Introduction
Humans are growing older as a result of improvements in public health, producing an era of accelerated aging. Preservation of healthy aging and maintenance of functional ability is increasingly the focus of public health care in older humans [1, 2]. To achieve this goal, an integrated model reflecting the complex and multifaceted aging-related health and functioning based on daily life is highly desired [3, 4]. Rhesus macaques are the most extensively studied nonhuman primate animals exhibiting parallel aging characteristics and a majority of the age-related diseases of humans [5, 6]. Despite these close genetic relationships and the high degree of similarity of aging phenotypes, the repeatability and reproducibility of experimental findings in rhesus macaques has not been satisfactory [3]. One potential reason is the high heterogeneity in individual health and functioning that accumulates from the young-adult age to advanced age [7]. Therefore, although a large variety of scales have been used to assess aging-related health and functioning in humans, a well-accepted and widely used scale is still lacking for the assessment of elderly humans [8, 9]. It is definitely more difficult to develop an aging-related health and functioning scale in rhesus macaques because they are incapable of self-rating or being interviewed.
Although numerous studies have indicated that cardiovascular aging, motor aging [10] and mental aging all show similar declines in macaques [6] and in humans, the aging-related traits and trajectories were different between nonhuman primates and humans. From a biological perspective, arterial stiffness, which is highly prevalent in humans, is rare in aged monkeys with the development of ectasia being common in animals fed a normal diet [11]. Moreover, aging-related biological changes can be distinguished only by advanced techniques and comprehensive examinations. From a psychosocial perspective, it has been difficult to develop an observation-based scale because of the much lower psychosocial performance with a narrow range between the best and the worst levels of performance. Using the cognitive domain of aging-related health as an example, the cognitive capability in adult monkeys was comparable to that in an 8–10 month human infant [11]. Therefore, the largest scaling range of cognitive capability in monkeys was comparable to that from an infant to a newborn in humans. With such a narrow range of variation, daily observation with the naked eye could not make any particular assessment with a high degree of reliability. That is, such confined psychosocial performance of animals attenuated the availability of distinguishable items based on daily observation. To resolve this dilemma, routine studies on aging-related neurological changes have required training of animals over a period of time. This long training period allows numerous external factors to bias learning capability and longitudinal adaptability, greatly attenuating the repeatability and reproducibility of experiments with these nonhuman primate models; however, the potential impacts of such biases have long been neglected.
In the present study, we attempted to develop a rapid, easily operationalized scale based on daily observation to assess aging-related health and functioning in a rhesus macaque group. With high respect to the multidimensional nature of health and functioning, we have incorporated the social domain of health into the present nonhuman primate “society” for better efficacy. We believe that the present scale is valuable not only for human aging research but also for improving the repeatability and reproducibility of studies with nonhuman primate models.
Results
The reliability of the AHFS in rhesus macaques
The reliability was 0.877 (Cronbach’s alpha coefficient), indicating that the items in the scale measure as a latent construct with 87.75% internal consistency. Notably, during the development of the scale, we compared Cronbach’s alpha coefficient by denoting the male or female animals as milder aging (score=1). We found that when the female sex was rated with a milder aging score, higher reliability scores were obtained. Because higher socioeconomic status has been closely correlated with better health during aging in humans [14], we supposed that the male animals with higher levels of social dominance might display better health. However, according to the reliability test, females performed better with slower aging based on the daily life assessment.
To further validate the reliability of the scale, we estimated half-split reliability by randomly splitting the 12 items into two halves, each comprising six items. Half one included four items on dietary uptake, age and sex, and the remaining six items were in half two. The reliability of half one was 0.831, whereas that of half two was 0.687. From these results, we suggested that dietary uptake performance played a substantially independent role in aging-related health and functioning.
