Introduction
Due to the advances in the detection of early-stage cancers and cancer treatment, the population of cancer survivors has increased by approximately 4 folds in the United States in the past 30 years [1–3]. Almost two-thirds of cancer survivors live more than 5 years after the initial diagnosis, which increases the risk of developing a second primary malignancy (SPM) [4–8]. A SPM is defined as a cancer which arises independently in a new organ or tissue at least 2 months after the diagnosis of the prior primary cancer. Over 10% of younger adult cancer patients and around 25% of older adult cancer patients have been diagnosed with a SPM [9, 10]. It has been reported to be associated with genetic susceptibility [11, 12], the carcinogenic adverse effects of cancer treatment [13], and behavioral risk factors such as smoking and alcohol intake [14–18].
Many studies have examined the risk of a SPM in patients with different types of prior cancer, such as lung, testis, head and neck, and thyroid cancers [19–21]. The prognosis of patients with stage IV lung cancer as a SPM was not affected by a prior cancer [22]. The prevalence of colorectal adenomas in breast cancer survivors is similar to that in patients with single colorectal adenomas [23]. Garg et al. [24] reported that early diagnosis and the absence of recurrence in patients with prior breast cancer who had abdominal carcinomatosis were significantly associated with the development of ovarian/peritoneal cancer as a SPM. Prior breast cancer and tamoxifen exposure did not affect the prognosis of women with uterine papillary serous carcinoma as a SPM [25]. However, the risk of breast cancer as a SPM in patients with a prior cancer and the cancer-specific survival for these patients are not known.
Thus, in the present study, we used data from the Surveillance, Epidemiology, and End Results (SEER) database to analyze the outcomes of patients who developed breast cancer as a SPM. This information may help guide long-term surveillance strategies for patients.
Results
Baseline characteristics of the primary cohort
The median (interquartile range, IQR) age at diagnosis of the prior cancer was 66 (20–96) years, and that of subsequent breast cancer was 68 (20–99) years. Most patients were Caucasian (1,097, 83.8%), and only a few (111, 8.5%) were black. Overall, 920 (70.2%) patients had their prior cancers diagnosed at TNM stage I-II, and 1094 (83.5%) had their breast cancers diagnosed at TNM stage I-II. The median (IQR) interval between 2 diagnoses was 17 (2–71) months. By the end of 2017, the patients were followed up for a median of 17 months (IQR, 0-69 months), and 219 (16.7%) patients died during follow-up: 153 (69.9%) died of cancer, and 66 (30.1%) died of other causes (Table 1).
Table 1. Summary description of demographic and clinical factors.
| At prior cancer diagnosis | At breast cancer diagnosis | |||
| Variable | Value | Variable | Value | |
| Age, years | Age, years | |||
| Mean(range) | 65.8(20-96) | Mean(range) | 67.8(20-99) | |
| Median(IQR) | 66 | Median(IQR) | 68 | |
| Race,n(%) | Race,n(%) | |||
| White | 1098(83.8%) | White | 1098(83.8%) | |
| Black | 111(8.5%) | Black | 111(8.5%) | |
| Other | 101(7.7%) | Other | 101(7.7%) | |
| Marital status | Marital status | |||
| Married | 997(76.1%) | Married | 916(69.9%) | |
| Unmarried | 313(23.9%) | Unmarried | 394(30.1%) | |
| TNM stage,n(%) | TNM stage,n(%) | |||
| I-II | 919(70.2%) | I-II | 1094(83.5%) | |
| III-IV | 391(29.8%) | III-IV | 216(16.5%) | |
| Interval between diagnoses, months | Follow up from BC diagnosis to death or end of study(months) | |||
| Mean(range) | 21.64 | Mean(range) | 20.96 | |
| Median(IQR) | 17(2–71) | Median(IQR) | 17(0-69) | |
Associations of demographic and clinicopathologic factors with breast cancer deaths in the primary cohort
The rates of high-grade disease (32.3% vs. 23.1%, P < 0.001), T3–4/N0/M0 disease (11.3% vs. 4.4%, P = 0.014), and distant disease (33.9% vs. 27.5%, P = 0.023) of breast cancer were significantly higher in patients who died of breast cancer than in those who died of a prior cancer, and the rates of metastasis of prior cancer (11.3% vs. 25.3%, P = 0.011) and administration of breast cancer treatment (37.1% vs. 63.4%, P = 0.018) were significantly lower in patients who died of breast cancer. Patients who died of breast cancer had a longer diagnosis interval (17.0 vs. 11.4 months, P = 0.040) and were older (73.5 vs. 69.1 years, P = 0.035) than those who died of a prior cancer (Table 2). Breast cancer treatment was associated with a decreased risk of breast cancer-specific mortality (BCSM) (hazard ratio = 0.695, 95% confidence interval [CI] = 0.586–0.725, P < 0.001), but not associated with a decreased risk of non-BCSM (Figure 2).
