Introduction
Age-related declines in physical function and subsequent disability are major contributors to morbidity and mortality in older adults [1,2]. Among those physical function measurements, gait speed has been reported to be a useful predictor of adverse outcomes, including incident mobility disability and mortality in older adults [3-6]. A recent study further supports that walking at a maximum pace might be useful for estimating subjective general health and skeletal muscle mass [7]. Although aerobic exercise training (AEX) is generally effective for improving physical function in older adults, there exists inter-individual variation in responses to standardized AEX interventions [8,9]. We previously demonstrated an overall benefit of AEX on improvements in fast-paced gait speed over a distance of 400 meters [10,11]. However, changes in fast-paced gait speed with AEX were highly variable, ranging from a decline of 0.13 m/s to an increase of 0.44 m/s [10]. Results from a previous study further indicate that 0.05 m/s is a small meaningful change in gait speed, while 0.10 m/s is a substantial change in gait speed in older adults, and importantly, small changes in gait speed are detectable in research and clinical settings [12]. Thus, understanding what physiologic factors contribute to gait speed changes with exercise could be important in tailoring successful interventions to prevent disability. Ideally, identification of novel biomarkers that predict gait speed responses to exercise training will be crucial for clinicians to select intervention regimens in a personalized manner.
Over the last decade, short non-coding microRNAs (miRNAs) have emerged as important regulators in multiple biological processes. These miRNAs are usually about 22 nucleotides long and regulate protein abundance by inhibiting protein translation or enhancing mRNA degradation [13]. miRNAs are involved in epigenetic control of muscle function, including proliferation, differentiation, hypertrophy, and contraction [14-19]. Although some miRNAs are tissue-specific, many can be detected in blood, where they are highly stable and resistant to endogenous RNAses [20-23]. Thus, circulating miRNAs are considered useful disease-specific diagnostic biomarkers and promising therapeutic targets [24-27].
Although recent studies demonstrate that circulating miRNAs are associated with aerobic capacity, muscle mass, and muscle strength [28-30], their relationship with physical function and disability is largely unknown. miRNA expression is altered in response to acute, as well as chronic exercise, which may help to elucidate molecular mechanisms that underlie cardiovascular and muscular adaptations [31-33]. Other studies have shown that miRNAs may function as paracrine/endocrine mediators [34], which could be regulated with endurance exercise, indicating a potential role of miRNAs in regulating responses to an exercise intervention [35-37]. However, to our knowledge, the effects of AEX on global miRNA expression profiles in older adults, and the relationship of individual variation in gait speed responses to miRNAs, have not been studied. The present study had two goals: (1) to examine changes in miRNA expression in response to an AEX intervention and (2) to identify circulating miRNA biomarkers related to individual changes in gait speed.
Results
High-throughput screening of circulating miRNAs in obese older adults by Nanostring nCounter analysis
Among the 800 miRNAs detected, 120 miRNAs were abundantly expressed in plasma (Figure 1). Among them, only 14 were found to have counts above 500, whereas the majority were between 50 and 500. Details of each miRNA are listed in Supplementary Table S1. In addition, circulating levels of 4 miRNAs were significantly changed after AEX (pre vs. post): miR-376a-5p (3.71 ± 2.92 vs. 4.98 ± 2.26, p = 0.004) increased, while miR-16-5p (9.91 ± 0.86 vs. 9.60 ± 0.82, p = 0.039), miR-27a-3p (4.06 ± 2.54 vs. 2.74 ± 2.88, p = 0.022), and miR-28-3p (2.65 ± 2.79 vs. 1.63 ± 2.52, p = 0.043) all decreased.
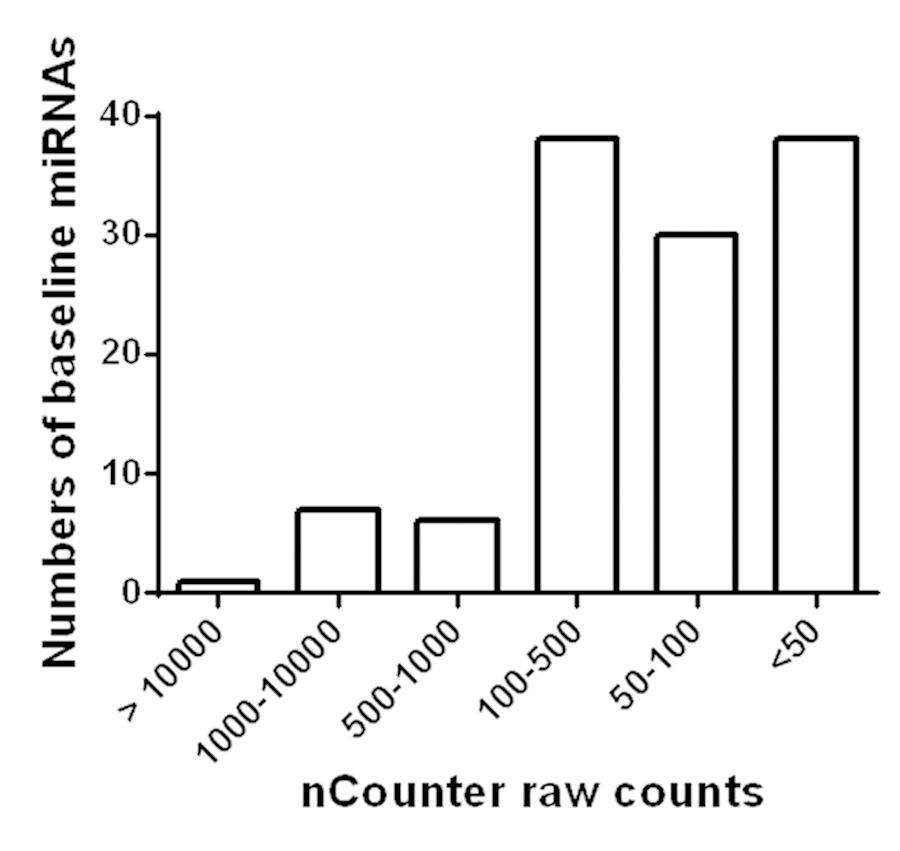
Figure 1. Detection of miRNAs in circulation of obese older adults. Based on Nanostring nCounter analysis raw counts, 120 miRNAs were found to be abundantly present in circulation of obese older adults.
Association between gait speed changes and circulating miRNA levels
None of the 4 miRNAs that were altered with AEX (miR-376a-5p, miR-16-5p, miR-27a-3p, and miR-28-3p) were associated with gait speed changes in response to AEX (data not shown); thus, we analyzed whether other abundant circulating miRNAs were associated with gait speed changes. We found that changes in gait speed were negatively associated with baseline levels of miR-181a-5p, but not with percent changes in miR-181a-5p with AEX (Figure 2). In addition, changes in gait speed showed a trend for a positive correlation with baseline levels of miR-92a-3p, but negatively correlated with percent changes in miR-92a-3p levels (Figure 3). These associations persisted or became stronger after adjustment for age and gender. Since gait speed changes were not associated with changes in body mass (data not shown), we did not include change in body mass in the model.