The validity of the AHFS in rhesus macaques
To validate the construction of the scale, we conducted an exploratory factor analysis of the 12 items. The adequacy of the scale was calculated by KMO (0.815), which indicated that these items harbor a dimension-reducible construct. We then divided the 12 items into three distinguishable factors using varimax rotation (Figure 2A). According to the results, three clustered factors together explained 76.4% of the variance in aging-related health and daily functioning. We further estimated the contribution to each factor using the “loading” statistic (Figure 2B–2D). Loadings above 0.5 were considered to contribute to a relatively independent role, helping “denoise” the complexity of aging-related health and functioning. Factor 1 comprised six significantly interrelated items, among which volume of dietary uptake explained the highest level of variance of the 12 items in the AHFS.
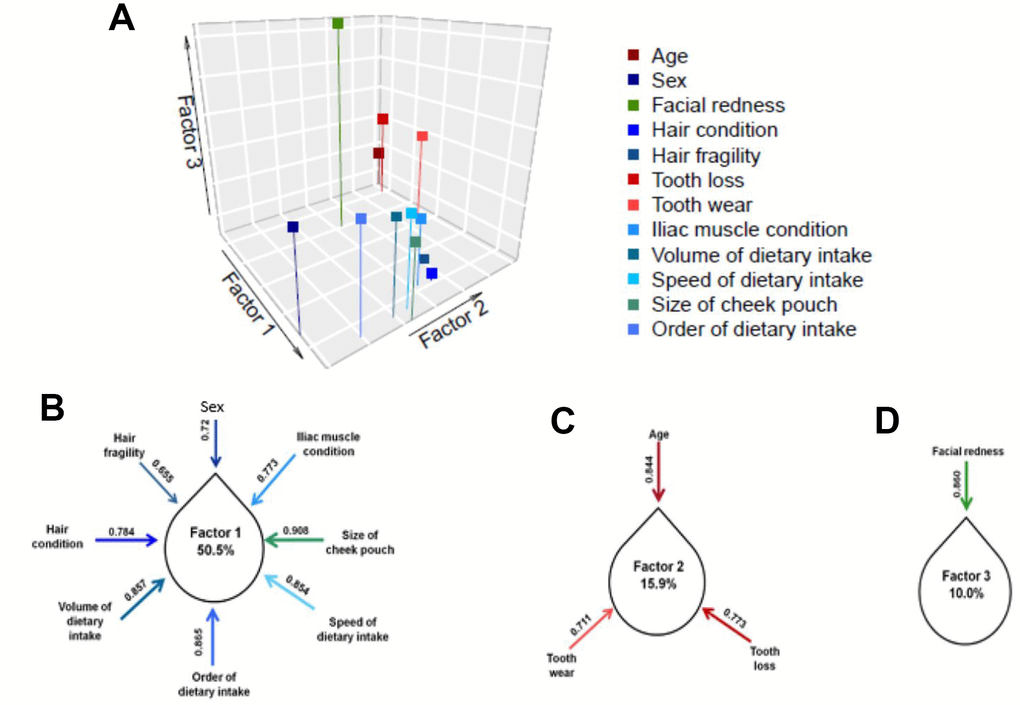
Figure 2. Using varimax rotation, 12 items on aging-related health and functioning yielded three exploratory factors (Factor 1, Factor 2 and Factor 3). The factor loading of each item within the explanatory factor is shown in (B–C). Factor 1, focusing on dietary uptake and physical performance, explained greater than half of the overall variance; Factor 2, focusing on tooth condition, explained 15.9% of the overall variance; and Factor 3, consisting only of perceived age, explained 10% of the overall variance. Notably, each item was considered to significantly contribute to the overall aging-related health and functioning (loading>0.5).
Evaluating the applicability of the AHFS using hierarchical cluster analysis
Hierarchical cluster analysis was conducted to validate the actual applicability of the scale. Individuals’ scores for aging-related health and function were clustered in all subjects as well as within each sex. According to the comparison of the respective presented dendrograms, three clusters were taken, and the optimal number of clusters reflected the severe-aging clustered subgroup, moderate-aging clustered subgroup and mild-aging clustered subgroup. Notably, the average scores of three clustered subgroups displayed significant disparity from each other in all samples, all females and all males (Table 4).
