Introduction
Frailty is among the most complex and problematic conditions in older individuals. It is a state of increased vulnerability to stressors and adverse outcomes due to a multisystem loss of homeostasis [1]. The importance of frailty is highlighted by its consistent association with all-cause mortality and adverse aging outcomes, such as institutionalization, physical limitations, disability, recurrent hospitalizations, falls and fractures [2,3]. Frailty has also been shown to be strongly predictive of post-operative mortality, complications, and prolonged length of stay in older surgical patients [4]. In individuals aged 65 years and older, a dose-responsive reduction in survival probability has been observed with increasing levels of frailty [2]. It has been estimated that that up to 5% of deaths among older individuals could be delayed by preventing frailty [2].
There are currently two common models of frailty: the Fried frailty phenotype [5] and the Rockwood frailty index (FI) [6]. The former sees frailty as a syndrome with loss of physical function and it classifies individuals as non-frail, pre-frail and frail, whereas the latter sees frailty as a continuous risk state assessed as the number of accumulated health deficits. Although both measures are valid predictors of adverse outcomes and demonstrate overlap in the identification of frailty [7], the FI – being a continuous measure – shows more sensitivity at the lower end of the frailty continuum [8]. In addition, the FI may provide better resolution in younger populations as it also predicts adverse outcomes among individuals who are classified as non-frail by the Fried phenotypic model [9]. Nevertheless, despite the acknowledged need to screen all individuals aged 70 years and older for frailty [10], a consensus is still lacking as to how to best assess it in clinical settings.
Whether these frailty instruments also predict mortality among the younger individuals (under 65 years), whose levels of frailty are generally much lower and times to death are longer, is less-studied. Sex differences in this respect have also received little attention. A growing body of evidence links frailty to the development of cardiovascular disease (CVD) [11,12] and dementia [13], and the presence of frailty is also known to worsen the outcomes of these diseases as well as cancer [14]. However, less is known about the association of frailty with deaths due to these causes and especially in community settings. To this end, we analyzed the predictive ability of a 42-item Rockwood-based FI, herein referred to as the FI, for all-cause mortality in a population-based cohort of individuals aged from 29 to 95 years at baseline with a 30-year mortality follow-up. We also elucidated the relationship between the FI and CVD-, cancer-, dementia- and other-cause mortality.
Results
The FI in the study sample
Characteristics of the study population are presented in Table 1. The distribution of the FI was skewed with a long right tail (Supplementary Figure 1) and the association with age was exponential in both men and women (Figure 1a). The maximum value of FI was 0.619 in women and 0.536 in men. These findings align with the existing literature on the maximum FI value, 0.7 rather than 1.0, which indicates that regardless of the study population and the deficits included in the FI, survival beyond FI 0.7 is unlikely [6,15]. Although the median level of frailty was relatively low in this sample (Table 1), sex differences across the age range and a higher mortality risk for men at all levels of FI were apparent (Figure 1b).
Table 1. Characteristics of the study population.
| All (n=1477) Median (IQR) | Men (n=623) Median (IQR) | Women (n=854) Median (IQR) | p* | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age (yr) | 63.2 | (21.0) | 62.9 | (19.4) | 63.5 | (21.8) | 0.037 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BMI (kg/m2) | 24.2 | (4.4) | 24.7 | (3.8) | 23.6 | (4.8) | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Smoking statusa | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Never | 1032 | (70.7) | 397 | (64.0) | 635 | (75.6) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Previous | 87 | (6.0) | 52 | (8.4) | 35 | (4.2) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Current | 341 | (23.4) | 171 | (27.6) | 170 | (20.2) | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Educationa | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Primary education | 800 | (57.7) | 325 | (55.5) | 475 | (59.4) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Lower secondary or Vocational education | 383 | (27.6) | 148 | (25.3) | 235 | (29.4) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Upper secondary education | 109 | (7.8) | 62 | (10.6) | 47 | (5.9) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Tertiary education | 49 | (6.9) | 51 | (8.7) | 43 | (5.4) | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| FI | 0.077 | (0.095) | 0.071 | (0.083) | 0.083 | (0.107) | 0.007 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| FI categorizeda | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Relatively fit | 288 | (19.5) | 122 | (19.6) | 166 | (19.4) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Less fit | 622 | (42.1) | 279 | (44.5) | 343 | (40.2) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Least fit | 401 | (27.2) | 175 | (28.1) | 226 | (26.5) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Frail | 166 | (11.2) | 47 | (7.5) | 119 | (13.9) | 0.002 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| FI categorizeda younger individuals (<65 yrs) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Relatively fit | 224 | (27.8) | 101 | (28.6) | 123 | (27.1) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Less fit | 378 | (48.0) | 173 | (49.0) | 214 | (47.1) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Least fit | 155 | (19.2) | 66 | (18.7) | 89 | (19.6) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Frail | 41 | (5.0) | 13 | (3.7) | 28 | (6.2) | 0.422 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| FI categorizeda older individuals (≥65 yrs) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Relatively fit | 64 | (9.6) | 21 | (7.7) | 43 | (10.8) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Less fit | 235 | (35.1) | 106 | (39.3) | 129 | (32.3) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Least fit | 246 | (36.7) | 109 | (40.4) | 137 | (34.3) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Frail | 125 | (18.7) | 34 | (12.6) | 91 | (22.8) | 0.003 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| All-cause mortality | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Died during follow-upa | 975 | (66.1) | 432 | (69.3) | 543 | (63.6) | 0.021 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Median time to death | 14.8 | (12.8) | 13.7 | (13.0) | 15.5 | (12.8) | 0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cancer mortality | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Died during follow-upa | 232 | (15.7) | 143 | (23.0) | 89 | (10.4) | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Median time to death | 12.2 | (11.4) | 11.5 | (12.7) | 13.1 | (9.6) | 0.223 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CVD mortality | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Died during follow-upa | 347 | (23.5) | 160 | (25.7) | 187 | (21.9) | 0.090 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Median time to death | 13.7 | (12.3) | 13.2 | (12.7) | 14.6 | (12.0) | 0.081 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Dementia mortality | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Died during follow-upa | 78 | (5.3) | 20 | (3.2) | 58 | (6.8) | 0.002 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Median time to death | 19.0 | (7.8) | 19.4 | (6.1) | 18.0 | (9.0) | 0.672 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Other causes of mortality | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Died during follow-upa | 258 | (17.5) | 89 | (14.3) | 169 | (19.8) | 0.006 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Median time to death | 14.1 | (11.7) | 14.1 | (11.2) | 13.6 | (11.8) | 0.743 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Note: n=1442 for information on BMI, n=1460 for smoking status and n=1386 for education. The follow-up time was 30 years for all-cause mortality and 27 years for cause-specific mortality. Other causes of mortality include those not dying of cancer, CVD or dementia. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| aValues are n (%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| *p for difference between men and women, Mann-Whitney’s test for median values and χ2 -test for dichotomous variables | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Abbreviations: BMI, body mass index; CVD, cardiovascular disease; FI, frailty index; IQR, interquartile range | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
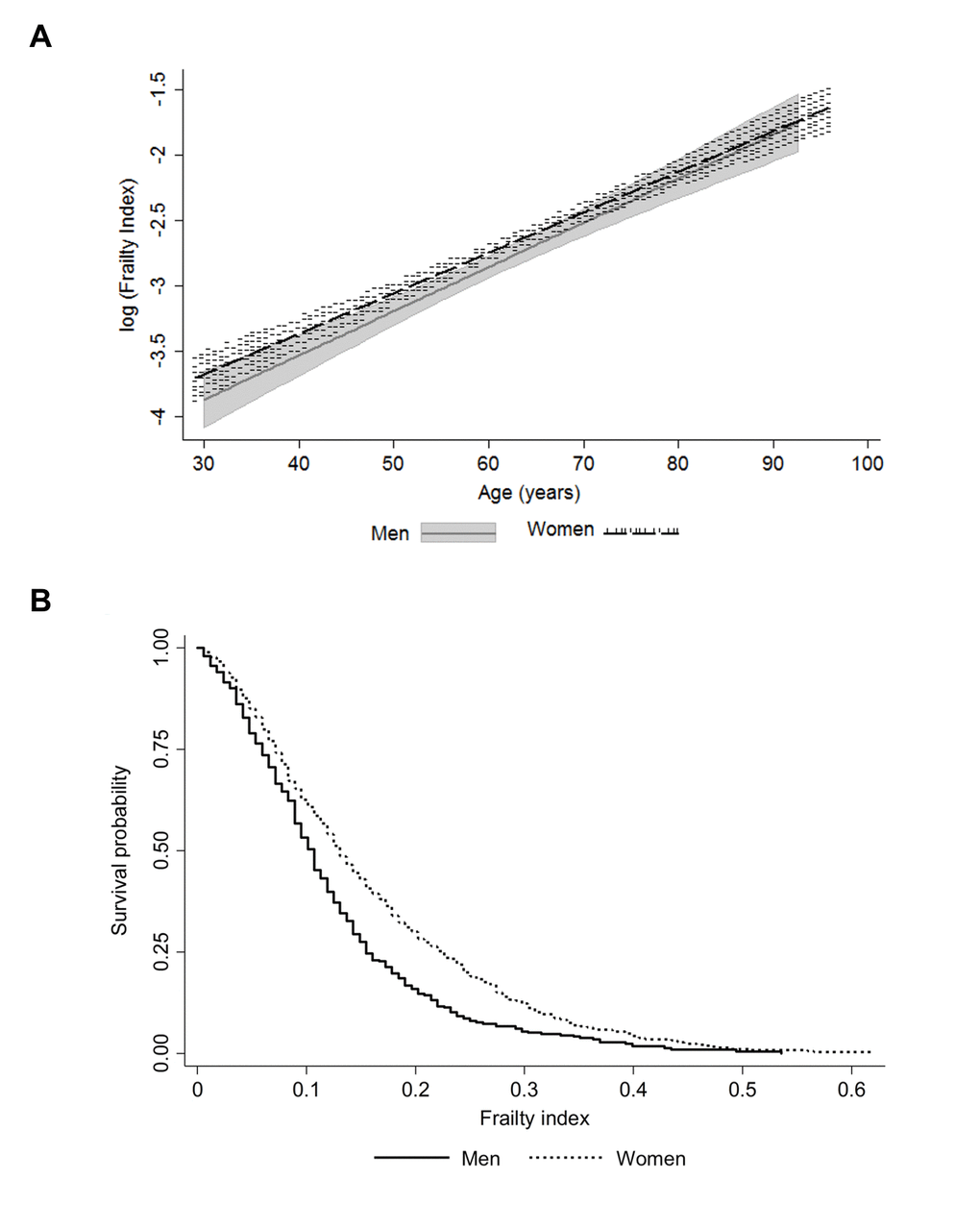
Figure 1. Association of the frailty index with age (a); the shaded areas around the lines represent the 95% confidence intervals for the mean) and mortality by sex (b).
All-cause mortality
Men had a higher all-cause mortality rate and a lower median time to death than women during the 30-year follow-up (Table 1). The categorized FI levels demonstrated a dose-response increase in mortality risk with increased frailty in both men and women (SSupplementary Figure 2). Of the tested variables (age, FI, education, smoking and BMI) only age, smoking and FI remained significantly associated with mortality in the multivariate Cox models for men and women (Table 2). For BMI, only a linear association with mortality was considered, as no sign of an existence of a U-shaped relationship was observed. This was verified as the -2 log likelihood between the Cox regression model with the linear term of BMI and the model with both the linear term and the quadratic term of BMI was non-significant (p=0.128). When the models were further stratified by age, the associations between FI and mortality were similar in all but the older men where the association was non-significant (Table 2).
