Introduction
Cardiovascular diseases and cognitive impairment are frequent and increasingly prevalent, especially in older patients and patients with end-stage renal disease (ESRD) [1–4]. Both chronic kidney disease, especially ESRD, and cardiovascular diseases have been identified as independent risk factors for the development of microvascular damage and cerebral small vessel disease, which can lead to structural cerebrovascular changes and cognitive impairment [5–9]. It is, however, unknown how cardiovascular structure and function associates with brain structure and function in older patients with ESRD.
In ESRD nephrogenic factors as uremic toxins, anaemia and inflammation, are potential underlying mechanisms for the development of cerebrovascular changes and cognitive impairment [9]. Furthermore, cardiovascular risk factors can lead to microvascular damage in both the brain and kidney [9, 10]. The association between cardiovascular structure and function with both cerebrovascular changes and cognitive impairment can be divided into two possible mechanisms in the general population, namely increased arterial stiffness and impaired systolic heart function. Arterial stiffness can cause microvascular damage in the brain due to an increased impact of pulsatility on the microvasculature, which possibly alters brain structure or cognitive functioning [11–13]. Furthermore, impaired systolic heart function could cause cerebral ischemia because of hypoperfusion in the brain due to decreased cardiac output [14–17]. To what extent an altered cardiac structure and function play a role in cerebrovascular changes and cognitive impairment in older patients with ESRD remains unclear.
Figure 1 shows the hypothesis of the current study as well as the potential underlying mechanisms. The aim of this study was to investigate the association of cardiovascular structure and function with cerebrovascular changes and cognitive function in older patients with ESRD.
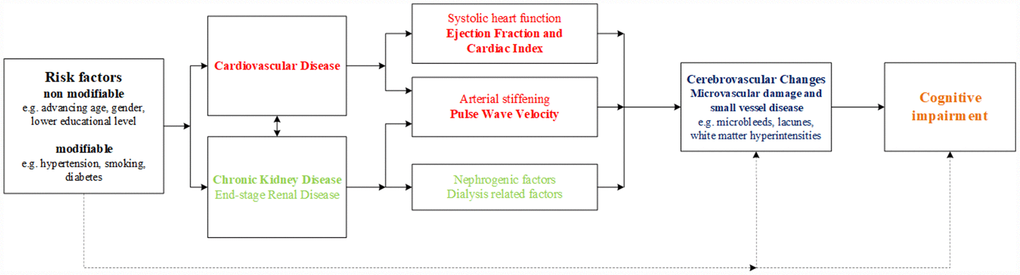
Figure 1. The heart-kidney-brain axis. Hypothesis of the current study and the potential underlying pathophysiological mechanisms in the heart-kidney-brain axis.
Results
Of the 157 patients included in the ‘The Cognitive decline in Older Patients with End stage renal disease’ (COPE) study, cardiac magnetic resonance imaging (MRI) scans were available for 85 participants, see the flowchart in Figure 2. Baseline characteristics of all patients are shown in Table 1. Mean±standard deviation (SD) of age was 75.6±6.9years and 56 (66%) patients were male. Mean±SD eGFR at time of inclusion was 15.8±4.2ml/min/1.73m2. The origin of primary kidney disease was non-vascular in 36% and vascular (mainly diabetes and hypertension) in 64% of all patients. Median [interquartile range (IQR)] pulse wave velocity (PWV) was 9.6m/s [7.8-13.0], ejection fraction (EF) 62% [51–66] and cardiac index (CI) 2.5l/min/m2 [2.1-3.0]. Global cognition in the total population was not impaired, measured by the MMSE with median [IQR] 28 [27–30] out of 30 points and also clock drawing with a median [IQR] of 12 [11–13]. Differences in baseline characteristics for each subgroup of PWV, EF and CI are shown in Supplementary Tables 1–3.
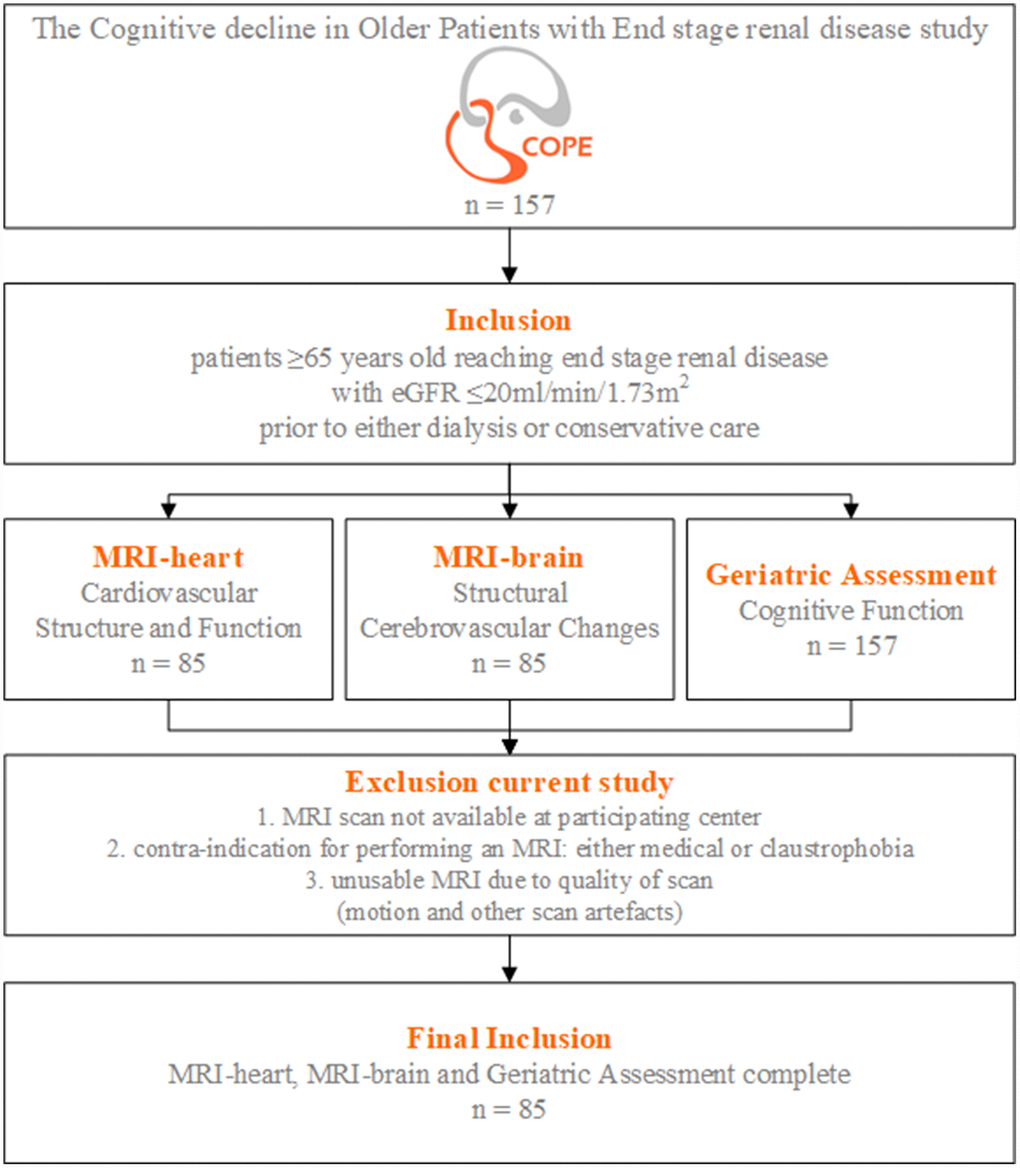
Figure 2. Flowchart study population. Inclusion and exclusion criteria of the COPE (The Cognitive decline in Older Patients with End stage renal disease) study.
Table 1. Baseline characteristics total population (n=85).
| Male gender, n (%) | 56 (65.9) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age, years; mean ± SD | 75.6 ± 6.9 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Race, Caucasian, n (%) | 75 (88.2) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Higher educational level, n (%) | 32 (37.6) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Primary kidney disease, n (%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Non-vascular cause | 30 (35.7) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Vascular cause | 54 (64.3) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Comorbidity, n (%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Diabetes mellitus | 32 (37.6) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Peripheral vascular disease | 16 (18.8) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cerebral vascular accident | 23 (27.1) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Heart failure | 7 (8.2) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Coronary heart disease | 18 (21.2) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atrial fibrillation | 17 (20.5) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Alcohol consumption, n (%) | 45 (52.9) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Current smoking, n (%) | 14 (16.5) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| History of smoking, n (%) | 49 (57.6) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Medication use, n (%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Polypharmacy (the use of ≥5 medications) | 75 (88.2) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Antihypertensive medication | 79 (92.9) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Beta-blockers | 44 (51.8) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Diuretics | 50 (58.8) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Objective measures, mean ± SD | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Blood pressure (mmHg) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Systolic | 150.3 ± 22.2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Diastolic | 81.6 ± 11.8 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| eGFR (ml/min/1.73m2) | 15.8 ± 4.2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Urea (mg/dL) | 21.3 ± 6.3 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Phosphate (mmol/L) | 1.32 ± 0.29 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Albuminuria (mg/24 hours) | 771 ± 882 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Troponin (ng/L) | 0.052 ± 0.070 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| NT-proBNP (ng/L) | 879 ± 1208 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cardiovascular function, measured by MRI, median [IQR] | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Pulse wave velocity (m/s) | 9.6 [7.8-13.0] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Ejection fraction (%) | 62 [51–66] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cardiac index (l/min/m2) | 2.5 [2.1-3.0] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cerebrovascular changes, measured by MRI, n (%) or mean ± SD | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Presence of microbleeds | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Non-lobar | 18 (21.2) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Lobar | 32 (37.6) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Presence of lacunes* | 39 (45.9) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Total white matter hyperintensities | 16.1 ± 8.0 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cognitive function performance, mean ± SD or median [IQR] | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Global cognition | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Mini-Mental State Examination (points) | 28 [27–30] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Visuoconstruction | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Clock drawing | 12 [11–13] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Memory | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 15-WVLT immediate recall | 32.3 ± 10.1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 15-WVLT delayed recall | 6.3 ± 3.0 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Visual Association Test | 12 [11–12] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Executive function | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TMT-B (sec) | 157.0 ± 72.5 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SCWT III (sec) | 166.0 ± 90.0 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SCWT III corrected for SCWT II (sec) | 84.0 ± 81.6 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Psychomotor Speed | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LDST (correct in 60 sec) | 22.9 ± 7.1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TMT-A (sec) | 62.0 ± 39.0 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SCWT II (sec) | 82.5 ± 33.7 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| *Lacunes; both gliotic and hemorrhagic parenchymal defects subcortical, in brain stem and basal ganglia. Abbreviations: 15-WVLT, 15-Word Verbal Learning Test; eGFR, estimated glomerular filtration rate; LDST, Letter Digit Substitution Test; NT-proBNP, N-terminal pro b-type natriuretic peptide; SD, standard deviation; SCWT, Stroop Color and Word Test; TMT, Trail Making Test. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Cerebrovascular changes
Table 2 shows the association between cardiac parameters and cerebrovascular changes. No statistically significant associations were observed between cardiac parameters and cerebrovascular changes. Patients with a high PWV, and therefore high aortic stiffness, more often than with low PWV, had more structural cerebrovascular changes, including more microbleeds (both non-lobar and lobar) and lacunes, and a higher mean total white matter hyperintensities (WMH), although differences were of unknown clinical relevance and not statistically significant. Similar non-significant results were seen for patients with a low compared to high EF and a low compared to high CI. A sensitivity analysis based on median PWV, EF and CI and a sensitivity analysis excluding patients with a history of CVA yielded similar results.