Table 4. Three aging-related clusters in different animal subgroups.
| Subgroups based on clustered analysis | n | Average score | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| All animals* | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Severe-aging subgroup | 4 | 29.500 ± 0.577 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Moderate-aging subgroup | 27 | 20.259 ± 2.536 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Mild-aging subgroup | 26 | 14.231 ± 1.107 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Female animals* | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Severe-aging subgroup | 4 | 22.000 ± 1.414 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Moderate-aging subgroup | 7 | 17.571 ± 0.787 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Mild-aging subgroup | 18 | 14.278 ± 1.074 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Male animals* | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Severe-aging subgroup | 4 | 29.500 ± 0.577 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Moderate-aging subgroup | 18 | 20.445 ± 1.907 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Mild-aging subgroup | 6 | 13.500 ± 0.548 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| * P<0.05 comparing between two clusters | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
The clusters of individual scores in all samples, females and males, were also demonstrated by tree plots (Figure 3). The magnitude of an individual’s score for aging-related health and functioning is represented by the average interval between clusters according to the sequential similarity. In general, the discriminatory capability of the scale validated the applicability of the AHFS as good.
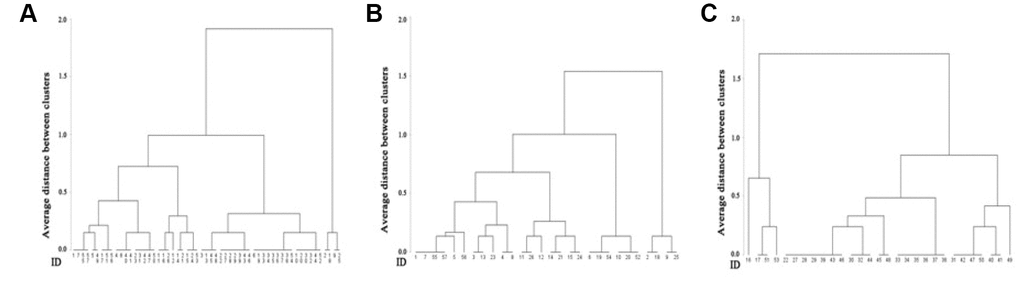
Figure 3. Depicts three cluster trees based on scores for aging-related health and functioning in all subjects. (A) All females (B) and all males (C). The distinct patterns of the three trees indicate that the scale can finely separate distinct samples with respect to similarity
Furthermore, the scores of each item in the three clustered subgroups were also estimated and compared (data not shown). The similar clustered patterns between these composite items and the summed score were revealed, which indicated that the scale could be well separated with respect to the similarity of its composite items.
Discussion
It is more difficult to quantify aspects of the long and complicated aging-related health and functioning in nonhuman primates than in humans because neither self-rating nor interview procedures are possible in nonhuman animals. We constructed a subjective scale based on daily life observations in healthy captive rhesus macaques. It has been documented that rhesus macaques age at approximately 3 times the rate of humans, with puberty occurring between 2.5 and 4.5 years and a median life span of 27 years [19]. Therefore, we included subjects ranging from young-adult age to young-old age to trace the long and subjective healthy aging process. Due to the complexity of aging, aging health assessments or healthy aging measurements are a continuing challenge in humans. We constructed the present scale based on daily life with the aim to easily and rapidly score captive animals with the least invasiveness. With the present scale, the majority of items could be rated noninvasively, with the exception of tooth condition and muscle palpation in males, which required a very short duration of anesthesia. Furthermore, social traits that nonhuman primates express due to their colony lifestyle could present a sophisticated translational model of aging in humans. For the present scale, candidate items were screened using a step-by-step strategy with the consensus of a multidisciplinary panel. The general three-point scale was believed to easily discriminate the scores for each animal with high intrarater agreement. Therefore, we supposed that the present AHFS could characterize overall health and functioning based on daily life. Notably, accurate measurement of aging-related changes is very difficult. For example, motor functioning can be qualified only using complicated digital-based examination [20]. Therefore, this daily life scale contains the notable merit of practicality. The application of the AHFS could help reduce the bias of baseline variation in animal status to produce higher repeatability and reproducibility in experiments.