Table 2. Cox regression for all-cause mortality with a 30-year follow-up. Age, smoking and FI were all assessedanalyzed in the same model.
| All | Young (<65 years) | Old (≥65 years) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| HR | 95%CI | p | HR | 95%CI | p | HR | 95%CI | p | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Men | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age | 1.12 | 1.11-1.14 | <0.001 | 1.12 | 1.09-1.15 | <0.001 | 1.11 | 1.08-1.13 | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Smoking | 1.23 | 1.10-1.38 | <0.001 | 1.20 | 0.99-1.44 | 0.055 | 1.26 | 1.09-1.46 | 0.002 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| FI | 1.04 | 1.01-1.07 | 0.008 | 1.05a | 1.01-1.10 | 0.021 | 1.03 | 0.99-1.07 | 0.097 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Women | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age | 1.13 | 1.12-1.15 | <0.001 | 1.13 | 1.10-1.16 | <0.001 | 1.14 | 1.10-1.17 | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Smoking | 1.20 | 1.04-1.37 | 0.011 | 1.31 | 1.09-1.58 | 0.004 | 1.08 | 0.88-1.31 | 0.456 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| FI | 1.08a | 1.06-1.11 | <0.001 | 1.11 | 1.07-1.17 | <0.001 | 1.07a | 1.04-1.10 | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Note: Numbers of deaths/individuals in each model were as follows: all men 430/620; young men 166/352; old men 264/268; all women 532/840; young women 156/452; old women 376/388. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| aProportional hazards assumption not met: a time-varying coefficient produced and presented in Supplementary Figure 3. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Abbreviations: CI, confidence interval; FI, frailty index; HR, hazard ratio | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
The Schoenfeld and scaled Schoenfeld residual plots with the time-varying coefficients for FI from the models where FI violated the PH assumption (younger men, all women and older women) are presented in Supplementary Figure 3. Overall, the violations were mild; no clear patterns creating the non-zero slopes were observed and the time-varying coefficients indicated 0.4-0.8% decrease in the HR for FI per year. Hence the HR for FI in these models is interpreted as a time-averaged effect.
Cause-specific mortality
Cause-specific mortality rates and median times to death during the 27-year follow-up are presented in Table 1. The Kaplan-Meier survivor function plots for the competing risks and the median FI in each group are presented in Figure 2. The results of the age and smoking status-adjusted cause-specific hazard (CHR) and sub-distribution hazard (SHR) models are presented in Table 3. In women, the CHR models demonstrated that FI predicted deaths due to cancer (HR 1.06, 95%CI 1.01-1.12), CVD (HR 1.13, 95%CI 1.09-1.17) and other causes (HR 1.07, 95%CI 1.03-1.12). In the Cox models run as a sensitivity analysis for the consensus classifications for CVD and cancer deaths, the HRs for the FI remained essentially the same (cancer mortality HR 1.06, 95%CI 1.00-1.13, p=0.05; CVD mortality HR 1.12, 95%CI 1.08-1.16), yet the former association was attenuated to borderline significance. Adjusting the CVD mortality model for CVD status at baseline did not change the results for FI (HR 1.11, 95%CI 1.06-1.16), whereas adjusting the cancer mortality model for cancer diagnosis at study baseline attenuated the FI’s association towards null (HR 1.05, 95%CI 1.00-1.11, p=0.072). In the SHR models for women, the association with CVD mortality was significant (SHR 1.07, 95%CI 1.02-1.11) and a significant inverse association was observed with dementia mortality (SHR 0.87, 95%CI 0.80-0.96). In men, the CHR models demonstrated that FI predicted only other-cause mortality (HR 1.08, 95%CI 1.02-1.14) whereas the SHR models demonstrated no significant associations.
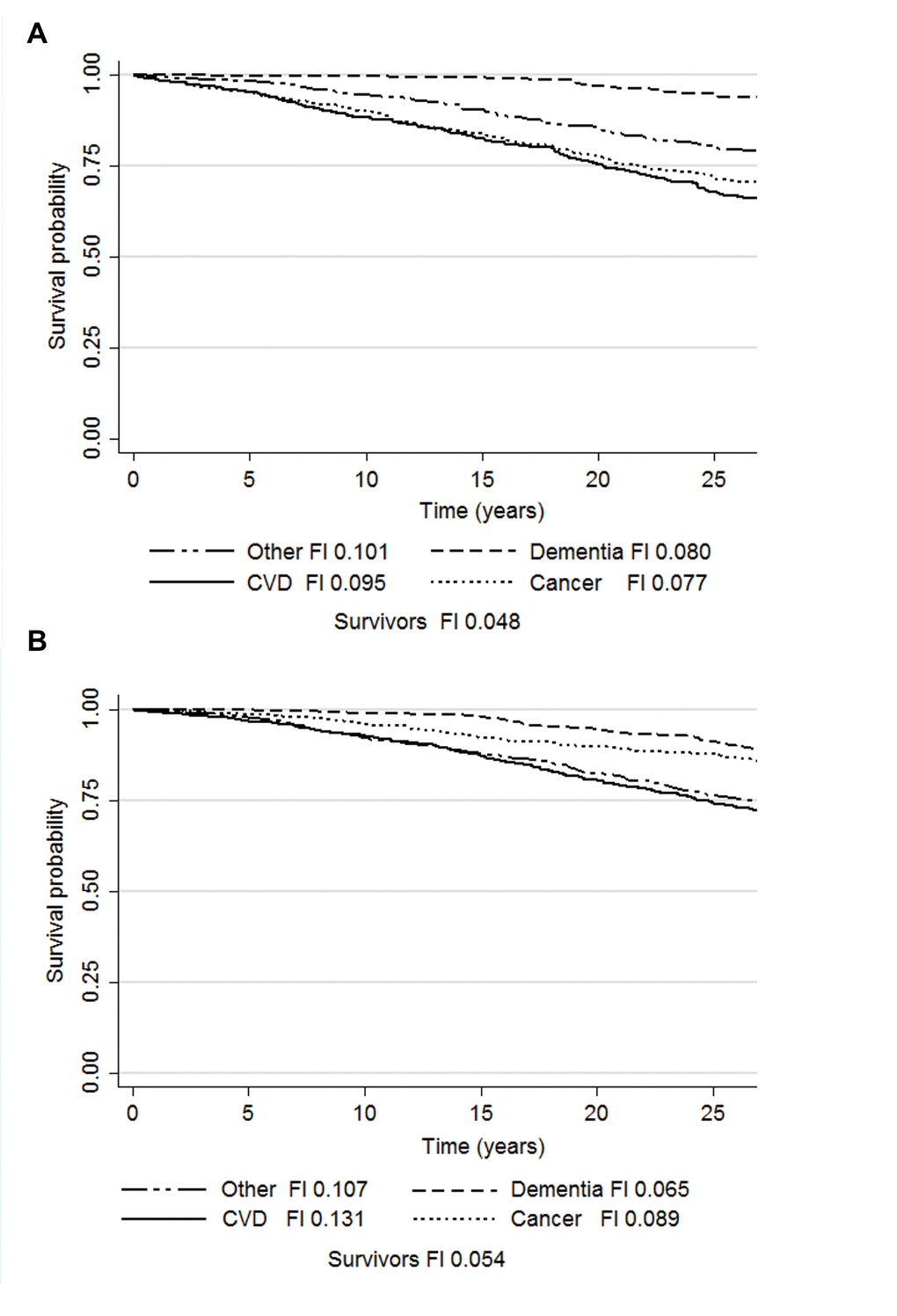
Figure 2. Kaplan-Meier survival probabilities and median frailty index levels according to the causes of death in men (a) and women (b). Abbreviations: CVD, cardiovascular disease; FI, frailty index.
Table 3. Cause-specific mortality with a 27-year follow-up. Age, smoking and FI were all analyzed in the same model.