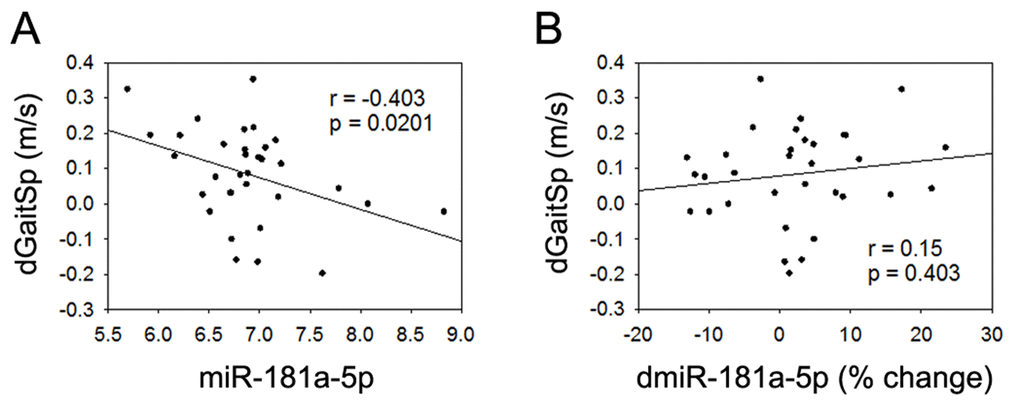
Figure 2. miR-181a-5p and gait speed changes. (A) Association between changes in gait speed (dGaitSp) and baseline levels of miR-181a-5p; (B) Association between changes in gait speed (dGaitSp) and percent changes in miR-181a-5p (dmiR-181a-5p (% change)).
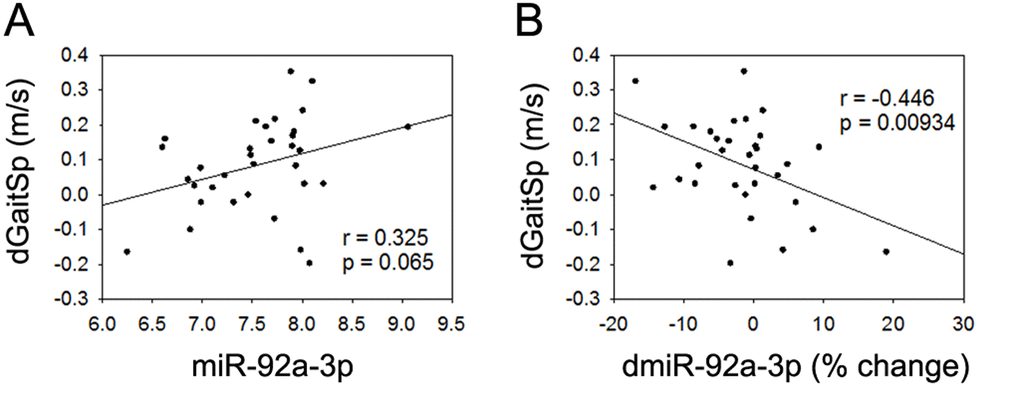
Figure 3. miR-92a-3p and gait speed changes. (A) Association between changes in gait speed (dGaitSp) and baseline levels of miR-92a-3p; (B) Association between changes in gait speed (dGaitSp) and percent changes in miR-92a-3p (dmiR-92a-3p (% change)).
Combined miR-181a-5p and miR-92a-3p baseline levels were strongly associated with gait speed changes with AEX
Given the detected associations or trend of associations between gait speed changes and baseline miR-181a-5p or miR-92a-3p, we next tested if the linear regression combined baseline miR-181a-5p and miR-92a-3p could yield an even stronger association with gait speed changes. Based on a linear combination of the baseline levels of the 2 miRNAs weighted by their regression coefficients as others have used [38], we used a linear combination of miR-181a-5p and miR-92a-3p (-0.208+0.08(miR-181a-5p)-0.057(miR-92a-3p)), and found that the combined baseline level of these miRNAs had an even stronger negative association with gait speed changes than either miRNA alone (Figure 4), even after adjustment for age and gender (p=0.004). This finding implies that combined baseline levels of miR-181a-5p and miR-92a-3p could be useful biomarkers for prediction of gait speed responses to AEX in obese older adults.
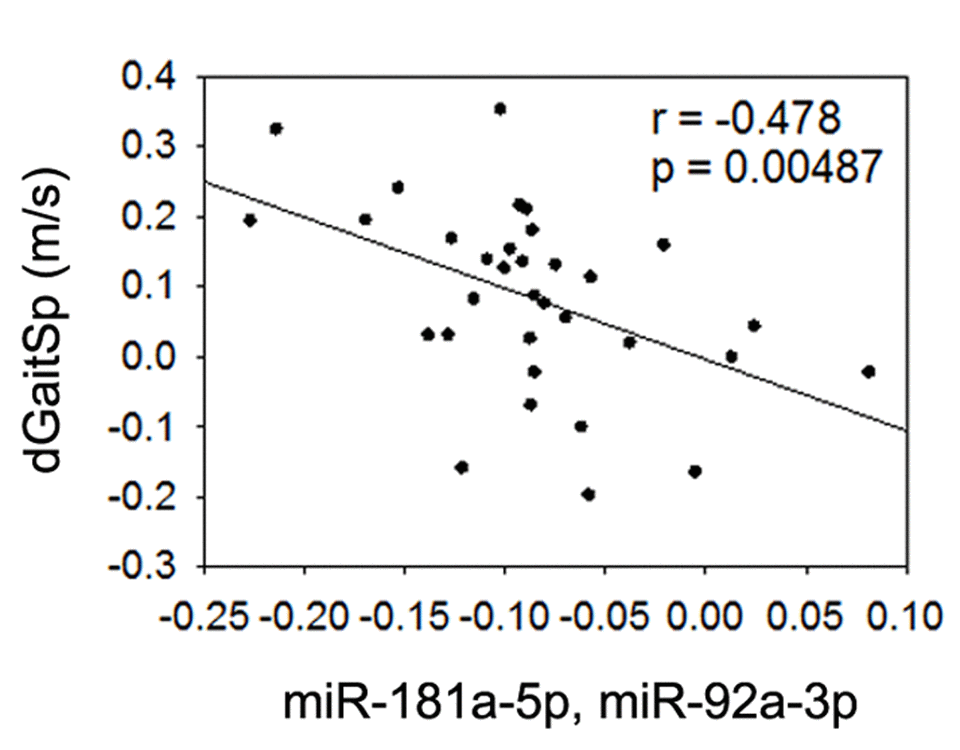
Figure 4. Combined miR-181a-5p and miR-92a-3p and gait speed changes. Association between changes in gait speed (dGaitSp) and combined baseline levels of miR-181a-5p and miR-92a-3p.
Baseline level adjusted gait speed changes show similar trends of association with miRNAs as those observed between absolute gait speed changes and miRNAs
The above determined associations were based on absolute gait speed changes (dGaitSp (m/s)) after AEX. Given that the initial baseline gait speed varied among individuals (Table 1) and may affect the individual gait speed response to AEX, we further analyzed the association between the identified miRNAs and baseline level-adjusted gait speed changes: dGaitSp (% change) = 100 × (GaitSpPost-AEX – GaitSpPre-AEX)/ GaitSpPre-AEX. As shown in Table 2, dGaitSp (% change) shows similar trends of association with miRNAs as that of dGaitSp (m/s). Notably, both dGaitSp (% change) and dGaitSp (m/s) were significantly associated with this linear regression combined variable using baseline levels of miR-181a-5p and miR-92a-3p. This finding confirmed that combined baseline levels of miR-181a-5p and miR-92a-3p are useful for prediction of gait speed responses to AEX in obese older adults, which is independent of the baseline level gait speed, as well as age and gender.