In the development of the scale, we selected three domains based on daily life activity: perceived aging, musculoskeletal aging and social aging. Perceived age was reflected by three items. The facial dermis in aged rhesus macaques is thin and fragile [6], which results in the easily recognized darkness of the redness on the face, especially in the periorbital region. Therefore, two experts recommended grading the darkness of facial redness in the rhesus macaques. Intriguingly, we found that perceived facial redness alone explained 10% of the variation in overall aging-related health and functioning. Hair condition, tested in a noninvasive manner, was simultaneously rated. It has been documented that hair condition is closely correlated with immune-endocrine imbalance [21–23], increased social stress [22, 23] and malnutrition [24], all of which can accelerate aging-related decreases in health and functioning. Regarding musculoskeletal aging, we selected the iliac muscle mass as the representative muscle to grade aging-related muscle loss. Although rhesus macaques display a process that matches sarcopenia in humans, a duration of approximately nine years is required to observe 20% muscle loss in elderly macaques [16]. Furthermore, apparent muscle loss is not distinguishable until the animal is >25 years old [16]. Therefore, we used three-point scoring to discriminate the chronic loss of iliac muscle mass. Oral health, as an integral component of overall health, is consistently arousing attention in aging research [25]. It is well known that dental hard tissues do not remodel with aging as bones do. Therefore, we rated tooth loss and tooth wear to reflect the continuous loss of dental hard tissues. Aging-related tooth conditions are also affected by microhabitat determinants, including food, dietary properties and behaviors, in rhesus macaques [26]. Therefore, scoring tooth condition can help integrate the assessment of aging-related health and functioning. The largest aspect of daily life we rated in the scale was social aging, which was reflected in the dietary uptake performance or eating behavior. We supposed that these dietary uptake measures indicated not only nutrition uptake but also essential social activity. Here, we averaged four items, which included the volume of dietary uptake, the speed of eating, the size of the cheek pouch and the order of dietary uptake. Notably, the order of dietary uptake was the only independent social variable that directly assessed the effect of social hierarchy on aging-related health and functioning. Practically, the distance between the interested female and the male was measured to better assess the order of dietary uptake during mealtime. We believe that dietary uptake performance can comprehensively reflect the entangled aging-related health and functioning involving physical function, social status and neuromuscular performance. The term “eating capability” has been quantified for understanding the complicated correlation during dietary management in elderly humans [27]. During dietary uptake, neuromuscular orchestration of forelimb use [28], bimanual coordination [29] and orofacial muscle control [27] can be assessed. Regarding the present scale, both the highest correlation and the greatest loading for an exploratory factor were observed in the four eating-related items. In summary, the present scale that demonstrated high intraobserver reliability, well-explained construct validity and good applicability can help captive rhesus macaques become more valuable in the study of aging-related health and functioning. To the best of our knowledge, no scale has been developed on overall aging-related health and functioning in nonhuman primates. Therefore, no comparison could be carried out between distinct scales.
Of note, the scored aging-related health and functioning in rhesus macaques also displayed some aging-related paradoxes that humans have exhibited. A famous gender-survival paradox during aging has revealed that elderly female humans have worse health and lower functioning but greater longevity than males [30]. During the construction of the present scale, the health of the dominant males within the social hierarchy was not superior to that of females, in contrast to results that high socioeconomic status can retard aging [9, 15], this outcome reveals a gender-related health-social status paradox. Another phenomenon was the calorie-related health-survival paradox during aging. Although a chronic hypercaloric diet has been shown to result in obesity, multisystemic deterioration known as metabolic syndrome and accelerated aging, obese elderly individuals seem to live longer and to have better outcomes against infection compared to their age-matched thinner counterparts [31, 32]. Simultaneously, caloric restriction (CR), which has been found to result in longevity and retarded aging in many lower animals, does not consistently show this effect in nonhuman primates [33]. The largest two rhesus monkey cohorts exposed to a CR intervention initiated in the late 1980s have demonstrated contrasting results between health and survival [19, 34]. In the present rhesus macaque colony, females with lower social rank were observed to have later dietary uptake and smaller cheek pouches, which produced relatively hypocaloric diets and mild CR. However, as the complicated interactions of CR between longer life and better quality of life involved numerous critical co-factors [33, 35–37], whether the mild CR intervention in subjects with worse health and functioning would live longer warrants longitudinal observations. From the perspective of scale utilization, we suggested that these aging-related paradoxes in primates were attributable not only to the complex aging process but also to a lack of dynamic assessment of changeable aspects of health and functioning.