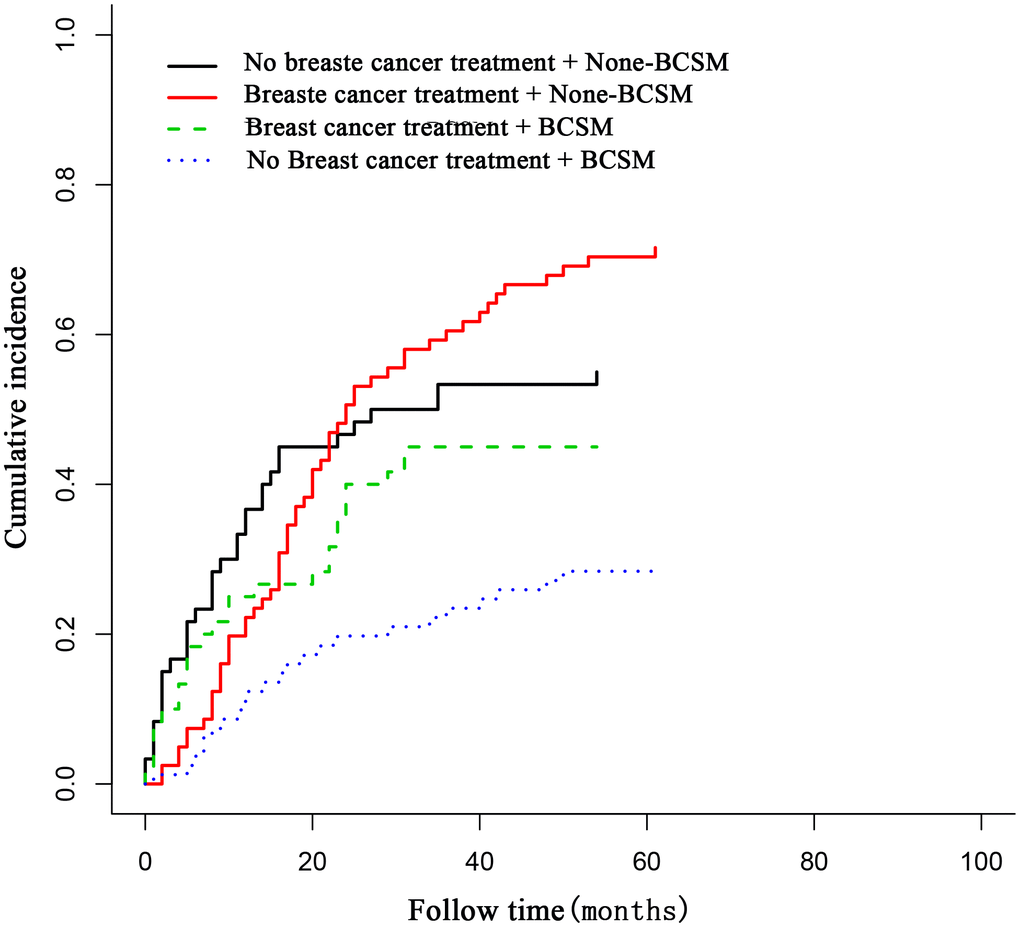
Figure 2. The association of breast cancer treatment with breast cancer deaths in patients with breast cancer as a second primary cancer. Breast cancer treatment was associated with decreased breast cancer deaths in these patients.
Table 2. Clinical and demographic factors associated with breast cancer death vs prior malignancy death.
| Character | Died from Breast cancer | Died from prior cancer | P value |
| Number of patients(%) | 62 | 91 | |
| High grade | 32.3% | 23.1% | <0.001 |
| Breast cancer stage T3-4, N0/M0 | 11.3% | 4.4% | 0.014 |
| Tx/N1-3/Mx or Tx/Nx/M1 Breast cancer | 33.9% | 27.5% | 0.023 |
| Tx/N1/Mx or Tx/Nx/M1 Prior cancer | 11.3% | 25.3% | 0.011 |
| Breast cancer treated | 37.1% | 63.4% | 0.018 |
| Interval between diagnoses, months | 17.0 | 11.4 | 0.04 |
| Age at Breast cancer diagnosis, years | 73.5 | 69.1 | 0.03 |
Survival of patients with breast cancer as the prior cancer or subsequent primary cancer in the second cohort
In the second cohort, 63,761 patients had breast cancer as their only cancer (primary breast cancer, PBC), and 9,955 had breast cancer as the second primary cancer (subsequent breast cancer, SBC). The 5-year BCSS and OS rates were significantly lower in patients with SBC than in those with PBC (BCSS: 91.0% vs. 94.0%, P < 0.001, Figure 4A; OS: 91.0% vs. 93.8%, P < 0.001, Figure 4B).