Table 1. Participant characteristics at baseline.
| Overall (n=33) | Range | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age (years) | 69.3 ± 3.6 | 65 ̶ 79 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Gender (M/F) | 24/9 | N/A | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Race (White/Non-White) | 28/5 | N/A | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BMI (kg/m2) | 34.0 ± 3.1 | 30 ̶ 42 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Self-reported comorbidity (yes/no) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hypertension | 19/14 | N/A | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Diabetes | 5/28 | N/A | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Arthritis | 25/8 | N/A | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chronic back pain | 10/23 | N/A | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Medication Use | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Antihypertensive | 23/10 | N/A | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Lipid-lowering | 17/15 | N/A | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Glucose control | 6/27 | N/A | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Anti-depressant | 10/23 | N/A | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Baseline gait speed (m/s) | 1.02 ± 0.19 | 0.56 ̶ 1.44 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| dGaitSp (m/s) | 0.08 ± 0.13 | -0.2 ̶ 0.35 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| dGaitSp (% change) | 9.26 ± 12.92 | -15.24 ̶ 38.89 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Table values are mean ± SD or sample size (N). N/A, not applicable. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Table 2. Pearson correlation analysis of associations between gait speed changes and plasma miRNAs (n=33).
| miR-181a-5p | dmiR-181a-5p (% change) | miR-92a-3p | dmiR-92a-3p (% change) | miR-181a-5p and miR-92a-3p | |
| dGaitSp (m/s) | R = -0.40 | R = 0.15 P = 0 | R = 0.33 | R = -0.45 | R = -0.48 |
| P = 0.02 | 0.40 | P = 0.07 | P = 0.009 | P = 0.005 | |
| dGaitSp (% change) | R = -0.34 | R = 0.13 | R = 0.31 | R = -0.44 | R = -0.42 |
| P = 0.05 | P = 0.49 | P = 0.08 | P = 0.02 | P = 0.01 |
Target genes shared between miR-181a-5p and miR-92a-3p are closely related to biological processes involved in the regulation of neural, skeletal muscle, and vascular function. To better understand the underlying mechanisms related to miR-181a-5p and miR-92a-3p’s involvement in gait speed responses to AEX, we next performed analysis of their target genes. Using the multiMiR R analysis, we detected 1,569 target genes for miR-181a-5p and 1,997 genes for miR-92a-3p, 353 of which were common to both. Go analyses yielded 139 BP Go terms for miR-181a-5p target genes, 176 terms for miR-92a-3p target genes, and 104 terms for the common genes. Using REViGO to remove redundant GO terms revealed that a complex set of genes involved in muscle, vascular and neural physiology regulation could be involved in the response to AEX among obese older adults (Figure 5).
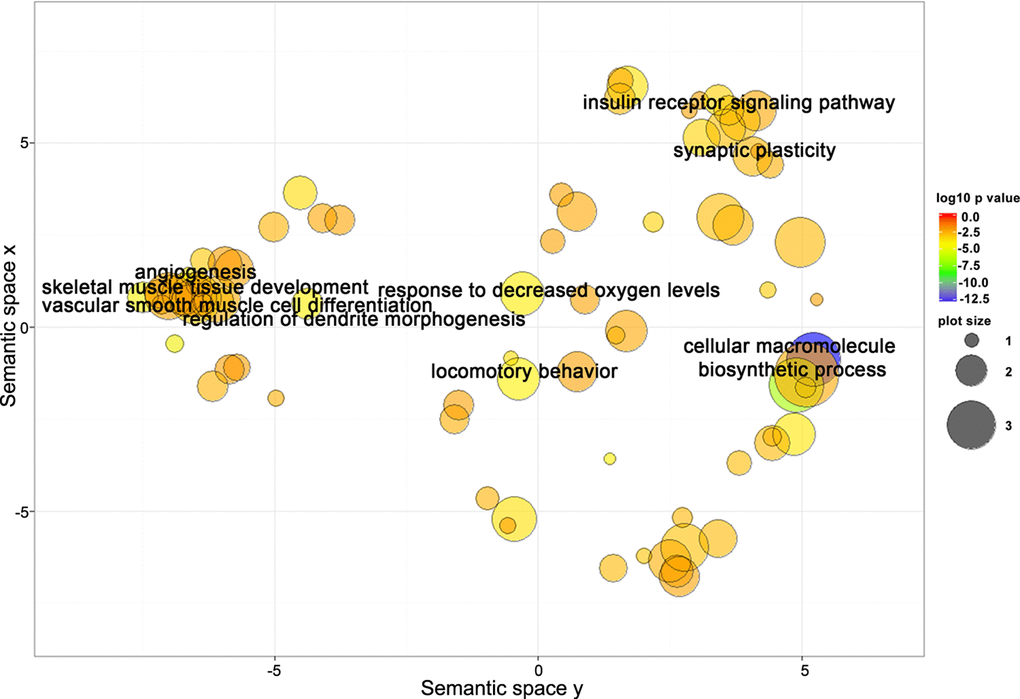
Figure 5. Target genes shared between miR-181a-5p and miR-92a-3p are closely related to biological processes involved in regulation of neural, skeletal muscle, and vascular function. Scatterplot showing semantic similarities of enriched GO terms. Bubble color indicates the GO term enrichment p-value. Bubble size indicates the frequency of the GO term in the reference database (EBI GOA database).
Discussion
The current study provides the first evidence that miR-181a-5p, miR-92a-3p, or their combination, may be useful biomarkers to predict gait speed response to AEX in older adults. Importantly, the identified associations between these two miRNAs and gait speed changes are independent of the baseline gait speed, indicating that these miRNAs may be involved in the regulation of gait speed responses to AEX through mediating their target gene-related signaling pathways, including but not limited to, muscle, vascular, and neural physiology regulation.
These findings add value to the development of ‘personalized medicine’ or ‘precision care’, which is an important concept that has been growing over the recent decades [39]. To date, regular exercise is the most effective therapy for reducing age-related loss of walking speed; however, even though there is non-uniformity of exercise-induced improvement in gait speed among individuals [40], prescription of exercise is often undertaken with a global approach rather than a individualized one [41]. Identification of novel biomarkers that predict gait speed response to therapy is needed before a personalized approach can be used clinically. This will help predetermine if an individual is likely to respond favorably to a certain exercise training mode, or whether a clinical practitioner should prescribe a different therapy.
Given their stability in circulation and the easy application in the clinical practice, circulating miRNAs have been widely studied and used as useful biomarkers for disease prediction, diagnosis, prognosis, and staging [42,43]. Yet, very few studies have investigated the impact of exercise training interventions on miRNA expression, either in tissue or in the circulation. In addition, research into the role of miRNAs in exercise-induced adaptations has predominantly focused on selected miRNA species with limited numbers based on their reported functions, for instance, inflammation, angiogenesis, hypoxia/ischemia adaptation and metabolism [44-48]. These studies indicate that circulating miRNAs could be affected by different exercise modalities, and thus could be potential useful biomarkers or mediators of exercise mode-specific training adaptations [49,50]. We simultaneously characterized 800 circulating miRNAs in a sample of obese older adults and our findings provide novel evidence for the potential use of circulating miRNAs for defining gait speed adaptations to AEX in these individuals.
An important difference between this work and previous studies is our use of the Nanostring nCounter analysis platform to measure miRNAs. The FDA-approved Nanostring nCounter analysis platform has been widely used for plasma and body fluid miRNA profiling, which has shown value as a diagnostic marker for various diseases [51-54]. The direct quantitation of miRNAs without any amplification with the Nanostring nCounter platform, and the normalization of the mean abundance of the top 100 expressed plasma miRNAs, make our data more reliable and easier to repeat on a larger scale. Although RT-qPCR based methods are widely used, it is yet unresolved how the data should be normalized [55]. Different studies use different normalization strategies to report miRNA expression levels; consequently, there is no consensus on the optimal approach [56]. However, there is some evidence that the combination of several internal reference miRNA normalizers might be more appropriate than a single universal normalize [57]. We did not perform RT-qPCR for our validation because of the concern that RT-qPCR usually uses an individual miRNA as internal control, and the effects of exercise on this selected internal control is unknown.
Using the Nanostring platform, we identified 120 abundant circulating miRNAs in the plasma. This number is comparable to the number of circulating miRNAs detected from other groups in healthy subjects using different quantitation platforms, including Solexa sequencing, Sanger sequencing, Agilent microarrays and Taqman qPCR array as was reviewed recently [58]. Compared to the reported top 20 plasma miRNAs from 7 healthy individuals measured with Agilent microarrays [58], 9 of our 120 miRNAs overlapped: miR-451a, miR-16-5p, miR-223-3p, miR-4454, miR-21-5p, miR-23a-3p, let-7a-5p, let-7b-5p, and let-7f-5p. This supports the notion that the Nanostring platform used in our study is as efficient as the other miRNA analysis platforms.