In summary, we developed an observer-based scale by integrating perceived aging, biophysical functioning and socially related performance based on the daily life of rhesus monkeys. The scale has been tested and has shown good interrater reliability, has satisfied construct validity and has acceptable applicability. The limited availability of advanced age samples, especially with healthy status, has hindered us from including adequate samples in the construction of the present scale. We are longitudinally observing and scoring this naturally aging cohort to further supplement the database and improve the scale on aging-related health and functioning. The wide application of the AHFS in rhesus macaques would be very helpful in disentangling the complexity of aging in humans.
Materials and Methods
Healthy animal screening
We observed all laboratory-maintained group-housed rhesus macaques in the North Primate Research Center affiliated with the Institute of Laboratory Animal Sciences, Chinese Academy of Medical Sciences & Peking Union Medical College from November 2018 to March 2019. Approximately two-thirds of the rhesus monkeys aged 4 to 20 were initially screened as candidates. A total of 57 rhesus monkeys identified as consistently healthy animals with normal functioning were maintained to construct a scale termed the Aging-related Health and Functioning Scale (AHFS). The animals with transient abnormalities, including decreased dietary uptake, diarrhea, fatigue and solitary habits during the observation, were excluded. All selected animals were research-naïve and pregnancy-free during the observation. The sex ratio was 1:1.04 (M:F=28:29). All subjects were of known age, parity, mass, and social rank according to colony records. All protocols were approved by the Institutional Animal Care and Use Committee of the Institute of Laboratory Animal Sciences, Chinese Academy of Medical Sciences & Peking Union Medical College.
A step-by-step selection of items under panel consensus
A panel including three researchers in the field of human aging, two researchers who study nonhuman primates and five experienced rhesus macaque caretakers was assembled. A step-by-step item selection was performed to screen candidate items for the AHFS. First, the three human aging researchers were consulted to present candidate domains and related items in reference to human aging. Then, items that could be easily differentiated and were relatively noninvasive and occurred in daily life were selected by the two nonhuman primate researchers. Finally, the five rhesus macaque caretakers carried out the preliminary assessment to determine which items could be practically rated. A general 3-point scoring system was used with minor exceptions. The higher the score was, the more severe the age-related aspect of health and functioning.
Statistical analysis
All observed results on an ordinal scale were analyzed as continuous values. The mean ± standard deviation (SD) was calculated between repetitive assessments. Using the 25th and 75th percentiles as cutoffs, three age groups were defined: junior (8–12 years old), adult (13–16 years old) and senior (17–20 years old). Pearson correlations were used to determine the relationship between the scored AHFS and each of the rated items as well as between pairs of items. In order to examine the reliability of the scale, Cronbach’s alpha value and Cronbach’s alpha half-split value was calculated. In order to validate the construct of the scale, an exploratory factor analysis was performed. The number of underlying exploratory factors in the scale was determined by eigenvalues. Varimax rotation was performed to achieve the simplest structure of these exploratory factors. The factor loadings of each item within its exploratory factor were extracted within the rotated structure. In order to validate the applicability of the scale, a hierarchical cluster analysis was carried out based on the individual’s scored aging-related health and function. The optimal number of clusters was determined based on a comparison of the respective presented dendrograms. Similarity was measured based on the interval of intraindividual scores within each group. All analyses were conducted in SPSS (17.0) and R (3.5.0). A P value less than 0.05 was considered statistically significant.
