durand F, et al. Diagnosis and management of acute kidney injury in patients with cirrhosis: revised consensus recommendations of the International Club of Ascites. J Hepatol. 2015; 62:968–74. https://doi.org/10.1016/j.jhep.2014.12.029 [PubMed]