We compared our findings with a recent study examining the miRNA plasma signature in response to acute and prolonged AEX training in young individuals [59]. It appears that young and old individuals may use different circulating miRNAs in mediating responses to exercise. For instance, levels of miR-92a were down-regulated after 12 weeks of AEX in the young individuals [59], while we saw no apparent effect of 5 months of AEX on plasma levels of miR-92a in our older subjects. We further compared our findings with another study by Barr et al. [60,61] that analyzed circulating miRNA responses to a 16-week combined intervention including an energy restriction diet (250 kcal/d) and mixed resistance/aerobic exercise (walking and swimming) (250 kcal/d), focused specifically on 13 preselected plasma miRNAs from middle-aged (35-59 years) individuals. Ten out of the 13 miRNAs they focused on (miR-126-3p, miR-142-3p, miR-223-3p, miR-148b-3p, miR-199a-3p-199b-3p, miR-21-5p, miR-221-3p, miR-423-5p, miR-148a-3p, and miR-140-5p) were also detected by our Nanostring platform analyses in older adults. However, in their study, miR-140-5p, miR-223-3p, and miR-221-3p were modulated by the combined diet and exercise training, whereas we found that miR-376a-3p, miR-16-5p, miR-27a-3p and miR-28-3p were altered by AEX. These differences suggest that individual miRNAs may respond differently based on an individual’s age and the type of intervention.
Our study is also the first to associate plasma miRNAs and gait speed changes in response to sustained AEX. Previous studies indicate that miR-181a-5p or its targets (ROPN1L and SLC37A3) are involved in processes highly relevant to exercise response, including immune function, apoptosis, membrane traffic of proteins and transcription regulation [62,63]. In addition, circulating miR-181a-5p decreases with aging and correlates closely with vascular inflammation and immunity [64,65][REMOVED HYPERLINK FIELD]. Similarly, miR-92a-3p plays a key role in regulation of vascular growth [66] and its serum levels increase with aging [67]. Given our limited sample size and the relatively narrow age range of our participants, we did not detect an age-related difference in miR-181a-5p or miR-92a-3p (data not shown). However, our study shows for the first time that signatures of combined baseline levels of miR-181a-5p or miR-92a-3p could be useful noninvasive biomarkers to predict gait speed responses to AEX in obese older adults. It is worth noting that gait speed changes were not associated with any of the four miRNAs that we found to be changed with AEX (miR-376a-3p, miR-27a-3p, miR-16-5p, and miR-28-3p). In fact, plasma miRNAs associated with changes in gait speed do not appear to be affected by AEX, which is consistent with a recent finding that blood-born miRNA patterns that are useful as biomarkers are not necessarily altered by overall fitness and exercise [68]. Therefore, we propose that the miRNA target genes and consequently their related signaling pathways might be regulated differentially, and may subsequently lead to inter-individual variation in reponses to AEX. Further mechanistic studies on changes in miRNA-regulated target gene networks in muscle and other tissues/systems could help detect novel targets as potential useful tools to adjust physiologic adaptations to AEX in older adults.
Limitations and future directions
Our study has some limitations. Although most of the skeletal muscle-enriched myomiRs [69] (e.g. miR-1, miR-133a, miR-133b, miR-206, and miR-499) were detected by real-time PCR analysis in circulation after various exercise interventions [48,49,70-72], none of them were detected in plasma in our assay. This could have been limited by the sensitivity of Nanostring technology, the relatively lower amount of myomiRs released into circulation, or their higher uptake into recipient cells in other tissues. Without skeletal muscle and other tissues collected in parallel with the plasma in our study, we cannot fully interpret regulation of those specific myomiRs. Similarly, the source and target recipient cells of circulating miR-181a-5p and miR-92a-3p could not be further analyzed and verified. Our target gene prediction and interaction analyses indicated that the predicted miR-181a-5p, miR-92a-3p target genes are closely related to regulation of muscle, vascular and neural physiology, and other important pathways relevant to the physiological response to exercise, including but not limited to apoptosis, transcription regulation, and response to oxygen levels [62]. Yet, without examining skeletal muscle samples, we could not determine the direct or indirect physiological effects of those circulating miRNAs on the skeletal muscle systems. Thus, the current experimental design and data do not provide definitive evidence that miR-181 and miR-92a regulate AEX-induced gait speed changes in obese older adults. It also does not provide a thorough understanding of the molecular pathways that are possibly involved in this gait speed response, which will be the direction of our future mechanistic study.
Notably, we observed no significant associations between several other measured variables (chair rise time, short physical performance battery, blood lipid levels, VO2max, glucose levels, or body composition) and gait speed changes. This indicates that although these metabolic and physiological changes are important responses to aerobic training, they are not necessarily closely related with gait speed changes in obese older adults. The identified miRNAs thus are likely involved in other signaling pathways that regulate gait speed response to AEX in obese older adults, such as cytokine/inflammatory responses. Since we did not measure the levels of cytokines and other inflammation markers, we cannot rule out those possible mechanisms at this time. In addition, all participants in this study completed the intervention with high compliance (≥80% attendance). Thus, it is unlikely that differences in the dose of training contributed to variation in gait speed and miRNA responses to AEX. Although 4 miRNAs were found to change with AEX, none of them were found to be associated with gait speed changes. This could have been limited by the relatively small sample size in this study. Given that the 4 AEX changed miRNAs have been previously reported to be regulated by exercise at the tissue level (i.e., skeletal and cardiac muscle) [62,73-78], it is worth doing further analysis in future studies to explore their related molecular mechanisms.
Given the relatively few participants in this study, we cannot rule out the possibility that some of the observed differences and associations were statistically significant simply due to chance. A future larger and more definitive study with a non-exercise control group is necessary to validate miRNA expression changes with aerobic exercise and determine their role in predicting inter-individual variation in gait speed.
Materials and Methods
Study design and participants
This study included 33 older adults who completed a 5-month AEX intervention with high compliance (≥80% attendance) [10]. Participants were recruited to be 65-79 years of age, obese (BMI = 30-37 kg/m2) with a sedentary lifestyle for the past 6 months, not dependent on a cane or walker, not part of another research study, non-smokers, and free of osteoporosis, abnormal kidney function, insulin-dependent or uncontrolled diabetes, or uncontrolled high blood pressure. Body composition, lower extremity function, and fasting lipid and glucose levels were measured as previously described [10,79]. Prevalent comorbidities were assessed by self-report and medication use. Clinical characteristics of the study participants are shown in Table 1. The study was approved by the Wake Forest School of Medicine Institutional Review Board and all participants provided written informed consent.
Exercise intervention
Participants performed supervised treadmill walking 4 days per week for 5 months. Exercise sessions progressed from 15–20 minutes at 50% heart rate reserve (HRR, assessed during a graded exercise treadmill test) during the 1st week to 30 minutes at 65–70% HRR by the end of the 6th week and included a 5-minute warm-up, 5-minute cool-down, and light stretching. Heart rate, treadmill grade/speed, exercise duration, and the amount of energy expended were recorded each session to monitor compliance to the exercise prescription.
Gait speed
Gait speed was assessed with a fast-paced 400-meter walk test [80]. The test-retest reliability and validity of the 400-meter walk test in middle-aged and older adults have been reported previously [81,82]. Participants were instructed to walk the 400-meter distance (10 laps on a flat indoor surface 20 m in length) as quickly as possible. Time to complete the walk was recorded in seconds. Standardized encouragement was given every lap.