| Cause-specific hazards | Subdistribution hazards | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| HR | 95%CI | p | SHR | 95%CI | p | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Men | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cancer mortality | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age | 1.07 | 1.05-1.09 | <0.001 | 1.02 | 1.01-1.04 | 0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Smoking | 1.19 | 0.98-1.45 | 0.087 | 1.10 | 0.91-1.34 | 0.325 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| FI | 1.03 | 0.97-1.09 | 0.357 | 0.99 | 0.91-1.06 | 0.769 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CVD mortality | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age | 1.15 | 1.12-1.18 | <0.001 | 1.08 | 1.06-1.09 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Smoking | 1.18 | 1.04-1.37 | 0.065 | 0.97 | 0.80-1.19 | 0.777 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| FI | 1.03 | 0.99-1.08 | 0.158 | 0.99 | 0.94-1.04 | 0.759 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Dementia mortality | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age | 1.25 | 1.17-1.33 | <0.001 | 1.05 | 1.02-1.08 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Smoking | 0.89 | 0.49-1.61 | 0.699 | 0.71 | 0.41-1.23 | 0.224 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| FI | 1.06 | 0.91-1.23 | 0.467 | 0.97 | 0.83-1.13 | 0.672 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Other causes | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age | 1.16 | 1.12-1.19 | <0.001 | 1.06 | 1.04-1.08 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Smoking | 1.45 | 1.15-1.80 | 0.004 | 1.17 | 0.92-1.50 | 0.198 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| FI | 1.08 | 1.02-1.14 | 0.005 | 1.04 | 0.98-1.11 | 0.178 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Women | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cancer mortality | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age | 1.08 | 1.06-1.11 | <0.001 | 1.03 | 1.02-1.05 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Smoking | 1.31 | 0.98-1.75 | 0.067 | 1.20 | 0.89-1.62 | 0.232 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| FI | 1.06 | 1.01-1.12 | 0.031 | 1.00 | 0.95-1.05 | 0.866 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CVD mortality | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age | 1.14 | 1.11-1.16 | <0.001 | 1.06 | 1.04-1.07 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Smoking | 1.06 | 0.98-1.75 | 0.648 | 0.94 | 0.72-1.23 | 0.640 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| FI | 1.13 | 1.09-1.17 | <0.001 | 1.07a | 1.02-1.11 | 0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Dementia mortality | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age | 1.18 | 1.14-1.22 | <0.001 | 1.06 | 1.04-1.08 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Smoking | 1.23 | 0.82-1.82 | 0.315 | 0.96 | 0.64-1.44 | 0.855 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| FI | 0.94 | 0.85-1.04 | 0.247 | 0.87 | 0.80-0.96 | 0.004 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Other causes | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age | 1.15 | 1.12-1.17 | <0.001 | 1.07 | 1.05-1.09 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Smoking | 1.37 | 1.09-1.72 | 0.007 | 1.19 | 0.95-1.49 | 0.136 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| FI | 1.07 | 1.03-1.12 | 0.001 | 0.99 | 0.95-1.03 | 0.725 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| aProportional hazards assumption not met: HR for FI*time interaction=0.988 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Abbreviations: CI, confidence interval; CVD, cardiovascular disease; FI, frailty index; HR, hazard ratio | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Discussion
In this study, we created a 42-item Rockwood FI in a population-based Swedish cohort and validated it by showing that its distribution and associations with age and sex are in agreement with those reported previously [6,8,9,16]. The categorized FI also demonstrated a dose-response relationship with mortality, in keeping with the observations by Rockwood et al. (2011) [16]. An increase in the FI, when treated as a continuous variable, was directly associated with all-cause mortality throughout the 30-year follow-up. The association was independent of age, smoking status, education and BMI. Stratification by sex and into younger (<65 years) and older (≥65 years) ages revealed that the association was strongest among younger women where accumulation of one deficit was associated with an 11% increase in the mortality risk. Among older women, the corresponding increase in the risk was 7%. In the younger men accumulation of one deficit was associated with a 5% increase in the mortality risk, whereas among older men the association was non-significant. The two approaches taken to analyze the relationships between FI and cause-specific mortality, the CHR and SHR, unequivocally demonstrated that higher FI is associated with CVD mortality in women. That is, FI predicts CVD mortality in the presence of competing risks and regardless of the nature of the associations between the FI and the other causes of death. A suggestive association was observed for cancer mortality in women as the association was significant only in the CHR model. In men, the FI was predictive only of other-cause mortality and the finding was restricted to the CHR model.