Acknowledgments
We appreciate the statistics aid from Dr. Wang Han and Dr. Zun-song Hu. We appreciate the consultations on item screening from Professor Qin Li and Dr. Jing-yu Zhang. We appreciate the arduous work on scoring rhesus macaques from Hai-quan Shang, Lei Wang, Yunpeng Jin and Huiwu Li.
Conflicts of Interest
The authors have no conflicts of interest to declare.
Funding
The study was funded by Grant of Natural Science Foundation of China (NSFC No. 81671383), Grant of Natural Science Foundation of China for young scientists (No. 81600629) and Chinese Academy of Medical Sciences Innovation Fund for Medical Sciences (No. 2016-12M-2-006-001).
References
- 1. Young Y, Frick KD, Phelan EA. Can successful aging and chronic illness coexist in the same individual? A multidimensional concept of successful aging. J Am Med Dir Assoc. 2009; 10:87–92. https://doi.org/10.1016/j.jamda.2008.11.003 [PubMed]
- 2. WHO. What Is Healthy Ageing. 2018. https://www.who.int/ageing/healthy-ageing/en/.
- 3. Mattison JA, Vaughan KL. An overview of nonhuman primates in aging research. Exp Gerontol. 2017; 94:41–45. https://doi.org/10.1016/j.exger.2016.12.005 [PubMed]
- 4. Asadi Noughabi A, Alhani F, Piravi H. Health hybrid concept analysis in old people. Glob J Health Sci. 2013; 5:227–32. https://doi.org/10.5539/gjhs.v5n6p227 [PubMed]
- 5. Roth GS, Mattison JA, Ottinger MA, Chachich ME, Lane MA, Ingram DK. Aging in rhesus monkeys: relevance to human health interventions. Science. 2004; 305:1423–26. https://doi.org/10.1126/science.1102541 [PubMed]
- 6. Simmons HA. Age-Associated Pathology in Rhesus Macaques (Macaca mulatta). Vet Pathol. 2016; 53:399–416. https://doi.org/10.1177/0300985815620628 [PubMed]
- 7. Mitnitski A, Howlett SE, Rockwood K. Heterogeneity of Human Aging and Its Assessment. J Gerontol A Biol Sci Med Sci. 2017; 72:877–84. https://doi.org/10.1093/gerona/glw089 [PubMed]
- 8. Skirbekk VF, Staudinger UM, Cohen JE. How to Measure Population Aging? The Answer Is Less than Obvious: A Review. Gerontology. 2019; 65:136–144. https://doi.org/10.1159/000494025 [PubMed]
- 9. de la Fuente J, Caballero FF, Sánchez-Niubó A, Panagiotakos DB, Prina AM, Arndt H, Haro JM, Chatterji S, Ayuso-Mateos JL. Determinants of Health Trajectories in England and the United States: An Approach to Identify Different Patterns of Healthy Aging. J Gerontol A Biol Sci Med Sci. 2018; 73:1512–18. https://doi.org/10.1093/gerona/gly006 [PubMed]
- 10. Zhang Z, Andersen A, Smith C, Grondin R, Gerhardt G, Gash D. Motor slowing and parkinsonian signs in aging rhesus monkeys mirror human aging. J Gerontol A Biol Sci Med Sci. 2000; 55:B473–80. https://doi.org/10.1093/gerona/55.10.B473 [PubMed]
- 11. Uno H, Poff B. Coronary arterial ectasia, a predominant type of coronary sclerosis in aged captive rhesus monkeys (Macaca mulatta). Am J Pathol. 1983; 111:315–22. [PubMed]
- 12. Diamond A, Goldman-Rakic PS. Comparison of human infants and rhesus monkeys on Piaget’s AB task: evidence for dependence on dorsolateral prefrontal cortex. Exp Brain Res. 1989; 74:24–40. https://doi.org/10.1007/BF00248277 [PubMed]
- 13. Clingerman KJ, Summers L. Development of a body condition scoring system for nonhuman primates using Macaca mulatta as a model. Lab Anim (NY). 2005; 34:31–36. https://doi.org/10.1038/laban0505-31 [PubMed]