Table 2. Association of cardiovascular function parameters and cerebrovascular changes.
| Better cardiovascular function | Worse cardiovascular function | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Pulse wave velocity ≤10m/s n = 45 | Pulse wave velocity >10m/s n = 38 | p-value | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Crude | Adjusted | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Presence of microbleeds, % | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Non-lobar | 7 (16.7) | 11 (28.9) | 0.189 | 0.575 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Lobar | 13 (31.0) | 18 (47.4) | 0.132 | 0.105 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Presence of lacunes, % | 18 (42.9) | 19 (50.0) | 0.522 | 0.616 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Total WMH, mean ± SE | 15.5 ± 1.2 | 16.6 ± 1.4 | 0.561 | 0.438 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Ejection fraction ≥50% n = 65 | Ejection fraction <50% n = 19 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Presence of microbleeds, % | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Non-lobar | 13 (20.6) | 5 (27.8) | 0.520 | 0.563 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Lobar | 22 (34.9) | 10 (55.6) | 0.114 | 0.121 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Presence of lacunes, % | 29 (46.0) | 10 (55.6) | 0.476 | 0.767 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Total WMH, mean ± SE | 15.8 ± 0.9 | 16.9 ± 2.4 | 0.609 | 0.778 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cardiac index >2.2l/min/m2 n = 56 | Cardiac index ≤2.2l/min/m2 n = 27 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Presence of microbleeds, % | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Non-lobar | 13 (23.6) | 5 (20.0) | 0.718 | 0.457 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Lobar | 23 (41.8) | 8 (32.0) | 0.403 | 0.382 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Presence of lacunes, % | 28 (50.9) | 10 (40.0) | 0.365 | 0.443 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Total WMH, mean ± SE | 17.0 ± 1.2 | 14.2 ± 1.3 | 0.151 | 0.166 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| P-values are assessed using linear or logistic regression models, unadjusted and adjusted for age and sex, comparing low versus high pulse wave velocity, ejection fraction and cardiac index. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Lacunes include both gliotic and hemorrhagic parenchymal defects subcortical, in brain stem and basal ganglia. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Abbreviations: SE, standard error; WMH, white matter hyperintensities. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Cognitive function
Table 3 shows the association between cardiac parameters and cognitive function in three different domains, namely memory, executive function and psychomotor speed. The scores of all cognitive function tests were worse for patients with a high compared to low PWV in all three domains, statistical significance was reached for the Trail Making Test B (TMTB) with a difference of 39 seconds (p=0.009), for Trail Making Test A (TMTA) with a difference of 23 seconds (p=0.008) and for the Letter Digit Substitution Test (LDST) with a difference of 4 correct answers in 60 seconds (p=0.021). After adjustment for age, sex and education, only the TMTA, measuring psychomotor speed, remained statistically significant (p=0.030). No clinically relevant nor statistically significant associations were found in cognitive function comparing patients with low compared to high EF. Patients with a low compared to high CI had a worse memory function (both immediate and delayed recall), which remained statistically significant for delayed recall after adjustment (p=0.003). No statistically significant differences were found in executive function or psychomotor speed. A sensitivity analysis based on median PWV, EF and CI and a sensitivity analysis excluding patients with a history of CVA yielded similar results.
Table 3. Association of cardiovascular function parameters and cognitive function.
| Better Cardiovascular function | Worse Cardiovascular function | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PWV ≤10m/s n=45 | PWV >10m/s n=38 | p-value | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Crude | Adjusted | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Memory | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 15-Word Verbal Learning Test immediate recall ↓ | 33.3 ± 1.2 | 31.6 ± 2.0 | 0.455 | 0.899 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 15-Word Verbal Learning Test delayed recall ↓ | 6.6 ± 0.4 | 5.9 ± 0.6 | 0.307 | 0.719 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Executive function | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Trail Making Test B (sec) ↑ | 140.6 ± 9.6 | 179.3 ± 12.2 | 0.009 | 0.184 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SCWT III (sec) ↑ | 158.3 ± 15.6 | 173.5 ± 11.8 | 0.439 | 0.339 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SCWT III corrected for SCWT II (sec) ↑ | 80.5 ± 14.0 | 87.6 ± 11.1 | 0.692 | 0.148 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Psychomotor speed | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Letter Digit Substitution Test (correct in 60 sec) ↓ | 24.6 ± 0.9 | 20.9 ± 1.3 | 0.021 | 0.179 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Trail Making Test A (sec) ↑ | 51.6 ± 2.5 | 74.6 ± 8.7 | 0.008 | 0.030 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Stroop Color and Word Test II (sec) ↑ | 77.8 ± 3.9 | 87.0 ± 6.7 | 0.241 | 0.361 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| EF ≥50% n=65 | EF <50% n=19 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Memory | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 15-Word Verbal Learning Test immediate recall ↓ | 32.1 ± 1.2 | 32.5 ± 2.8 | 0.881 | 0.621 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 15-Word Verbal Learning Test delayed recall ↓ | 6.4 ± 0.4 | 5.7 ± 0.8 | 0.426 | 0.664 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Executive function | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Trail Making Test B (sec) ↑ | 160.4 ± 8.9 | 153.4 ± 19.2 | 0.809 | 0.728 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SCWT III (sec) ↑ | 166.7 ± 11.7 | 164.8 ± 19.1 | 0.940 | 0.669 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SCWT III corrected for SCWT II (sec) ↑ | 86.4 ± 10.3 | 75.9 ± 19.8 | 0.633 | 0.392 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Psychomotor Speed | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Letter Digit Substitution Test (correct in 60 sec) ↓ | 23.4 ± 0.8 | 21.3 ± 1.9 | 0.265 | 0.383 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Trail Making Test A (sec) ↑ | 59.2 ± 4.4 | 73.4 ± 12.3 | 0.186 | 0.274 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Stroop Color and Word Test II (sec) ↑ | 80.9 ± 3.2 | 88.9 ± 12.8 | 0.377 | 0.459 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CI >2.2 l/min/m2 n=56 | CI≤2.2 l/min/m2 n=27 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Memory | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 15-Word Verbal Learning Test immediate recall ↓ | 33.2 ± 1.4 | 30.1 ± 1.9 | 0.207 | 0.219 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 15-Word Verbal Learning Test delayed recall ↓ | 6.9 ± 0.4 | 4.9 ± 0.5 | 0.004 | 0.003 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Executive function | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Trail Making Test B (sec) ↑ | 149.6 ± 8.7 | 176. ± 16.6 | 0.156 | 0.191 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SCWT III (sec) ↑ | 164.5 ± 13.1 | 171.4 ± 15.6 | 0.748 | 0.978 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SCWT III corrected for SCWT II (sec) ↑ | 85.1 ± 12.9 | 84.3 ± 10.0 | 0.966 | 0.712 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Psychomotor Speed | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Letter Digit Substitution Test (correct in 60 sec) ↓ | 23.4 ± 0.9 | 21.5 ± 1.5 | 0.266 | 0.349 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Trail Making Test A (sec) ↑ | 59.9 ± 4.4 | 68.1 ± 10.1 | 0.384 | 0.414 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Stroop Color and Word Test II (sec) ↑ | 80.2 ± 4.6 | 87.1 ± 6.5 | 0.387 | 0.428 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Values are mean ± SE. ↓↑ indicates that a higher (↑) or lower (↓) score means a worse cognitive function. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| P-values are assessed using linear regression models, unadjusted and multivariate adjusted for age, sex and education, comparing low versus high pulse wave velocity, ejection fraction and cardiac index. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Abbreviations: CI, cardiac index; EF, ejection fraction; PWV, pulse wave velocity | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Discussion
The main findings of this explorative study are as follows. First, higher PWV associated with all measures of cognitive impairment, albeit this was only statistically significant for the association of PWV with the TMTA, measuring psychomotor speed. Second, no statistically significant differences in the association between cardiovascular structure and function and structural cerebrovascular changes were found.
Although the association of arterial stiffness and brain pathology has been described previously in patients with ESRD, studies are limited, and included in general only cerebrovascular changes [18] or cognition [19], and in case of the latter limited tests for global cognition, instead of differentiating between various functional domains [20, 21]. In the general population, the influence of arterial stiffness on both cerebrovascular changes and cognitive function has been more extensively studied. Cardiovascular risk factors such as hypertension result in arterial stiffening that can be measured in the aorta as increased PWV, an important and independent determinant of arterial disease [11, 12]. Due to an impaired Windkessel effect aortic stiffness might increase the impact of cardiac pulsations on the cerebral microvasculature leading to cerebral small vessel diseases and cognitive impairment [13], as was recently confirmed by a systematic review [22]. Studies have shown independent associations of impaired systolic heart function on cerebrovascular changes or cognitive function, probably due to cerebral ischemia because of hypoperfusion in the brain due to decreased cardiac output [14–17], including in patients with ESRD [23]. However, it might have a more multifactorial dependency, like for instance whether patients clinically have heart failure. Previous studies have shown associations of heart failure [14], and also associations of both EF and CI in patients with heart failure [15], with cerebrovascular changes and cognitive impairment. Furthermore, treatment of heart failure, with for example cardiac resynchronization therapy or heart transplantation, can improve cognitive function, partly due to improvement of cerebral blood flow [24–26]. Taken together, our findings that arterial stiffness may be an underlying mechanism of development of cognitive impairment is in line with known literature in both the general population, but also in patients with ESRD.
We investigated two hypotheses as underlying pathophysiological mechanisms in the heart-kidney-brain axis, namely arterial stiffness and systolic heart function (Figure 1). Few associations reached statistical significance, possibly due to the relatively low number of participants in the study. However, for PWV, all 4 cerebrovascular changed parameters, and all 8 cognitive associates point in the same direction. The magnitudes of association in most cognitive parameters were well above what could be considered clinically relevant, which is unlikely the result of chance. Therefore, we conclude that our results suggest that PWV is a potential predictor of cognitive function in older patients with ESRD. These results, however, should be considered as "suggestive" and need replication in larger cohorts. The contributing role of systolic cardiac function on cerebrovascular changes and cognitive impairment seemed limited in our COPE population, possibly due to a low percentage of patients with manifest symptoms of clinical heart failure (7%). In addition, although EF and CI are both parameters of systolic cardiac function, values within the same patients are not always concordant. In our population, patients with a low EF had a relatively normal mean CI and vice versa. Cardiac output has been pointed out previously to be a superior reflection of systemic blood flow and cerebral blood flow than EF, especially in patients without heart failure [27]. It might explain that low CI was a better predictor of cognitive impairment (memory domain) than low EF in our population.
Strengths and limitations
The COPE study is a unique observational study in older patients with ESRD with a mean eGFR of 16ml/min/1.73m2, prior to either dialysis or conservative care, with comprehensive measurements of cardiovascular function, cerebrovascular changes and cognitive function.
However, some limitations should be mentioned. Patient numbers were relatively limited, and although cardiovascular comorbidity was common, patients had a relatively normal cardiac function, with only 19 patients available with EF ≤50% and 27 patients with CI <2.2l/min/m2, limiting the power to find an association between cardiovascular function and cerebrovascular changes or cognitive function of a smaller magnitude. This limited us to merely observe possible trends suitable for future research.
Conclusions
In conclusion, this exploratory study suggests that a higher PWV is associated with lower cognitive function, but not with increased cerebrovascular changes in older patients with ESRD, suggesting that arterial stiffness may be an underlying mechanism of development of cognitive impairment.