RNA extraction from plasma samples
Blood samples were collected into EDTA-treated tubes in the morning after an overnight fast before and 36-48 hours after the last intervention bout of AEX. Plasma was isolated by spinning blood at 2,000 × g for 20 min at 4 °C and stored at −80°C for later analysis. After thawing, the plasma was centrifuged at 2,000 × g for 10 min at 4 °C to remove debris and deplete platelets, and 500 µl supernatant was used for total RNA extraction using kit #5100 (Norgen Biotek, Thorold, Ontario, Canada) according to the manufacturer’s protocol. Eluted total RNA (200 µl) was precipitated overnight with sodium acetate/ethanol with a linear acrylamide carrier (Ambion, Austin, TX, USA) for maximum nucleic acid recovery. RNA pellets were concentrated in 15 µl ddH2O; 3 µl samples were submitted for profiling on the multiplexed nCounter platform (Nanostring Technologies, Seattle, WA, USA).
MiRNA expression profiling with Nanostring nCounter analysis
RNA samples were prepared by ligating a specific DNA tag (miR-tag) onto the 3’ end of each mature miRNA, according to the manufacturer’s instructions. Assay Kit NS_H_MIR_V2.1 (Nanostring Technologies) was used to anneal miRNAs to target specific barcode probes. No amplification was required. Excess tags were removed by restrict digestion at 37 °C. Hybridizations were carried out by combining 5 µl of each miRNA-miR-Tag sample with 20 µl of nCounter Reporter probes in hybridization buffer and 5 µl of nCounter Capture probes (for a total reaction volume of 30 µl) at 65°C for 16–20 hours. Excess probes were removed using a two-step magnetic bead-based purification on the nCounter Prep Station. Abundance of specific target molecules was quantified by imaging the immobilized fluorescent reporters in the sample cartridge with a CCD camera and counting the individual fluorescent barcode probes for each miRNA target (800 human miRNAs) using the nCounter Digital Analyzer. Data were normalized to the top 100 expressed miRNAs [83]. The background was corrected using the nSolver software package. A 30.21 count threshold was set using negative controls based upon the equation: mean + 3× SD. MiRNAs with more than 70% counts below this threshold were excluded.
MiRNA target gene prediction and interaction analysis
The multiMiR R package with retrieval of miRNA-target interactions from 14 external databases was used to predict target genes. Furthermore, we performed enrichment analysis for gene ontology using the TopGo package. For miRNA targeted gene sets, Fisher’s exact test was implemented, and the enriched Biological Process (BP) Go terms were selected based on a p-value <0.01. The GO list was further summarized using REViGO, which removed redundant GO terms. Data were visualized in semantic similarity-based scatterplots.
Data analysis
MiRNA data were analyzed using the nSolver software (Nanostring Technologies) and SPSS software from IBM (Armonk, NY, USA). MiRNA raw counts were log2 transformed and all data were expressed as mean ± standard deviation (SD). The Student’s t test was used to compare continuous variables. Categorical data were compared using the Chi-square test. Pearson product-moment correlation was used to measure the strength of the association between pairs of variables. A linear combination of identified miRNAs was also calculated to further assess associations with gait speed. Analysis of covariance was used to determine if associations between gait speed and miRNAs were independent of age and gender. A p-value ≤0.05 was considered statistically significant.
Supplementary Materials
Acknowledgements
We thank Karen Potvin Klein, MA, ELS in the Wake Forest Translational Science Institute (UL1 TR001420 to McClain) for her editorial contributions to this manuscript.
Conflicts of Interest
The authors declare that they have no conflict of interest.
Funding
This work was supported by departmental funding (T. Z. and T. B.) and the National Institutes of Health (P30 AG21332 to S. K., and R01 AG020583 to B.N.).
References
- 1. Branch LG, Jette AM. A prospective study of long-term care institutionalization among the aged. Am J Public Health. 1982; 72:1373–79. https://doi.org/10.2105/AJPH.72.12.1373 [PubMed]
- 2. Hirvensalo M, Rantanen T, Heikkinen E. Mobility difficulties and physical activity as predictors of mortality and loss of independence in the community-living older population. J Am Geriatr Soc. 2000; 48:493–98. https://doi.org/10.1111/j.1532-5415.2000.tb04994.x [PubMed]
- 3. Perera S, Patel KV, Rosano C, Rubin SM, Satterfield S, Harris T, Ensrud K, Orwoll E, Lee CG, Chandler JM, Newman AB, Cauley JA, Guralnik JM, et al. Gait Speed Predicts Incident Disability: A Pooled Analysis. J Gerontol A Biol Sci Med Sci. 2016; 71:63–71. https://doi.org/10.1093/gerona/glv126 [PubMed]
- 4. Studenski S, Perera S, Patel K, Rosano C, Faulkner K, Inzitari M, Brach J, Chandler J, Cawthon P, Connor EB, Nevitt M, Visser M, Kritchevsky S, et al. Gait speed and survival in older adults. JAMA. 2011; 305:50–58. https://doi.org/10.1001/jama.2010.1923 [PubMed]
- 5. Hardy SE, Perera S, Roumani YF, Chandler JM, Studenski SA. Improvement in usual gait speed predicts better survival in older adults. J Am Geriatr Soc. 2007; 55:1727–34. https://doi.org/10.1111/j.1532-5415.2007.01413.x [PubMed]
- 6. Abellan van Kan G, Rolland Y, Andrieu S, Bauer J, Beauchet O, Bonnefoy M, Cesari M, Donini LM, Gillette Guyonnet S, Inzitari M, Nourhashemi F, Onder G, Ritz P, et al. Gait speed at usual pace as a predictor of adverse outcomes in community-dwelling older people an International Academy on Nutrition and Aging (IANA) Task Force. J Nutr Health Aging. 2009; 13:881–89. https://doi.org/10.1007/s12603-009-0246-z [PubMed]
- 7. Kim HJ, Park I, Lee HJ, Lee O. The reliability and validity of gait speed with different walking pace and distances against general health, physical function, and chronic disease in aged adults. J Exerc Nutrition Biochem. 2016; 20:46–50. https://doi.org/10.20463/jenb.2016.09.20.3.7 [PubMed]
- 8. Bouchard C, Blair SN, Church TS, Earnest CP, Hagberg JM, Häkkinen K, Jenkins NT, Karavirta L, Kraus WE, Leon AS, Rao DC, Sarzynski MA, Skinner JS, et al. Adverse metabolic response to regular exercise: is it a rare or common occurrence? PLoS One. 2012; 7:e37887. https://doi.org/10.1371/journal.pone.0037887 [PubMed]