- 14. Ailshire JA, Crimmins EM. Psychosocial Factors Associated with Longevity in the United States: Age Differences between the Old and Oldest-Old in the Health and Retirement Study. J Aging Res. 2011; 2011:530534. https://doi.org/10.4061/2011/530534 [PubMed]
- 15. Xu X, Liang J, Bennett JM, Botoseneanu A, Allore HG. Socioeconomic stratification and multidimensional health trajectories: evidence of convergence in later old age. J Gerontol B Psychol Sci Soc Sci. 2015; 70:661–71. https://doi.org/10.1093/geronb/gbu095 [PubMed]
- 16. Colman RJ, McKiernan SH, Aiken JM, Weindruch R. Muscle mass loss in Rhesus monkeys: age of onset. Exp Gerontol. 2005; 40:573–81. https://doi.org/10.1016/j.exger.2005.05.001 [PubMed]
- 17. Uotinen V, Rantanen T, Suutama T. Perceived age as a predictor of old age mortality: a 13-year prospective study. Age Ageing. 2005; 34:368–72. https://doi.org/10.1093/ageing/afi091 [PubMed]
- 18. Christensen K, Thinggaard M, McGue M, Rexbye H, Hjelmborg JV, Aviv A, Gunn D, van der Ouderaa F, Vaupel JW. Perceived age as clinically useful biomarker of ageing: cohort study. BMJ. 2009; 339:b5262. https://doi.org/10.1136/bmj.b5262 [PubMed]
- 19. Colman RJ, Anderson RM, Johnson SC, Kastman EK, Kosmatka KJ, Beasley TM, Allison DB, Cruzen C, Simmons HA, Kemnitz JW, Weindruch R. Caloric restriction delays disease onset and mortality in rhesus monkeys. Science. 2009; 325:201–04. https://doi.org/10.1126/science.1173635 [PubMed]
- 20. Vaskov AK, Irwin ZT, Nason SR, Vu PP, Nu CS, Bullard AJ, Hill M, North N, Patil PG, Chestek CA. Cortical Decoding of Individual Finger Group Motions Using ReFIT Kalman Filter. Front Neurosci. 2018; 12:751. https://doi.org/10.3389/fnins.2018.00751 [PubMed]
- 21. Novak MA, Hamel AF, Coleman K, Lutz CK, Worlein J, Menard M, Ryan A, Rosenberg K, Meyer JS. Hair loss and hypothalamic-pituitary-adrenocortical axis activity in captive rhesus macaques (Macaca mulatta). J Am Assoc Lab Anim Sci. 2014; 53:261–66. https://doi.org/10.1002/ajp.22547 [PubMed]
- 22. Steinmetz HW, Kaumanns W, Dix I, Heistermann M, Fox M, Kaup FJ. Coat condition, housing condition and measurement of faecal cortisol metabolites—a non-invasive study about alopecia in captive rhesus macaques (Macaca mulatta). J Med Primatol. 2006; 35:3–11. https://doi.org/10.1111/j.1600-0684.2005.00141.x [PubMed]
- 23. Wooddell LJ, Hamel AF, Murphy AM, Byers KL, Kaburu SS, Meyer JS, Suomi SJ, Dettmer AM. Relationships between affiliative social behavior and hair cortisol concentrations in semi-free ranging rhesus monkeys. Psychoneuroendocrinology. 2017; 84:109–15. https://doi.org/10.1016/j.psyneuen.2017.06.018 [PubMed]
- 24. Hamel AF, Menard MT, Novak MA. Fatty acid supplements improve hair coat condition in rhesus macaques. J Med Primatol. 2017; 46:248–51. https://doi.org/10.1111/jmp.12271 [PubMed]
- 25. Guiglia R, Musciotto A, Compilato D, Procaccini M, Lo Russo L, Ciavarella D, Lo Muzio L, Cannone V, Pepe I, D’Angelo M, Campisi G. Aging and oral health: effects in hard and soft tissues. Curr Pharm Des. 2010; 16:619–30. https://doi.org/10.2174/138161210790883813 [PubMed]