Larger studies should replicate and extend on these findings, as identifying the mechanisms involved in cerebrovascular changes and cognitive impairment can be the first step towards prevention strategies. Prevention is of utmost importance, as the Framingham Heart Study have showed that earlier diagnosis and effective treatment of risk factors or proven vascular disease, can possibly lead to a decline in incidence of dementia [28]. Furthermore, future research should also focus on other potential biomarkers as miRNAs or metabolomics to unravel specific pathophysiological mechanisms in this interaction between the heart, kidney and brain and thereafter on potential interventions to prevent cerebrovascular changes and cognitive impairment.
Materials and Methods
Data of ‘The Cognitive decline in Older Patients with End stage renal disease’ (COPE) study were used, a Dutch prospective, multicenter cohort study. A detailed description of the rationale and design of COPE, including all in- and exclusion criteria, has been published previously [29]. In summary, patients ≥65 years old reaching ESRD with eGFR ≤20ml/min/1.73m2 (CKD stage 4 and 5) were included, prior to either dialysis or conservative care. The main study objective was to study the association between underlying pathophysiological mechanisms and cognitive decline in patients with ESRD. For this purpose, magnetic resonance imaging (MRI) of the heart and brain and extensive neurocognitive testing were performed. For the current analysis patients without a cardiac MRI were excluded, see flowchart in Figure 2. Written informed consent was obtained from all study participants. The study protocol was approved by the medical ethics committees (METC) of all participating centers (Leiden University Medical Center [LUMC, Leiden], HAGA Hospital [Den Haag], Dialysis Center Zoetermeer [Zoetermeer], Reinier de Graaf Group [Delft] and Jeroen Bosch Hospital [Den Bosch]).
Magnetic resonance imaging
All MRI scans were made on a 3T Philips Achieva MRI scanner (Philips, Best, The Netherlands). Brain MRI was made with a 32 channel receive coil, heart MRI with an 8-channel receive coil.
Cardiovascular structure and function
The cardiac MRI protocol included flow sensitive imaging by phase contrast MRI for pulse wave velocity (PWV), measuring aortic stiffness [29]. Furthermore, the protocol included TFE (turbo field echo) multi-slice multi-phase cine-imaging of the left ventricle for systolic function, including ejection fraction (EF) and cardiac index (CI). Ejection fraction is the percentage of blood ejected out of the ventricles with each contraction (stroke volume divided by the end diastolic volume in %). Cardiac index is calculated as cardiac output (stroke volume multiplied by heart rate) and corrected for body surface area with use of the Du Bois formula (in l/min/m2) [30]. After exclusion of scans with poor quality (artifacts mainly caused by movement or distortions) PWV was available for 83 patients, EF for 84 patients and CI for 83 patients.
Cerebrovascular changes
The brain MRI protocol included 3D FLAIR (fluid attenuated inversion recovery) and T2-weighted brain MRI images, which were scored for the presence of markers of small vessel disease, including white matter hyperintensities (WMH) and lacunes. Susceptibility weighted imaging was used to score the presence and distribution of cerebral microbleeds. Cerebrovascular changes were rated as presence of (non) lobar microbleeds, presence of lacunes and grade of WMH according to the Scheltens score [31]. For three patients the brain MRI was of insufficient quality for the rating, due to motion artefacts.
Cognitive function
Detailed description of the comprehensive geriatric assessment and neuropsychological tests used in COPE has been published previously [29]. Outcome variables were derived from seven widely used neuropsychological tests in five different cognitive domains. First, global cognition was measured by the Mini-Mental State Examination (MMSE), with a general cut-off point of 24 out of 30 points [32]. Visuoconstructible abilities were assessed using the clock drawing test, which is often used as dementia screening test, with scores ranging from 0 to 14 points based on accuracy [33, 34]. Memory was assessed using two tests, namely the 15-Word Verbal Learning Test (15-WVLT), which measures immediate recall (total outcome of five presentations) and delayed recall after 20 minutes [35–37]. Memory was also assessed using the Visual Association Test (VAT) of which the score is based on the number of completed associations reported in two trials [38]. Executive functioning was assessed using the Trail Making Test B (TMTB), which is a switching task. The score of the TMTB is the number of seconds required to complete the task [39, 40]. Furthermore, the Stroop Color and Word Test (SCWT) card III (interference card) was used [41–43]. The SCWT consists of three parts, namely reading color names (card I), naming colored patches (card II) and naming color names printed in incongruously colored ink (card III). The time in seconds required to read the names or to identify colors is recorded. We used an abbreviated version of the test with 40 elements [44]. Psychomotor speed was assessed with the Letter Digit Substitution Test (LDST), Trail Making Test A (TMTA) and SCWT card II (naming colored patches). The LDST is a modification of the procedurally identical Symbol-Digits Modalities Test, with an outcome variable of total number of correct entries completed in 60 seconds [45, 46]. The score of the TMTA, testing visual attention, is the counted the same as the TMTB [39, 40].
Statistical analysis
Cardiac parameters are dichotomized by clinical cut-off values based on current guidelines [47, 48]. These cut-off values are ≤10m/s and >10m/s for PWV, respectively no aortic stiffness versus aortic stiffness, ≤50% and >50% for EF, and <2.2l/min/m2 and ≥2.2L/min/m2 for CI, both respectively low versus high. All categorical data are presented as numbers with percentages. All continuous data are presented as mean ± standard error or ± standard deviation, or median with interquartile range. Baseline differences between cardiac parameters are assessed using an independent t-test, Mann-Whitney U test or chi-square test. Multivariate linear or logistic regression models are used to assess the associations of cardiac function with cerebrovascular changes and cognitive function. All analyses were adjusted for prespecified confounders, namely age and gender, and also education in case of cognitive function analyses. The data were analyzed using IBM SPSS Statistics version 23. P-values lower than 0.05 were considered statistically significant.