- 9. Bouchard C, Rankinen T. Individual differences in response to regular physical activity. Med Sci Sports Exerc. 2001 (Suppl ); 33:S446–51. https://doi.org/10.1097/00005768-200106001-00013 [PubMed]
- 10. Chmelo EA, Crotts CI, Newman JC, Brinkley TE, Lyles MF, Leng X, Marsh AP, Nicklas BJ. Heterogeneity of physical function responses to exercise training in older adults. J Am Geriatr Soc. 2015; 63:462–69. https://doi.org/10.1111/jgs.13322 [PubMed]
- 11. Henderson RM, Leng XI, Chmelo EA, Brinkley TE, Lyles MF, Marsh AP, Nicklas BJ. Gait speed response to aerobic versus resistance exercise training in older adults. Aging Clin Exp Res. 2016. https://doi.org/10.1007/s40520-016-0632-4 [PubMed]
- 12. Perera S, Mody SH, Woodman RC, Studenski SA. Meaningful change and responsiveness in common physical performance measures in older adults. J Am Geriatr Soc. 2006; 54:743–49. https://doi.org/10.1111/j.1532-5415.2006.00701.x [PubMed]
- 13. Grimson A, Farh KK, Johnston WK, Garrett-Engele P, Lim LP, Bartel DP. MicroRNA targeting specificity in mammals: determinants beyond seed pairing. Mol Cell. 2007; 27:91–105. https://doi.org/10.1016/j.molcel.2007.06.017 [PubMed]
- 14. Dey BK, Gagan J, Dutta A. miR-206 and -486 induce myoblast differentiation by downregulating Pax7. Mol Cell Biol. 2011; 31:203–14. https://doi.org/10.1128/MCB.01009-10 [PubMed]
- 15. Moresi V, Williams AH, Meadows E, Flynn JM, Potthoff MJ, McAnally J, Shelton JM, Backs J, Klein WH, Richardson JA, Bassel-Duby R, Olson EN. Myogenin and class II HDACs control neurogenic muscle atrophy by inducing E3 ubiquitin ligases. Cell. 2010; 143:35–45. https://doi.org/10.1016/j.cell.2010.09.004 [PubMed]
- 16. Nakasa T, Ishikawa M, Shi M, Shibuya H, Adachi N, Ochi M. Acceleration of muscle regeneration by local injection of muscle-specific microRNAs in rat skeletal muscle injury model. J Cell Mol Med. 2010; 14:2495–505. https://doi.org/10.1111/j.1582-4934.2009.00898.x [PubMed]
- 17. Small EM, O’Rourke JR, Moresi V, Sutherland LB, McAnally J, Gerard RD, Richardson JA, Olson EN. Regulation of PI3-kinase/Akt signaling by muscle-enriched microRNA-486. Proc Natl Acad Sci USA. 2010; 107:4218–23. https://doi.org/10.1073/pnas.1000300107 [PubMed]
- 18. van Rooij E, Quiat D, Johnson BA, Sutherland LB, Qi X, Richardson JA, Kelm RJ
Jr , Olson EN. A family of microRNAs encoded by myosin genes governs myosin expression and muscle performance. Dev Cell. 2009; 17:662–73. https://doi.org/10.1016/j.devcel.2009.10.013 [PubMed] - 19. Williams AH, Valdez G, Moresi V, Qi X, McAnally J, Elliott JL, Bassel-Duby R, Sanes JR, Olson EN. MicroRNA-206 delays ALS progression and promotes regeneration of neuromuscular synapses in mice. Science. 2009; 326:1549–54. https://doi.org/10.1126/science.1181046 [PubMed]
- 20. Duttagupta R, Jiang R, Gollub J, Getts RC, Jones KW. Impact of cellular miRNAs on circulating miRNA biomarker signatures. PLoS One. 2011; 6:e20769. https://doi.org/10.1371/journal.pone.0020769 [PubMed]
- 21. Hoy AM, Buck AH. Extracellular small RNAs: what, where, why? Biochem Soc Trans. 2012; 40:886–90. https://doi.org/10.1042/BST20120019 [PubMed]
- 22. Weber DG, Casjens S, Rozynek P, Lehnert M, Zilch-Schöneweis S, Bryk O, Taeger D, Gomolka M, Kreuzer M, Otten H, Pesch B, Johnen G, Brüning T. Assessment of mRNA and microRNA Stabilization in Peripheral Human Blood for Multicenter Studies and Biobanks. Biomark Insights. 2010; 5:95–102. https://doi.org/10.4137/BMI.S5522 [PubMed]
- 23. Xu L, Yang BF, Ai J. MicroRNA transport: a new way in cell communication. J Cell Physiol. 2013; 228:1713–19. https://doi.org/10.1002/jcp.24344 [PubMed]
- 24. Heneghan HM, Miller N, Lowery AJ, Sweeney KJ, Newell J, Kerin MJ. Circulating microRNAs as novel minimally invasive biomarkers for breast cancer. Ann Surg. 2010; 251:499–505. https://doi.org/10.1097/SLA.0b013e3181cc939f [PubMed]
- 25. Mitchell PS, Parkin RK, Kroh EM, Fritz BR, Wyman SK, Pogosova-Agadjanyan EL, Peterson A, Noteboom J, O’Briant KC, Allen A, Lin DW, Urban N, Drescher CW, et al. Circulating microRNAs as stable blood-based markers for cancer detection. Proc Natl Acad Sci USA. 2008; 105:10513–18. https://doi.org/10.1073/pnas.0804549105 [PubMed]
- 26. Wang GK, Zhu JQ, Zhang JT, Li Q, Li Y, He J, Qin YW, Jing Q. Circulating microRNA: a novel potential biomarker for early diagnosis of acute myocardial infarction in humans. Eur Heart J. 2010; 31:659–66. https://doi.org/10.1093/eurheartj/ehq013 [PubMed]
- 27. Wang K, Zhang S, Marzolf B, Troisch P, Brightman A, Hu Z, Hood LE, Galas DJ. Circulating microRNAs, potential biomarkers for drug-induced liver injury. Proc Natl Acad Sci USA. 2009; 106:4402–07. https://doi.org/10.1073/pnas.0813371106 [PubMed]
- 28. Bye A, Røsjø H, Aspenes ST, Condorelli G, Omland T, Wisløff U. Circulating microRNAs and aerobic fitness--the HUNT-Study. PLoS One. 2013; 8:e57496. https://doi.org/10.1371/journal.pone.0057496 [PubMed]
- 29. Donaldson A, Natanek SA, Lewis A, Man WD, Hopkinson NS, Polkey MI, Kemp PR. Increased skeletal muscle-specific microRNA in the blood of patients with COPD. Thorax. 2013; 68:1140–49. https://doi.org/10.1136/thoraxjnl-2012-203129 [PubMed]
- 30. Mooren FC, Viereck J, Krüger K, Thum T. Circulating microRNAs as potential biomarkers of aerobic exercise capacity. Am J Physiol Heart Circ Physiol. 2014; 306:H557–63. https://doi.org/10.1152/ajpheart.00711.2013 [PubMed]
- 31. Aoi W, Sakuma K. Does regulation of skeletal muscle function involve circulating microRNAs? Front Physiol. 2014; 5:39. https://doi.org/10.3389/fphys.2014.00039 [PubMed]
- 32. Kirby TJ, McCarthy JJ. MicroRNAs in skeletal muscle biology and exercise adaptation. Free Radic Biol Med. 2013; 64:95–105. https://doi.org/10.1016/j.freeradbiomed.2013.07.004 [PubMed]
- 33. McGregor RA, Poppitt SD, Cameron-Smith D. Role of microRNAs in the age-related changes in skeletal muscle and diet or exercise interventions to promote healthy aging in humans. Ageing Res Rev. 2014; 17:25–33. https://doi.org/10.1016/j.arr.2014.05.001 [PubMed]
- 34. Turchinovich A, Samatov TR, Tonevitsky AG, Burwinkel B. Circulating miRNAs: cell-cell communication function? Front Genet. 2013; 4:119. https://doi.org/10.3389/fgene.2013.00119 [PubMed]
- 35. Nielsen S, Scheele C, Yfanti C, Akerström T, Nielsen AR, Pedersen BK, Laye MJ. Muscle specific microRNAs are regulated by endurance exercise in human skeletal muscle. J Physiol. 2010; 588:4029–37. https://doi.org/10.1113/jphysiol.2010.189860 [PubMed]