Our results add to the understanding of the significance of frailty in several aspects. We had an exceptionally long follow-up and observed that the frailty-mortality relationship extends up to 30 years from baseline. In addition, half our sample consisted of younger individuals who are understudied for frailty in comparison to old individuals. We demonstrated that FI was a strong predictor of mortality especially in younger women, where the risk conferred by the increase in frailty was greater than that among the older. This finding is in line with previous results [16] that demonstrated a similar phenomenon in an age, sex and education-adjusted model of 12-year mortality. Hence, our results further verify the usefulness of FI in early risk stratification. The reason why the association between FI and mortality was not statistically significant in older men is unknown, yet we identified a data-driven explanation for it. CVD was the major cause of mortality among older men and as the FI did not predict CVD deaths in men, the association with all-cause mortality was hence likely attenuated. Similar findings have been reported in a Finnish study where the association between the Fried frailty phenotype and 4-year mortality was no longer significant in old men after adjusting for covariates such as smoking, education and functional capacity [17]. Nevertheless, it should be noted that both the FI and the frailty phenotype have been found to predict mortality in older men in several studies [2,3].
Our findings are in accordance with a recent meta-analysis reporting that both frailty and pre-frailty predict CVD-related mortality in individuals aged 65 years and older [11]. Sex-differences were not, however, specifically addressed in that work. Related findings have been presented in patients with established CVD or after an acute cardiovascular event where the presence of frailty – defined using different indicators – has been an independent predictor of all-cause mortality [18,19]. Although the individuals in these studies have generally been older than in our study and the follow-ups have been shorter, our findings are given some support by an Icelandic study that demonstrated a predictive validity of frailty on incident CVD, including deaths due to cardiovascular causes [12]. The association was stronger in women than in men and independent of subclinical atherosclerotic manifestations. Because frailty and subclinical CVD may share common biological pathways, a causal relationship is unlikely. Nevertheless, a mutual reciprocal relationship has been proposed; frailty may lead to CVD and CVD can also lead to frailty [19]. Notwithstanding that both scenarios are possible, our results provide new insight to the matter by demonstrating that FI predicts CVD mortality, regardless of CVD status at baseline, age and smoking. This observation may also be indicative of the type of vulnerability that the FI reflects in women. Given that our follow-up for cause-specific mortality spanned 27 years and the median time to CVD death in women was 14.6 years, our observation is of interest when considering FI as an early risk assessment tool.
The results on cancer mortality in women warrant further confirmation as the association was significant only in the CHR model. This association was also attenuated after adjusting for cancer diagnosis at study baseline, indicating that it was largely driven by those individuals who already had cancer. The SHR for cancer-specific death in women was 1.00, suggesting that there is no cause-specific association between FI and cancer death. The cause-specific risk is likely largely overruled by the absolute risk observed between FI and CVD deaths. The CHR model result nevertheless suggests that there is an instantaneous risk between increased frailty and cancer mortality. Although this an interesting finding that warrants further investigation, we wish to caution against too strong interpretation asthe number of cancer deaths in women was rather small. It is nevertheless noteworthy that despite the higher number of cancer deaths among men, no associations were observed, suggesting that the potential cancer mortality-related vulnerability reflected by the FI is specific to women. Our results in women align with the observations in a study of breast cancer patients where frailty was shown to be a predictor of both all-cause and breast cancer mortality irrespective of treatment differences between the robust and the frail individuals [20]. A systematic review has also demonstrated that frailty is an independent predictor of up to 10-year all-cause mortality in patients with various cancers [14]. Together with these observations, our results give support to the notion that frailty can predispose to cancer-related vulnerability, possibly through treatment intolerance and postoperative complications [14].
The finding of a lower FI being predictive of dementia mortality in the SHR model may be attributable to the fact that those who died of dementia had lower FI levels at baseline and longer median times to death compared to the other causes. The SHR model interprets this as an association between lower FI and dementia mortality, as in the risk set there is a large proportion of individuals who had already died due to the other causes and had higher FI values. Furthermore, there is also a biological way to interpret the result; those who died of dementia can be considered as physically healthy agers as they escaped deaths due to other causes at midlife and younger old ages and survived up to very old ages. In this sense, lower FI relates to healthy aging and resilience. Lastly, the association may also reflect a bias in reporting dementia as a cause of death; it has become more common nowadays, so those who have dementia recorded as a cause of death are by necessity older. However, as the association in the CHR model was non-significant, our data do not allow conclusive inferences on this relationship. However, we can conversely argue that the FI does not capture the vulnerability to dementia death in community settings, yet in older ages frailty has been shown to be predictive of dementia diagnosis [13].