Supplementary Materials
Author Contributions
Laurien E. Zijlstra, Conception and design of the study; analysis and interpretation of data; drafting of the manuscript; providing intellectual content of critical importance to the work described; final approval of the manuscript, Stella Trompet, Conception and design of the study; analysis and interpretation of data; revising of the manuscript; providing intellectual content of critical importance to the work described; final approval of the manuscript, J. Wouter Jukema, Conception and design of the study; analysis and interpretation of data; revising of the manuscript; providing intellectual content of critical importance to the work described; final approval of the manuscript, Lucia J.M. Kroft, Conception and design of the study; analysis and interpretation of data; revising of the manuscript; providing intellectual content of critical importance to the work described; final approval of the manuscript, Jeroen de Bresser, Conception and design of the study; analysis and interpretation of data; revising of the manuscript; providing intellectual content of critical importance to the work described; final approval of the manuscript, Matthias J.P. van Osch, Conception and design of the study; analysis and interpretation of data; revising of the manuscript; providing intellectual content of critical importance to the work described; final approval of the manuscript, Sebastiaan Hammer, Conception and design of the study; analysis and interpretation of data; revising of the manuscript; providing intellectual content of critical importance to the work described; final approval of the manuscript, Marie-Noëlle Witjes, Revising of the manuscript; providing intellectual content of critical importance to the work described; final approval of the manuscript, Marjolijn van Buren*, Conception and design of the study; analysis and interpretation of data; revising of the manuscript; providing intellectual content of critical importance to the work described; final approval of the manuscript, Simon P. Mooijaart*, Conception and design of the study; analysis and interpretation of data; revising of the manuscript; providing intellectual content of critical importance to the work described; final approval of the manuscript. * Van Buren and Mooijaart (shared last authors) contributed equally to this work.
Conflicts of Interest
M.J.P. van Osch reports receiving research support from Philips. No other competing conflicts of interest, financial or otherwise, are declared by the other authors.
Funding
This study was supported by a grant of Roche Nederland BV. The sponsor did not play a role in the preparation of this article.
References
- 1. Petersen RC, Lopez O, Armstrong MJ, Getchius TS, Ganguli M, Gloss D, Gronseth GS, Marson D, Pringsheim T, Day GS, Sager M, Stevens J, Rae-Grant A. Practice guideline update summary: Mild cognitive impairment: Report of the Guideline Development, Dissemination, and Implementation Subcommittee of the American Academy of Neurology. Neurology. 2018; 90:126–35. https://doi.org/10.1212/WNL.0000000000004826 [PubMed]
- 2. Pippias M, Stel VS, Abad Diez JM, Afentakis N, Herrero-Calvo JA, Arias M, Tomilina N, Bouzas Caamaño E, Buturovic-Ponikvar J, Čala S, Caskey FJ, Castro de la Nuez P, Cernevskis H, et al. Renal replacement therapy in Europe: a summary of the 2012 ERA-EDTA Registry Annual Report. Clin Kidney J. 2015; 8:248–61. https://doi.org/10.1093/ckj/sfv014 [PubMed]
- 3. Saran R, Li Y, Robinson B, Ayanian J, Balkrishnan R, Bragg-Gresham J, Chen JT, Cope E, Gipson D, He K, Herman W, Heung M, Hirth RA, et al. US Renal Data System 2014 Annual Data Report: Epidemiology of Kidney Disease in the United States. Am J Kidney Dis. 2015 (1 Suppl 1); 66:Svii, S1–305. https://doi.org/10.1053/j.ajkd.2015.05.001 [PubMed]
- 4. Saran R, Robinson B, Abbott KC, Agodoa LY, Bhave N, Bragg-Gresham J, Balkrishnan R, Dietrich X, Eckard A, Eggers PW, Gaipov A, Gillen D, Gipson D, et al. US Renal Data System 2017 Annual Data Report: Epidemiology of Kidney Disease in the United States. Am J Kidney Dis. 2018 (3 Suppl 1); 71:A7. https://doi.org/10.1053/j.ajkd.2018.01.002 [PubMed]
- 5. Thompson CS, Hakim AM. Living beyond our physiological means: small vessel disease of the brain is an expression of a systemic failure in arteriolar function: a unifying hypothesis. Stroke. 2009; 40:e322–30. https://doi.org/10.1161/STROKEAHA.108.542266 [PubMed]
- 6. Pantoni L. Cerebral small vessel disease: from pathogenesis and clinical characteristics to therapeutic challenges. Lancet Neurol. 2010; 9:689–701. https://doi.org/10.1016/S1474-4422(10)70104-6 [PubMed]
- 7. Livingston G, Sommerlad A, Orgeta V, Costafreda SG, Huntley J, Ames D, Ballard C, Banerjee S, Burns A, Cohen-Mansfield J, Cooper C, Fox N, Gitlin LN, et al. Dementia prevention, intervention, and care. Lancet. 2017; 390:2673–734. https://doi.org/10.1016/S0140-6736(17)31363-6 [PubMed]
- 8. Ikram MA, Vernooij MW, Hofman A, Niessen WJ, van der Lugt A, Breteler MM. Kidney function is related to cerebral small vessel disease. Stroke. 2008; 39:55–61. https://doi.org/10.1161/STROKEAHA.107.493494 [PubMed]