- 36. de Gonzalo-Calvo D, Dávalos A, Montero A, García-González Á, Tyshkovska I, González-Medina A, Soares SM, Martínez-Camblor P, Casas-Agustench P, Rabadán M, Díaz-Martínez AE, Úbeda N, Iglesias-Gutiérrez E. Circulating inflammatory miRNA signature in response to different doses of aerobic exercise. J Appl Physiol (1985). 2015; 119:124–34. https://doi.org/10.1152/japplphysiol.00077.2015 [PubMed]
- 37. Davidsen PK, Gallagher IJ, Hartman JW, Tarnopolsky MA, Dela F, Helge JW, Timmons JA, Phillips SM. High responders to resistance exercise training demonstrate differential regulation of skeletal muscle microRNA expression. J Appl Physiol (1985). 2011; 110:309–17. https://doi.org/10.1152/japplphysiol.00901.2010 [PubMed]
- 38. Dong H, Li J, Huang L, Chen X, Li D, Wang T, Hu C, Xu J, Zhang C, Zen K, Xiao S, Yan Q, Wang C, Zhang CY. Serum MicroRNA Profiles Serve as Novel Biomarkers for the Diagnosis of Alzheimer’s Disease. Dis Markers. 2015; 2015:625659. https://doi.org/10.1155/2015/625659 [PubMed]
- 39. Bouchard C, Antunes-Correa LM, Ashley EA, Franklin N, Hwang PM, Mattsson CM, Negrao CE, Phillips SA, Sarzynski MA, Wang PY, Wheeler MT. Personalized preventive medicine: genetics and the response to regular exercise in preventive interventions. Prog Cardiovasc Dis. 2015; 57:337–46. https://doi.org/10.1016/j.pcad.2014.08.005 [PubMed]
- 40. Buford TW, Hsu FC, Brinkley TE, Carter CS, Church TS, Dodson JA, Goodpaster BH, McDermott MM, Nicklas BJ, Yank V, Johnson JA, Pahor M. Genetic influence on exercise-induced changes in physical function among mobility-limited older adults. Physiol Genomics. 2014; 46:149–58 https://doi.org/10.1152/physiolgenomics.00169.2013 [PubMed]
- 41. Mann TN, Lamberts RP, Lambert MI. High responders and low responders: factors associated with individual variation in response to standardized training. Sports Med. 2014; 44:1113–24. https://doi.org/10.1007/s40279-014-0197-3 [PubMed]
- 42. Martinez B, Peplow PV. Blood microRNAs as potential diagnostic and prognostic markers in cerebral ischemic injury. Neural Regen Res. 2016; 11:1375–78. [PubMed]
- 43. Westphal M, Lamszus K. Circulating biomarkers for gliomas. Nat Rev Neurol. 2015; 11:556–66. https://doi.org/10.1038/nrneurol.2015.171 [PubMed]
- 44. Baggish AL, Hale A, Weiner RB, Lewis GD, Systrom D, Wang F, Wang TJ, Chan SY. Dynamic regulation of circulating microRNA during acute exhaustive exercise and sustained aerobic exercise training. J Physiol. 2011; 589:3983–94. https://doi.org/10.1113/jphysiol.2011.213363 [PubMed]
- 45. Baggish AL, Park J, Min PK, Isaacs S, Parker BA, Thompson PD, Troyanos C, D’Hemecourt P, Dyer S, Thiel M, Hale A, Chan SY. Rapid upregulation and clearance of distinct circulating microRNAs after prolonged aerobic exercise. J Appl Physiol (1985). 2014; 116:522–31. https://doi.org/10.1152/japplphysiol.01141.2013 [PubMed]
- 46. Cui SF, Li W, Niu J, Zhang CY, Chen X, Ma JZ. Acute responses of circulating microRNAs to low-volume sprint interval cycling. Front Physiol. 2015; 6:311. https://doi.org/10.3389/fphys.2015.00311 [PubMed]
- 47. Russell AP, Lamon S. Exercise, Skeletal Muscle and Circulating microRNAs. Prog Mol Biol Transl Sci. 2015; 135:471–96. https://doi.org/10.1016/bs.pmbts.2015.07.018 [PubMed]
- 48. Gomes CP, Oliveira- GP
Jr , Madrid B, Almeida JA, Franco OL, Pereira RW. Circulating miR-1, miR-133a, and miR-206 levels are increased after a half-marathon run. Biomarkers. 2014; 19:585–89. https://doi.org/10.3109/1354750X.2014.952663 [PubMed] - 49. Wardle SL, Bailey ME, Kilikevicius A, Malkova D, Wilson RH, Venckunas T, Moran CN. Plasma microRNA levels differ between endurance and strength athletes. PLoS One. 2015; 10:e0122107. https://doi.org/10.1371/journal.pone.0122107 [PubMed]
- 50. Banzet S, Chennaoui M, Girard O, Racinais S, Drogou C, Chalabi H, Koulmann N. Changes in circulating microRNAs levels with exercise modality. J Appl Physiol (1985). 2013; 115:1237–44. https://doi.org/10.1152/japplphysiol.00075.2013 [PubMed]
- 51. Zuo Z, Maiti S, Hu S, Loghavi S, Calin GA, Garcia-Manero G, Kantarjian HM, Medeiros LJ, Cooper LJ, Bueso-Ramos CE. Plasma circulating-microRNA profiles are useful for assessing prognosis in patients with cytogenetically normal myelodysplastic syndromes. Mod Pathol. 2015; 28:373–82. https://doi.org/10.1038/modpathol.2014.108 [PubMed]
- 52. Oikonomopoulos A, Polytarchou C, Joshi S, Hommes DW, Iliopoulos D. Identification of Circulating MicroRNA Signatures in Crohn’s Disease Using the Nanostring nCounter Technology. Inflamm Bowel Dis. 2016; 22:2063–69. https://doi.org/10.1097/MIB.0000000000000883 [PubMed]
- 53. Polytarchou C, Oikonomopoulos A, Mahurkar S, Touroutoglou A, Koukos G, Hommes DW, Iliopoulos D. Assessment of Circulating MicroRNAs for the Diagnosis and Disease Activity Evaluation in Patients with Ulcerative Colitis by Using the Nanostring Technology. Inflamm Bowel Dis. 2015; 21:2533–39. https://doi.org/10.1097/MIB.0000000000000547 [PubMed]
- 54. Pfeffer SR, Grossmann KF, Cassidy PB, Yang CH, Fan M, Kopelovich L, Leachman SA, Pfeffer LM. Detection of Exosomal miRNAs in the Plasma of Melanoma Patients. J Clin Med. 2015; 4:2012–27. https://doi.org/10.3390/jcm4121957 [PubMed]
- 55. Marabita F, de Candia P, Torri A, Tegnér J, Abrignani S, Rossi RL. Normalization of circulating microRNA expression data obtained by quantitative real-time RT-PCR. Brief Bioinform. 2016; 17:204–12. https://doi.org/10.1093/bib/bbv056 [PubMed]
- 56. Chugh P, Dittmer DP. Potential pitfalls in microRNA profiling. Wiley Interdiscip Rev RNA. 2012; 3:601–16. https://doi.org/10.1002/wrna.1120 [PubMed]
- 57. Shen Y, Tian F, Chen Z, Li R, Ge Q, Lu Z. Amplification-based method for microRNA detection. Biosens Bioelectron. 2015; 71:322–31. https://doi.org/10.1016/j.bios.2015.04.057 [PubMed]
- 58. Hruštincová A, Votavová H, Dostálová Merkerová M. Circulating MicroRNAs: Methodological Aspects in Detection of These Biomarkers. Folia Biol (Praha). 2015; 61:203–18. [PubMed]
- 59. Nielsen S, Åkerström T, Rinnov A, Yfanti C, Scheele C, Pedersen BK, Laye MJ. The miRNA plasma signature in response to acute aerobic exercise and endurance training. PLoS One. 2014; 9:e87308. https://doi.org/10.1371/journal.pone.0087308 [PubMed]