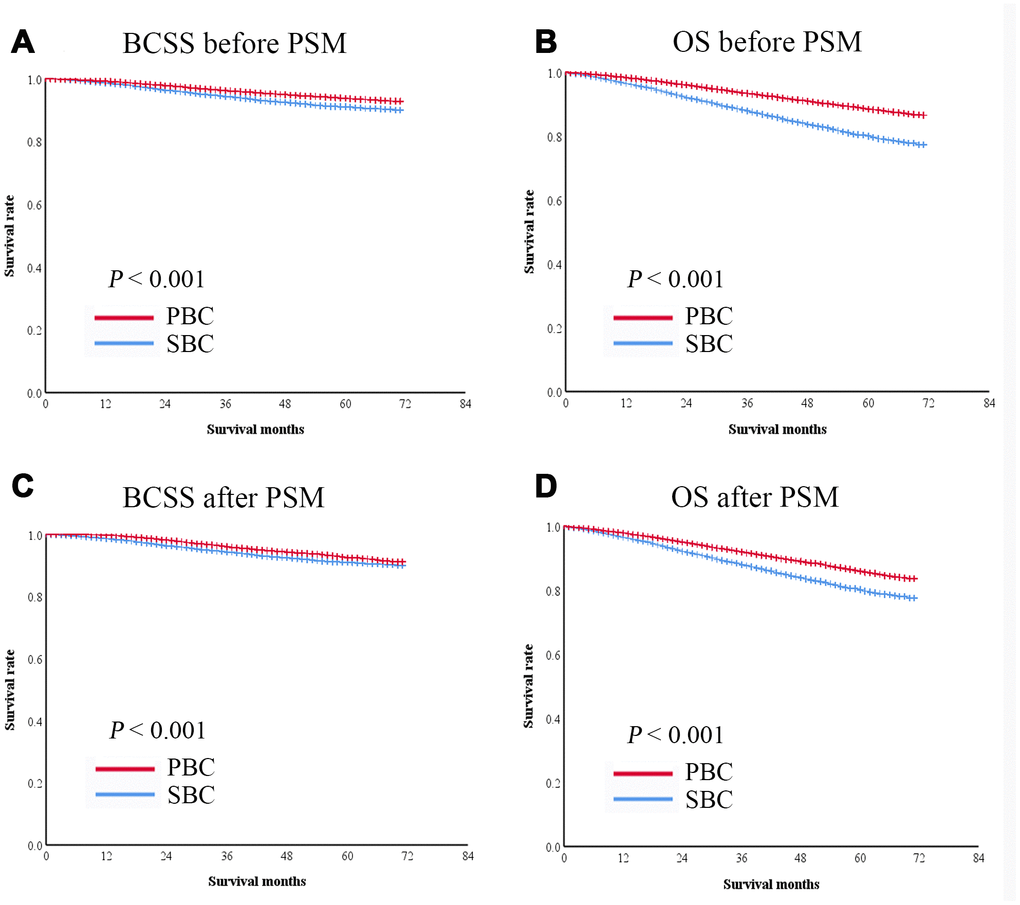
Figure 4. Kaplan-Meier survival curves of patients with breast cancer as the second primary cancer or the prior cancer. (A) BCSS was significantly shorter in patients with breast cancer as the second primary cancer than in those with breast cancer as the prior cancer in the entire cohort. (B) OS was significantly shorter in patients with breast cancer as the second primary cancer than in those with breast cancer as the prior cancer in the entire cohort. After PSM, both BCSS (C) and OS (D) were significantly lower in patients with breast cancer as the second primary cancer than in those with breast cancer as the prior cancer.
A 1:1 PSM was performed to balance the baseline characteristics of patients with SBC and PBC. After matching, 9,845 pairs of patients were included in the analysis (Table 3). The survival of the matched groups was in consistent with the survival of the entire cohort, the 5-year BCSS and OS rates were significantly lower in patients with SBC than in those with PBC (BCSS: 91.2% vs. 93.6%, P < 0.001, Figure 4C; OS: 80.5% vs. 86.1%, P < 0.001, Figure 4D).