The only model where the FI conferred a similar increased risk in men and women was the CHR approach on other-cause mortality. In fact, the other-cause mortality was the only model where FI was significant in men. Moreover, the median FIs at baseline showed greater differences across the causes of death i.e., CVD, cancer, dementia and other, in women than in men (Figure 2). Hence, it appears that the FI-related mortality risk is distributed evenly between the different causes in men, whereas in women it is largely confined to CVD deaths. FI can thus be considered to represent a different type of vulnerability in men and women – a finding that may add to the understanding of the frailty paradox with regards to the greater overall risk of death in men across all levels of frailty [21].
In conclusion, our results demonstrate that that FI is a prognostic survivorship factor already at younger ages (<65 years) and that the association is stronger in women. When different causes of death are considered, FI can best identify women at risk of CVD death and to a lesser degree the risk of other-cause death. In men, FI can be understood as more of a general vulnerability factor as the risk appears to be evenly distributed between the causes and confined to the other-cause mortality. However, more research on this topic is required as our population was rather small for a cause-specific mortality analysis. Moreover, this was the first study to assess the predictive value of FI on mortality in a competing-risks setting.
Materials and Methods
Participants
The Swedish Adoption/Twin Study of Aging (SATSA) [22] is a longitudinal population-based cohort of same-sexed twins that is drawn from the Swedish Twin Registry (STR) [23]. SATSA was initiated in 1984 (n=2018) and included both mailed questionnaires and in-person testing waves. The present study involves both single and pair responders (n=1477; 623 men, 854 women; aged 29-95 years) who returned the second questionnaire sent out in 1987. The questionnaires assess physical and mental health status, activities of daily living, wellbeing, health-related behavior with respect to smoking, alcohol consumption and use of prescribed drugs, family and social environments and personality dimensions (as documented in [24]). The study has received ethical approval from the Regional Ethics Review Board, Stockholm.
Assessment of frailty
We constructed the FI in SATSA based on the self-reported questionnaire data using the Rockwood deficit accumulation model according to the standard procedure [6,16]. The Rockwood FI uses health deficits that can be defined as symptoms, signs, disabilities and diseases that cover a wide range of systems, associate with health status and have a prevalence of ≥1% in the study population. The selected 42 items and scoring of the deficits are described in the Supplementary Table 1. All items had ≤10% missing data points and only individuals with ≤20% missing answers across the 42 FI items were included. After that, the patterns of missingness were examined, missing data were replaced by multiple imputation (MI) and a sensitivity analysis for the imputed data was performed (Supplementary Methods). Each individual’s FI was assessed by counting the number of deficits and dividing the count by the total number of deficits considered. The validity of the FI was assessed by examining its distribution and associations with age and sex. In addition, we tested whether at each given level of frailty, men were more likely to die than women. As further validation to test for a dose-response relationship with the FI and mortality, we subdivided the FI into four categories according to Rockwood et al. (2011): relatively fit (FI≤0.03), less fit (0.03< FI≤0.10), least fit (0.10
Mortality
All-cause mortality data, including dates of death, were obtained from linkages of the STR to Swedish national registers through the personal identification number assigned to all residents. The all-cause mortality data used in this study were updated on April 30, 2017, yielding a 30-year follow-up. Cause-specific mortality data were obtained from the Causes of Death Register (CDR) where the latest update was on December 31, 2014, producing a 27-year follow-up period. The CDR records include information about the underlying and contributory causes of death for all individuals who were registered as Swedish residents in the year of their death. Causes of death are recorded using the International Classification of Diseases (ICD) codes, with ICD-7 used prior to 1969, ICD-8 between 1969 and 1986, ICD-9 from 1987 until 1996, and ICD-10 from 1997 and onwards. We considered cancer, CVD (including stroke) and dementia as the specific causes of death and other causes not falling into these categories were denoted as “other”. The ICD codes included in each cause are presented in the Supplementary Table 2. When more than one cause of death was recorded, a consensus classification was used (Supplementary Table 3) and a sensitivity analysis was performed (Supplementary Methods). The causes of death classified as the “other” are shown in Supplementary Table 4.
Covariates
Age at baseline, sex, education, smoking status and BMI were considered as covariates in the survival analyses. Education, smoking status and BMI were assessed from questionnaire data. Education level was classified as 1=primary education, 2=lower secondary or vocational education, 3=upper secondary education and 4=tertiary education. Smoking status was classified as non-smoker (reference category), ex-smoker and current smoker. BMI was calculated as weight divided by height squared (kg/m2).
Statistical analyses
The associations between the study variables and sex were tested using the Mann-Whitney test or χ2 -test when appropriate. As the FI had a skewed distribution (Supplementary Figure 1), the association between log(FI) and age was tested using Pearson’s rho. Kaplan-Meier survival plots were used to assess differences in mortality rates by sex and FI categories. Age, sex, FI (sum of the deficits), BMI, education (primary education as reference category) and smoking status (non-smokers as reference category) were first tested for their association with mortality in univariate Cox regression models for the whole study population. Significant covariates were then tested in multivariate Cox models. Those covariates that remained significantly associated with all-cause mortality in the whole population were used as the final model covariates. The final models were stratified by sex and further subdivided into the younger and older using the standard definition of old age, ≥65 years, as the cut point. In all survival models, the sum of the FI deficits (of the 42 considered) was used to obtain hazard HRs that are interpretable as per increase by one FI deficit. In other statistical tests the sum of the deficits divided by 42 was used. Violation of the proportional hazards (PH) assumption was tested by including an interaction term with time with each variable and further inspected using the Schoenfeld and scaled Schoenfeld residual plots. If violation was observed, a time-varying coefficient (HR) was produced for that covariate.