- 60. Parr EB, Camera DM, Burke LM, Phillips SM, Coffey VG, Hawley JA. Circulating MicroRNA Responses between ‘High’ and ‘Low’ Responders to a 16-Wk Diet and Exercise Weight Loss Intervention. PLoS One. 2016; 11:e0152545. https://doi.org/10.1371/journal.pone.0152545 [PubMed]
- 61. Parr EB, Coffey VG, Cato LE, Phillips SM, Burke LM, Hawley JA. A randomized trial of high-dairy-protein, variable-carbohydrate diets and exercise on body composition in adults with obesity. Obesity (Silver Spring). 2016; 24:1035–45. https://doi.org/10.1002/oby.21451 [PubMed]
- 62. Tonevitsky AG, Maltseva DV, Abbasi A, Samatov TR, Sakharov DA, Shkurnikov MU, Lebedev AE, Galatenko VV, Grigoriev AI, Northoff H. Dynamically regulated miRNA-mRNA networks revealed by exercise. BMC Physiol. 2013; 13:9. https://doi.org/10.1186/1472-6793-13-9 [PubMed]
- 63. Russell AP, Lamon S, Boon H, Wada S, Güller I, Brown EL, Chibalin AV, Zierath JR, Snow RJ, Stepto N, Wadley GD, Akimoto T. Regulation of miRNAs in human skeletal muscle following acute endurance exercise and short-term endurance training. J Physiol. 2013; 591:4637–53. https://doi.org/10.1113/jphysiol.2013.255695 [PubMed]
- 64. Noren Hooten N, Fitzpatrick M, Wood WH
3rd , De S, Ejiogu N, Zhang Y, Mattison JA, Becker KG, Zonderman AB, Evans MK. Age-related changes in microRNA levels in serum. Aging (Albany NY). 2013; 5:725–40. https://doi.org/10.18632/aging.100603 [PubMed] - 65. Sun X, Sit A, Feinberg MW. Role of miR-181 family in regulating vascular inflammation and immunity. Trends Cardiovasc Med. 2014; 24:105–12. https://doi.org/10.1016/j.tcm.2013.09.002 [PubMed]
- 66. Hazra S, Henson GD, Morgan RG, Breevoort SR, Ives SJ, Richardson RS, Donato AJ, Lesniewski LA. Experimental reduction of miR-92a mimics arterial aging. Exp Gerontol. 2016; 83:165–70. https://doi.org/10.1016/j.exger.2016.08.007 [PubMed]
- 67. Zhang H, Yang H, Zhang C, Jing Y, Wang C, Liu C, Zhang R, Wang J, Zhang J, Zen K, Zhang C, Li D. Investigation of microRNA expression in human serum during the aging process. J Gerontol A Biol Sci Med Sci. 2015; 70:102–09. https://doi.org/10.1093/gerona/glu145 [PubMed]
- 68. Backes C, Leidinger P, Keller A, Hart M, Meyer T, Meese E, Hecksteden A. Blood born miRNAs signatures that can serve as disease specific biomarkers are not significantly affected by overall fitness and exercise. PLoS One. 2014; 9:e102183. https://doi.org/10.1371/journal.pone.0102183 [PubMed]
- 69. McCarthy JJ. The MyomiR network in skeletal muscle plasticity. Exerc Sport Sci Rev. 2011; 39:150–54. https://doi.org/10.1097/JES.0b013e31821c01e1 [PubMed]
- 70. Cui SF, Wang C, Yin X, Tian D, Lu QJ, Zhang CY, Chen X, Ma JZ. Similar Responses of Circulating MicroRNAs to Acute High-Intensity Interval Exercise and Vigorous-Intensity Continuous Exercise. Front Physiol. 2016; 7:102. https://doi.org/10.3389/fphys.2016.00102 [PubMed]
- 71. Zhang T, Birbrair A, Wang ZM, Messi ML, Marsh AP, Leng I, Nicklas BJ, Delbono O. Improved knee extensor strength with resistance training associates with muscle specific miRNAs in older adults. Exp Gerontol. 2015; 62:7–13. https://doi.org/10.1016/j.exger.2014.12.014 [PubMed]
- 72. Cui SF, Li W, Niu J, Zhang CY, Chen X, Ma JZ. Acute responses of circulating microRNAs to low-volume sprint interval cycling. Front Physiol. 2015; 6:311. https://doi.org/10.3389/fphys.2015.00311 [PubMed]
- 73. Wahl P, Wehmeier UF, Jansen FJ, Kilian Y, Bloch W, Werner N, Mester J, Hilberg T. Acute Effects of Different Exercise Protocols on the Circulating Vascular microRNAs -16, -21, and -126 in Trained Subjects. Front Physiol. 2016; 7:643. https://doi.org/10.3389/fphys.2016.00643 [PubMed]
- 74. Ogasawara R, Akimoto T, Umeno T, Sawada S, Hamaoka T, Fujita S. MicroRNA expression profiling in skeletal muscle reveals different regulatory patterns in high and low responders to resistance training. Physiol Genomics. 2016; 48:320–24. https://doi.org/10.1152/physiolgenomics.00124.2015 [PubMed]
- 75. Liu X, Trakooljul N, Hadlich F, Muráni E, Wimmers K, Ponsuksili S. MicroRNA-mRNA regulatory networking fine-tunes the porcine muscle fiber type, muscular mitochondrial respiratory and metabolic enzyme activities. BMC Genomics. 2016; 17:531. https://doi.org/10.1186/s12864-016-2850-8 [PubMed]
- 76. Lee DE, Brown JL, Rosa ME, Brown LA, Perry RA
Jr , Wiggs MP, Nilsson MI, Crouse SF, Fluckey JD, Washington TA, Greene NP. microRNA-16 Is Downregulated During Insulin Resistance and Controls Skeletal Muscle Protein Accretion. J Cell Biochem. 2016; 117:1775–87. https://doi.org/10.1002/jcb.25476 [PubMed] - 77. Fernandes T, Baraúna VG, Negrão CE, Phillips MI, Oliveira EM. Aerobic exercise training promotes physiological cardiac remodeling involving a set of microRNAs. Am J Physiol Heart Circ Physiol. 2015; 309:H543–52. https://doi.org/10.1152/ajpheart.00899.2014 [PubMed]
- 78. Dmitriev P, Barat A, Polesskaya A, O’Connell MJ, Robert T, Dessen P, Walsh TA, Lazar V, Turki A, Carnac G, Laoudj-Chenivesse D, Lipinski M, Vassetzky YS. Simultaneous miRNA and mRNA transcriptome profiling of human myoblasts reveals a novel set of myogenic differentiation-associated miRNAs and their target genes. BMC Genomics. 2013; 14:265. https://doi.org/10.1186/1471-2164-14-265 [PubMed]
- 79. Normandin E, Chmelo E, Lyles MF, Marsh AP, Nicklas BJ. Effect of Resistance Training and Caloric Restriction on the Metabolic Syndrome. Med Sci Sports Exerc. 2016. [PubMed]
- 80. Simonsick EM, Montgomery PS, Newman AB, Bauer DC, Harris T. Measuring fitness in healthy older adults: the Health ABC Long Distance Corridor Walk. J Am Geriatr Soc. 2001; 49:1544–48. https://doi.org/10.1046/j.1532-5415.2001.4911247.x [PubMed]
- 81. Pettee Gabriel KK, Rankin RL, Lee C, Charlton ME, Swan PD, Ainsworth BE. Test-retest reliability and validity of the 400-meter walk test in healthy, middle-aged women. J Phys Act Health. 2010; 7:649–57. https://doi.org/10.1123/jpah.7.5.649 [PubMed]
- 82. Rolland YM, Cesari M, Miller ME, Penninx BW, Atkinson HH, Pahor M. Reliability of the 400-m usual-pace walk test as an assessment of mobility limitation in older adults. J Am Geriatr Soc. 2004; 52:972–76. https://doi.org/10.1111/j.1532-5415.2004.52267.x [PubMed]
- 83. Fourie NH, Peace RM, Abey SK, Sherwin LB, Rahim-Williams B, Smyser PA, Wiley JW, Henderson WA. Elevated circulating miR-150 and miR-342-3p in patients with irritable bowel syndrome. Exp Mol Pathol. 2014; 96:422–25. https://doi.org/10.1016/j.yexmp.2014.04.009 [PubMed]