Table 3. Baseline characteristics of patients with PBC or SBC from the SEER database 2010–2011.
| Variables | Data before PSM | P value | Data after PSM | P value | ||
| PBC (63761) | SBC (9955) | PBC (9845) | SBC (9845) | |||
| Age(y), % | <0.001 | >0.99 | ||||
| 18-45 | 9485 | 615 | 600 | 600 | ||
| 46-65 | 32848 | 4014 | 3995 | 3995 | ||
| ≥65 | 21428 | 5326 | 5250 | 5250 | ||
| Race, % | <0.001 | >0.99 | ||||
| White | 50893 | 8388 | 8347 | 8347 | ||
| Black | 6641 | 895 | 854 | 854 | ||
| Others | 6227 | 672 | 644 | 644 | ||
| Marital status, % | <0.001 | >0.99 | ||||
| Married | 38474 | 5392 | 5340 | 5340 | ||
| Not married | 25287 | 4563 | 4505 | 4505 | ||
| Tumor differentiation, % | <0.001 | >0.99 | ||||
| I–II | 42439 | 7066 | 6984 | 6984 | ||
| III–IV | 21322 | 2889 | 2861 | 2861 | ||
| TNM Stage, % | <0.001 | >0.99 | ||||
| I | 31769 | 5963 | 5897 | 5897 | ||
| II–III | 31992 | 3992 | 3948 | 3948 | ||
| Breast subtype, % | <0.001 | >0.99 | ||||
| HER2-/HR+ | 46913 | 7508 | 7469 | 7469 | ||
| HER2+/HR+ | 6377 | 799 | 779 | 779 | ||
| HER2+/HR- | 2761 | 373 | 354 | 354 | ||
| Triple-negative | 7710 | 1275 | 1243 | 1243 | ||
| Surgery, % | <0.001 | >0.99 | ||||
| Lumpectomy | 37756 | 4679 | 4644 | 4644 | ||
| Mastectomy | 26005 | 5276 | <0.001 | 5201 | 5201 | |
| Radiation, % | >0.99 | |||||
| Yes | 27815 | 6298 | 6221 | 6221 | ||
| No/ Unknown | 35946 | 3657 | <0.001 | 3624 | 3624 | |
| Chemotherapy, % | >0.99 | |||||
| Yes | 36349 | 6847 | 6797 | 6797 | ||
| No/unknown | 27412 | 3108 | 3048 | 3048 | ||
Survival of patients with hormone receptor (HR)-positive breast cancer as the prior cancer or second primary cancer
Taking hormone receptor statuses into consideration, breast cancer as the subsequent primary cancer (SBC) was significantly associated with short BCSS and OS in HER2-/HR+ patients (both P < 0.001, Figure 5A, 5B) and HER2+/HR+ patients (P = 0.001and P < 0.001) (Figure 5E, 5F). However, no such associations were observed in HER2+/HR- and triple-negative patients (Figure 5C, 5D, 5G, 5H).
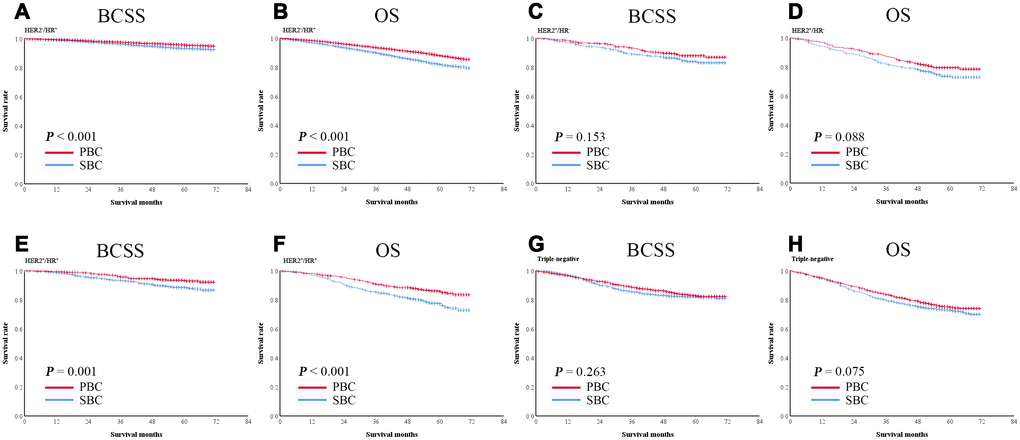
Figure 5. Kaplan-Meier survival curves of patients with breast cancer as the second primary cancer or the prior cancer and with different hormone receptor statuses. Breast cancer as the second primary cancer was significantly associated with shorter BCSS and OS in hormone receptor-positive subgroups (A–H).