- 9. Elias MF, Dore GA, Davey A. Kidney disease and cognitive function. Contrib Nephrol. 2013; 179:42–57. https://doi.org/10.1159/000346722 [PubMed]
- 10. Kurella Tamura M, Xie D, Yaffe K, Cohen DL, Teal V, Kasner SE, Messé SR, Sehgal AR, Kusek J, DeSalvo KB, Cornish-Zirker D, Cohan J, Seliger SL, et al. Vascular risk factors and cognitive impairment in chronic kidney disease: the Chronic Renal Insufficiency Cohort (CRIC) study. Clin J Am Soc Nephrol. 2011; 6:248–56. https://doi.org/10.2215/CJN.02660310 [PubMed]
- 11. Cecelja M, Chowienczyk P. Dissociation of aortic pulse wave velocity with risk factors for cardiovascular disease other than hypertension: a systematic review. Hypertension. 2009; 54:1328–36. https://doi.org/10.1161/HYPERTENSIONAHA.109.137653 [PubMed]
- 12. de Roos A, van der Grond J, Mitchell G, Westenberg J. Magnetic Resonance Imaging of Cardiovascular Function and the Brain: Is Dementia a Cardiovascular-Driven Disease? Circulation. 2017; 135:2178–95. https://doi.org/10.1161/CIRCULATIONAHA.116.021978 [PubMed]
- 13. Mitchell GF, van Buchem MA, Sigurdsson S, Gotal JD, Jonsdottir MK, Kjartansson Ó, Garcia M, Aspelund T, Harris TB, Gudnason V, Launer LJ. Arterial stiffness, pressure and flow pulsatility and brain structure and function: the Age, Gene/Environment Susceptibility—Reykjavik study. Brain. 2011; 134:3398–407. https://doi.org/10.1093/brain/awr253 [PubMed]
- 14. Almeida OP, Garrido GJ, Beer C, Lautenschlager NT, Arnolda L, Flicker L. Cognitive and brain changes associated with ischaemic heart disease and heart failure. Eur Heart J. 2012; 33:1769–76. https://doi.org/10.1093/eurheartj/ehr467 [PubMed]
- 15. Hoth KF, Poppas A, Moser DJ, Paul RH, Cohen RA. Cardiac dysfunction and cognition in older adults with heart failure. Cogn Behav Neurol. 2008; 21:65–72. https://doi.org/10.1097/WNN.0b013e3181799dc8 [PubMed]
- 16. Cacciatore F, Abete P, Ferrara N, Calabrese C, Napoli C, Maggi S, Varricchio M, Rengo F, and Osservatorio Geriatrico Campano Study Group. Congestive heart failure and cognitive impairment in an older population. J Am Geriatr Soc. 1998; 46:1343–48. https://doi.org/10.1111/j.1532-5415.1998.tb05999.x [PubMed]
- 17. Duschek S, Schandry R. Reduced brain perfusion and cognitive performance due to constitutional hypotension. Clin Auton Res. 2007; 17:69–76. https://doi.org/10.1007/s10286-006-0379-7 [PubMed]
- 18. Washida N, Wakino S, Hayashi K, Kuwahara T, Itoh H. Brachial-ankle pulse wave velocity predicts silent cerebrovascular diseases in patients with end-stage renal diseases. J Atheroscler Thromb. 2010; 17:165–72. https://doi.org/10.5551/jat.2097 [PubMed]
- 19. Tasmoc A, Donciu MD, Veisa G, Nistor I, Covic A. Increased arterial stiffness predicts cognitive impairment in hemodialysis patients. Hemodial Int. 2016; 20:463–72. https://doi.org/10.1111/hdi.12406 [PubMed]
- 20. Angermann S, Baumann M, Wassertheurer S, Mayer CC, Steubl D, Hauser C, Suttmann Y, Reichelt AL, Satanovskij R, Lorenz G, Lukas M, Haller B, Heemann U, et al. Pulse wave velocity is associated with cognitive impairment in hemodialysis patients. Clin Sci (Lond). 2017; 131:1483–93. https://doi.org/10.1042/CS20170087 [PubMed]
- 21. Karasavvidou D, Boutouyrie P, Kalaitzidis R, Kettab H, Pappas K, Stagikas D, Antonakis N, Tsalikakis D, Elisaf M, Laurent S. Arterial damage and cognitive decline in chronic kidney disease patients. J Clin Hypertens (Greenwich). 2018; 20:1276–84. https://doi.org/10.1111/jch.13350 [PubMed]
- 22. Singer J, Trollor JN, Baune BT, Sachdev PS, Smith E. Arterial stiffness, the brain and cognition: a systematic review. Ageing Res Rev. 2014; 15:16–27. https://doi.org/10.1016/j.arr.2014.02.002 [PubMed]
- 23. Bossola M, Laudisio A, Antocicco M, Tazza L, Colloca G, Tosato M, Zuccalà G. Cognitive performance is associated with left ventricular function in older chronic hemodialysis patients: result of a pilot study. Aging Clin Exp Res. 2014; 26:445–51. https://doi.org/10.1007/s40520-013-0191-x [PubMed]
- 24. Deshields TL, McDonough EM, Mannen RK, Miller LW. Psychological and cognitive status before and after heart transplantation. Gen Hosp Psychiatry. 1996 (6 Suppl); 18:62S–69S. https://doi.org/10.1016/S0163-8343(96)00078-3 [PubMed]
- 25. Dixit NK, Vazquez LD, Cross NJ, Kuhl EA, Serber ER, Kovacs A, Dede DE, Conti JB, Sears SF. Cardiac resynchronization therapy: a pilot study examining cognitive change in patients before and after treatment. Clin Cardiol. 2010; 33:84–88. https://doi.org/10.1002/clc.20710 [PubMed]
- 26. Gruhn N, Larsen FS, Boesgaard S, Knudsen GM, Mortensen SA, Thomsen G, Aldershvile J. Cerebral blood flow in patients with chronic heart failure before and after heart transplantation. Stroke. 2001; 32:2530–33. https://doi.org/10.1161/hs1101.098360 [PubMed]
- 27. Jefferson AL. Cardiac output as a potential risk factor for abnormal brain aging. J Alzheimers Dis. 2010; 20:813–21. https://doi.org/10.3233/JAD-2010-100081 [PubMed]
- 28. Satizabal CL, Beiser AS, Chouraki V, Chêne G, Dufouil C, Seshadri S. Incidence of Dementia over Three Decades in the Framingham Heart Study. N Engl J Med. 2016; 374:523–32. https://doi.org/10.1056/NEJMoa1504327 [PubMed]