To rigorously assess the relationship between FI and cause-specific mortality, we took two different approaches: the CHR using the conventional Cox regression and a cumulative incidence function (CIF) using the SHR model proposed by Fine and Fray [25] (see Supplementary Methods for details). In both approaches, deaths due to cancer, CVD, dementia and other causes were considered as the competing risks (mutually exclusive failures). In the former approach, all the competing events – in our case the deaths due to other causes than the one investigated – are censored at the time of the competing event and are thus removed from the risk set. In the SHR model, those experiencing a competing risk are kept within the risk set which increases the number of individuals in the risk set at the occurrence of each competing event. In other words, the standard survival model (CHR) corresponds to a hypothetical scenario where the only way to die is due to the cause under investigation. If the other causes of death are correlated with the one under investigation, the estimates may become biased and inferences invalid. Hence, the two approaches may produce different results and it has been advocated that both methods should be used side-by-side when assessingcompeting risks [26]. The SRHs are commonly attenuated compared to HRs derived from the CHR model and especially if the covariate of interest has also an effect on the competing risk(s) [27,28]. Differences also arise when the competing risks precede or outnumber the risk of interest.
If a significant association was found, an additional analysis performed by adjusting for the diagnosis of the given disease at the study baseline (see Supplementary Methods). In all models, clustering of the data in twin pairs was accounted for by computing cluster robust standard errors for the coefficients. P-values <0.05 were considered statistically significant. Statistical analyses were performed using Stata version 14.1 (College Station, TX: StataCorp LP) and SPSS version 24.0 (Armonk, NY: IBM Corp).
Supplementary Materials
Acknowledgements
We thank Professor Chandra Reynolds and Professor Deborah Finkel for careful reading of the manuscript and helpful comments. We also thank Professor Kenneth Rockwood for his valuable input in creating the frailty index in our sample.
Conflicts of Interest
The authors have no conflicts of interest to declare.
Funding
The SATSA study was supported by NIH grants R01 AG04563, AG10175, AG028555, the MacArthur Foundation Research Network on Successful Aging, the Swedish Council for Working Life and Social Research (FAS/FORTE) (97:0147:1B, 2009-0795), and the Swedish Research Council (825-2007-7460, 825-2009-6141, 521-2013-8689 and 2015-03255). This study is supported by the JPND/Swedish Research Council (2015-06796), FORTE (2013-2292), the Loo & Hans Osterman Foundation, the Foundation for Geriatric Diseases, the Magnus Bergwall Foundation, and the Strategic Research Program in Epidemiology at Karolinska Institutet.
References
- 1. Clegg A, Young J, Iliffe S, Rikkert MO, Rockwood K. Frailty in elderly people. Lancet. 2013; 381:752–62. https://doi.org/10.1016/S0140-6736(12)62167-9 [PubMed]
- 2. Shamliyan T, Talley KM, Ramakrishnan R, Kane RL. Association of frailty with survival: a systematic literature review. Ageing Res Rev. 2013; 12:719–36. https://doi.org/10.1016/j.arr.2012.03.001 [PubMed]
- 3. Vermeiren S, Vella-Azzopardi R, Beckwée D, Habbig AK, Scafoglieri A, Jansen B, Bautmans I, Bautmans I, Verté D, Beyer I, Petrovic M, De Donder L, Kardol T, et al, and Gerontopole Brussels Study group. Frailty and the Prediction of Negative Health Outcomes: A Meta-Analysis. J Am Med Dir Assoc. 2016; 17:1163.e1–17. https://doi.org/10.1016/j.jamda.2016.09.010 [PubMed]
- 4. Lin HS, Watts JN, Peel NM, Hubbard RE. Frailty and post-operative outcomes in older surgical patients: a systematic review. BMC Geriatr. 2016; 16:157. https://doi.org/10.1186/s12877-016-0329-8 [PubMed]
- 5. Fried LP, Tangen CM, Walston J, Newman AB, Hirsch C, Gottdiener J, Seeman T, Tracy R, Kop WJ, Burke G, McBurnie MA, and Cardiovascular Health Study Collaborative Research Group. Frailty in older adults: evidence for a phenotype. J Gerontol A Biol Sci Med Sci. 2001; 56:M146–56. https://doi.org/10.1093/gerona/56.3.M146 [PubMed]
- 6. Searle SD, Mitnitski A, Gahbauer EA, Gill TM, Rockwood K. A standard procedure for creating a frailty index. BMC Geriatr. 2008; 8:24. https://doi.org/10.1186/1471-2318-8-24 [PubMed]
- 7. Cigolle CT, Ofstedal MB, Tian Z, Blaum CS. Comparing models of frailty: the Health and Retirement Study. J Am Geriatr Soc. 2009; 57:830–39. https://doi.org/10.1111/j.1532-5415.2009.02225.x [PubMed]
- 8. Kulminski AM, Ukraintseva SV, Kulminskaya IV, Arbeev KG, Land K, Yashin AI. Cumulative deficits better characterize susceptibility to death in elderly people than phenotypic frailty: lessons from the Cardiovascular Health Study. J Am Geriatr Soc. 2008; 56:898–903. https://doi.org/10.1111/j.1532-5415.2008.01656.x [PubMed]