- 26. Cuozzo FP, Sauther ML, Gould L, Sussman RW, Villers LM, Lent C. Variation in dental wear and tooth loss among known-aged, older ring-tailed lemurs (Lemur catta): a comparison between wild and captive individuals. Am J Primatol. 2010; 72:1026–37. https://doi.org/10.1002/ajp.20846 [PubMed]
- 27. Laguna L, Sarkar A, Artigas G, Chen J. A quantitative assessment of the eating capability in the elderly individuals. Physiol Behav. 2015; 147:274–81. https://doi.org/10.1016/j.physbeh.2015.04.052 [PubMed]
- 28. Sacrey LA, Alaverdashvili M, Whishaw IQ. Similar hand shaping in reaching-for-food (skilled reaching) in rats and humans provides evidence of homology in release, collection, and manipulation movements. Behav Brain Res. 2009; 204:153–61. https://doi.org/10.1016/j.bbr.2009.05.035 [PubMed]
- 29. Donchin O, Gribova A, Steinberg O, Bergman H, Vaadia E. Primary motor cortex is involved in bimanual coordination. Nature. 1998; 395:274–78. https://doi.org/10.1038/26220 [PubMed]
- 30. Oksuzyan A, Petersen I, Stovring H, Bingley P, Vaupel JW, Christensen K. The male-female health-survival paradox: a survey and register study of the impact of sex-specific selection and information bias. Ann Epidemiol. 2009; 19:504–11. https://doi.org/10.1016/j.annepidem.2009.03.014 [PubMed]
- 31. Roth J, Sahota N, Patel P, Mehdi SF, Wiese MM, Mahboob HB, Bravo M, Eden DJ, Bashir MA, Kumar A, Alsaati F, Kurland IJ, Brima W, et al. Obesity paradox, obesity orthodox, and the metabolic syndrome: an approach to unity. Mol Med. 2017; 22:873–85. https://doi.org/10.2119/molmed.2016.00211 [PubMed]
- 32. Abdullah A, Wolfe R, Stoelwinder JU, de Courten M, Stevenson C, Walls HL, Peeters A. The number of years lived with obesity and the risk of all-cause and cause-specific mortality. Int J Epidemiol. 2011; 40:985–96. https://doi.org/10.1093/ije/dyr018 [PubMed]
- 33. Mendelsohn AR, Larrick JW. Dietary restriction: critical co-factors to separate health span from life span benefits. Rejuvenation Res. 2012; 15:523–29. https://doi.org/10.1089/rej.2012.1371 [PubMed]
- 34. Mattison JA, Roth GS, Beasley TM, Tilmont EM, Handy AM, Herbert RL, Longo DL, Allison DB, Young JE, Bryant M, Barnard D, Ward WF, Qi W, et al. Impact of caloric restriction on health and survival in rhesus monkeys from the NIA study. Nature. 2012; 489:318–21. https://doi.org/10.1038/nature11432 [PubMed]
- 35. Godfrey JR, Diaz MP, Pincus M, Kovacs-Balint Z, Feczko E, Earl E, Miranda-Dominguez O, Fair D, Sanchez MM, Wilson ME, Michopoulos V. Diet matters: glucocorticoid-related neuroadaptations associated with calorie intake in female rhesus monkeys. Psychoneuroendocrinology. 2018; 91:169–78. https://doi.org/10.1016/j.psyneuen.2018.03.008 [PubMed]
- 36. Michopoulos V, Reding KM, Wilson ME, Toufexis D. Social subordination impairs hypothalamic-pituitary-adrenal function in female rhesus monkeys. Horm Behav. 2012; 62:389–99. https://doi.org/10.1016/j.yhbeh.2012.07.014 [PubMed]
- 37. Mattison JA, Colman RJ, Beasley TM, Allison DB, Kemnitz JW, Roth GS, Ingram DK, Weindruch R, de Cabo R, Anderson RM. Caloric restriction improves health and survival of rhesus monkeys. Nat Commun. 2017; 8:14063. https://doi.org/10.1038/ncomms14063 [PubMed]