- 29. Berkhout-Byrne N, Kallenberg MH, Gaasbeek A, Rabelink TJ, Hammer S, van Buchem MA, van Osch MJ, Kroft LJ, Boom H, Mooijaart SP, van Buren M. The Cognitive decline in Older Patients with End stage renal disease (COPE) study - rationale and design. Curr Med Res Opin. 2017; 33:2057–64. https://doi.org/10.1080/03007995.2017.1341404 [PubMed]
- 30. Du Bois D, Du Bois EF. A formula to estimate the approximate surface area if height and weight be known. 1916. Nutrition. 1989; 5:303–11. https://doi.org/10.1001/archinte.1916.00080130010002 [PubMed]
- 31. Wardlaw JM, Smith EE, Biessels GJ, Cordonnier C, Fazekas F, Frayne R, Lindley RI, O’Brien JT, Barkhof F, Benavente OR, Black SE, Brayne C, Breteler M, et al, and STandards for ReportIng Vascular changes on nEuroimaging (STRIVE v1). Neuroimaging standards for research into small vessel disease and its contribution to ageing and neurodegeneration. Lancet Neurol. 2013; 12:822–38. https://doi.org/10.1016/S1474-4422(13)70124-8 [PubMed]
- 32. Folstein MF, Folstein SE, McHugh PR. “Mini-mental state”. A practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res. 1975; 12:189–98. https://doi.org/10.1002/(sici)1099-1166(199805)13:5<285::aid-gps753>3.3.co;2-m [PubMed]
- 33. Suhr J, Grace J, Allen J, Nadler J, McKenna M. Quantitative and qualitative performance of stroke versus normal elderly on six clock drawing systems. Arch Clin Neuropsychol. 1998; 13:495–502. https://doi.org/10.1093/arclin/13.6.495 [PubMed]
- 34. Adunsky A, Fleissig Y, Levenkrohn S, Arad M, Noy S. A comparative study of Mini-Mental Test, Clock Drawing task and Cognitive-FIM in evaluating functional outcome of elderly hip fracture patients. Clin Rehabil. 2002; 16:414–19. https://doi.org/10.1191/0269215502cr514oa [PubMed]
- 35. Rey A. L’examen psychologique dans les cas d’encephalopathie traumatique. Paris, France: Presses Universitaires de France; 1964.
- 36. Brand N, Jolles J. Learning and retrieval rate of words presented auditorily and visually. J Gen Psychol. 1985; 112:201–10. https://doi.org/10.1080/00221309.1985.9711004 [PubMed]
- 37. Van der Elst W, van Boxtel MP, van Breukelen GJ, Jolles J. Rey’s verbal learning test: normative data for 1855 healthy participants aged 24-81 years and the influence of age, sex, education, and mode of presentation. J Int Neuropsychol Soc. 2005; 11:290–302. https://doi.org/10.1017/S1355617705050344 [PubMed]
- 38. Lindeboom J, Schmand B, Tulner L, Walstra G, Jonker C. Visual association test to detect early dementia of the Alzheimer type. J Neurol Neurosurg Psychiatry. 2002; 73:126–33. https://doi.org/10.1136/jnnp.73.2.126 [PubMed]
- 39. Arnett JA, Labovitz SS. Effect of physical layout in performance of the Trail Making Test. Psychol Assess. 1995; 7:220–21. https://doi.org/10.1037/1040-3590.7.2.220
- 40. Schmand BHP, de Koning I. Normen voor Stroop kleur-woord tests, Trail Making test en Story Recall van de Rivermead Behavioural Memory Test. Amsterdam: Neuropsychology of the Netherlands Institute of Psychologists, 2003.
- 41. Houx PJ, Jolles J, Vreeling FW. Stroop interference: aging effects assessed with the Stroop Color-Word Test. Exp Aging Res. 1993; 19:209–24. https://doi.org/10.1080/03610739308253934 [PubMed]
- 42. Stroop J. Studies of interference in serial verbal reaction. J Exp Psychol. 1935; 18:643–62. https://doi.org/10.1037/h0054651
- 43. Van der Elst W, Van Boxtel MP, Van Breukelen GJ, Jolles J. The Stroop color-word test: influence of age, sex, and education; and normative data for a large sample across the adult age range. Assessment. 2006; 13:62–79. https://doi.org/10.1177/1073191105283427 [PubMed]
- 44. Klein M, Ponds RW, Houx PJ, Jolles J. Effect of test duration on age-related differences in Stroop interference. J Clin Exp Neuropsychol. 1997; 19:77–82. https://doi.org/10.1080/01688639708403838 [PubMed]
- 45. Smith A. The Symbol Digit Modalities Test. A neuropsychologic test for economic screening of learning and other cerebral disorders. Learning Disorders. 1968; 3:83–91.
- 46. van der Elst W, van Boxtel MP, van Breukelen GJ, Jolles J. The Letter Digit Substitution Test: normative data for 1,858 healthy participants aged 24-81 from the Maastricht Aging Study (MAAS): influence of age, education, and sex. J Clin Exp Neuropsychol. 2006; 28:998–1009. https://doi.org/10.1080/13803390591004428 [PubMed]
- 47. Williams B, Mancia G, Spiering W, Agabiti Rosei E, Azizi M, Burnier M, Clement DL, Coca A, de Simone G, Dominiczak A, Kahan T, Mahfoud F, Redon J, et al, and ESC Scientific Document Group. 2018 ESC/ESH Guidelines for the management of arterial hypertension. Eur Heart J. 2018; 39:3021–104. https://doi.org/10.1093/eurheartj/ehy339 [PubMed]
- 48. Ponikowski P, Voors AA, Anker SD, Bueno H, Cleland JG, Coats AJ, Falk V, González-Juanatey JR, Harjola VP, Jankowska EA, Jessup M, Linde C, Nihoyannopoulos P, et al. 2016 ESC Guidelines for the Diagnosis and Treatment of Acute and Chronic Heart Failure. Rev Esp Cardiol (Engl Ed). 2016; 69:1167. https://doi.org/10.1016/j.rec.2016.11.005 [PubMed]