- 9. Theou O, Brothers TD, Mitnitski A, Rockwood K. Operationalization of frailty using eight commonly used scales and comparison of their ability to predict all-cause mortality. J Am Geriatr Soc. 2013; 61:1537–51. https://doi.org/10.1111/jgs.12420 [PubMed]
- 10. Morley JE, Vellas B, van Kan GA, Anker SD, Bauer JM, Bernabei R, Cesari M, Chumlea WC, Doehner W, Evans J, Fried LP, Guralnik JM, Katz PR, et al. Frailty consensus: a call to action. J Am Med Dir Assoc. 2013; 14:392–97. https://doi.org/10.1016/j.jamda.2013.03.022 [PubMed]
- 11. Veronese N, Cereda E, Stubbs B, Solmi M, Luchini C, Manzato E, Sergi G, Manu P, Harris T, Fontana L, Strandberg T, Amieva H, Dumurgier J, et al. Risk of cardiovascular disease morbidity and mortality in frail and pre-frail older adults: results from a meta-analysis and exploratory meta-regression analysis. Ageing Res Rev. 2017; 35:63–73. https://doi.org/10.1016/j.arr.2017.01.003 [PubMed]
- 12. Veronese N, Sigeirsdottir K, Eiriksdottir G, Marques EA, Chalhoub D, Phillips CL, Launer LJ, Maggi S, Gudnason V, Harris TB. Frailty and Risk of Cardiovascular Diseases in Older Persons: The Age, Gene/Environment Susceptibility-Reykjavik Study. Rejuvenation Res. 2017rej.2016.1905. https://doi.org/10.1089/rej.2016.1905 [PubMed]
- 13. Kojima G, Taniguchi Y, Iliffe S, Walters K. Frailty as a Predictor of Alzheimer Disease, Vascular Dementia, and All Dementia Among Community-Dwelling Older People: A Systematic Review and Meta-Analysis. J Am Med Dir Assoc. 2016; 17:881–88. https://doi.org/10.1016/j.jamda.2016.05.013 [PubMed]
- 14. Handforth C, Clegg A, Young C, Simpkins S, Seymour MT, Selby PJ, Young J. The prevalence and outcomes of frailty in older cancer patients: a systematic review. Ann Oncol. 2015; 26:1091–101. https://doi.org/10.1093/annonc/mdu540 [PubMed]
- 15. Rockwood K, Mitnitski A. Limits to deficit accumulation in elderly people. Mech Ageing Dev. 2006; 127:494–96. https://doi.org/10.1016/j.mad.2006.01.002 [PubMed]
- 16. Rockwood K, Song X, Mitnitski A. Changes in relative fitness and frailty across the adult lifespan: evidence from the Canadian National Population Health Survey. CMAJ. 2011; 183:E487–94. https://doi.org/10.1503/cmaj.101271 [PubMed]
- 17. Kulmala J, Nykänen I, Hartikainen S. Frailty as a predictor of all-cause mortality in older men and women. Geriatr Gerontol Int. 2014; 14:899–905. https://doi.org/10.1111/ggi.12190 [PubMed]
- 18. Ekerstad N, Swahn E, Janzon M, Alfredsson J, Löfmark R, Lindenberger M, Andersson D, Carlsson P. Frailty is independently associated with 1-year mortality for elderly patients with non-ST-segment elevation myocardial infarction. Eur J Prev Cardiol. 2014; 21:1216–24. https://doi.org/10.1177/2047487313490257 [PubMed]
- 19. Afilalo J, Karunananthan S, Eisenberg MJ, Alexander KP, Bergman H. Role of frailty in patients with cardiovascular disease. Am J Cardiol. 2009; 103:1616–21. https://doi.org/10.1016/j.amjcard.2009.01.375 [PubMed]
- 20. Mandelblatt JS, Cai L, Luta G, Kimmick G, Clapp J, Isaacs C, Pitcher B, Barry W, Winer E, Sugarman S, Hudis C, Muss H, Cohen HJ, Hurria A. Frailty and long-term mortality of older breast cancer patients: CALGB 369901 (Alliance). Breast Cancer Res Treat. 2017; 164:107–17. https://doi.org/10.1007/s10549-017-4222-8 [PubMed]
- 21. Gordon EH, Peel NM, Samanta M, Theou O, Howlett SE, Hubbard RE. Sex differences in frailty: A systematic review and meta-analysis. Exp Gerontol. 2017; 89:30–40. https://doi.org/10.1016/j.exger.2016.12.021 [PubMed]
- 22. Finkel D, Pedersen NL. Processing Speed and Longitudinal Trajectories of Change for Cognitive Abilities: The Swedish Adoption/Twin Study of Aging. Neuropsychol Dev Cogn B Aging Neuropsychol Cogn. 2004; 11:325–45. https://doi.org/10.1080/13825580490511152
- 23. Magnusson PK, Almqvist C, Rahman I, Ganna A, Viktorin A, Walum H, Halldner L, Lundström S, Ullén F, Långström N, Larsson H, Nyman A, Gumpert CH, et al. The Swedish Twin Registry: establishment of a biobank and other recent developments. Twin Res Hum Genet. 2013; 16:317–29. https://doi.org/10.1017/thg.2012.104 [PubMed]
- 24. Pedersen NL, Swedish Adoption/Twin Study on Aging (SATSA), 1984, 1987, 1990, 1993, 2004, 2007, and 2010. 2015, Inter-university Consortium for Political and Social Research. .
- 25. Fine JP, Gray RJ. A Proportional Hazards Model for the Subdistribution of a Competing Risk. J Am Stat Assoc. 1999; 94:496–509. https://doi.org/10.1080/01621459.1999.10474144
- 26. Latouche A, Allignol A, Beyersmann J, Labopin M, Fine JP. A competing risks analysis should report results on all cause-specific hazards and cumulative incidence functions. J Clin Epidemiol. 2013; 66:648–53. https://doi.org/10.1016/j.jclinepi.2012.09.017 [PubMed]
- 27. Dignam JJ, Zhang Q, Kocherginsky M. The use and interpretation of competing risks regression models. Clin Cancer Res. 2012; 18:2301–08. https://doi.org/10.1158/1078-0432.CCR-11-2097 [PubMed]
- 28. Austin PC, Lee DS, Fine JP. Introduction to the Analysis of Survival Data in the Presence of Competing Risks. Circulation. 2016; 133:601–09. https://doi.org/10.1161/CIRCULATIONAHA.115.017719 [PubMed]