Factors associated with survival of patients with breast cancer as the primary cancer or subsequent breast cancer
We used univariate and multivariate analyses to determine clinicopathological factors which associated with survival. Univariate analysis showed that age, race, marital status, tumor differentiation, TNM stage, breast cancer subtype, breast cancer as the prior or second primary cancer (PBC vs SBC), surgery, radiotherapy, and chemotherapy were significant risk factors for both BCSS and OS (all P < 0.001) (Table 4). Cox regression multivariate analysis indicated that marital status, tumor differentiation, TNM, breast cancer subtype, breast cancer as the primary or subsequent breast cancer (PBC vs SBC), and radiotherapy were independent prognostic factors for both BCSS and OS, race was an independent prognostic factor for BCSS, and age and chemotherapy were independent prognostic factors for OS (all P < 0.05) (Table 4).
Table 4. Univariate and Multivariate Cox regression model analysis of BCSS and OS.
| Category | BCSS | OS | |||||||
| 5 years | Univariate | HR (95% CI) | Multivariate | 5 years | Univariate | HR (95% CI) | Multivariate | ||
| Age | P<0.001 | P<0.001 | |||||||
| 18-45 | 90% | Reference | 87.6% | Reference | |||||
| 45-65 | 93.6% | 0.899 (0.732-1.104) | 0.309 | 90.7% | 0.883 (0.737-1.059) | 0.179 | |||
| >65 | 91.9% | 1.368 (1.118-1.674) | 0.002 | 77.3% | 2.167 (1.817-2.586) | P<0.001 | |||
| Race | P<0.001 | P<0.001 | |||||||
| White | 92.3% | Reference | 83.5% | Reference | |||||
| Black | 86.2% | 1.261 (1.083-1.469) | 0.003 | 77.1% | 1.155 (1.034-1.291) | 0.011 | |||
| Other | 94.5% | 0.690 (0.535-0.890) | 0.004 | 88.4% | 0.749 (0.633-0.886) | 0.001 | |||
| Marital status | P<0.001 | P<0.001 | |||||||
| Married | 93.9% | Reference | 88.3% | Reference | |||||
| Not married | 90.7% | 1.307 (1.171-1.459) | P<0.001 | 77.4% | 1.606 (1.492-1.728) | P<0.001 | |||
| Tumor differentiation | P<0.001 | P<0.001 | |||||||
| I–II | 95.6% | Reference | 86.1% | Reference | |||||
| III–IV | 84.8% | 2.143 (1.891-2.429) | P<0.001 | 76.4% | 1.479 (1.361-1.6078) | P<0.001 | |||
| TNM Stage | P<0.001 | P<0.001 | |||||||
| I | 97.0% | Reference | 88.9% | Reference | |||||
| II–III | 85.4% | 4.438 (3.903-5.045) | P<0.001 | 74.8% | 2.553 (2.369-2.752) | P<0.001 | |||
| Breast subtype | P<0.001 | P<0.001 | |||||||
| HER2-/HR+ | 94.6% | Reference | 85.3% | Reference | |||||
| HER2+/HR+ | 90.9% | 1.246 (1.031-1.505) | 0.023 | 76.5% | 1.283 (1.130-1.458) | P<0.001 | |||
| HER2+/HR- | 85.9% | 1.517 (1.207-1.096) | P<0.001 | 81.8% | 1.557 (1.314-1.845) | P<0.001 | |||
| Triple-negative | 82.6% | 2.209 (1.769-2.327) | P<0.001 | 74.2% | 1.711 (1.545-1.894) | P<0.001 | |||
| Diagnosis | P<0.001 | P<0.001 | |||||||
| PBC | 93.7% | Reference | 86.1% | Reference | |||||
| SBC | 91.2% | 1.444 (1.297-1.607) | P<0.001 | 80.5% | 1.493 (1.392-1.602) | P<0.001 | |||
| Surgery | P<0.001 | P=0.272 | |||||||
| Lumpectomy | 94.0% | 83.6% | |||||||
| Mastectomy | 91.1% | 83.0% | |||||||
| Radiation | P<0.001 | P<0.001 | |||||||
| Yes | 94.4% | Reference | 88.9% | Reference | |||||
| No/ Unknown | 91.3% | 1.517 (1.349-1.707) | P<0.001 | 80.0% | 1.768 (1.630-1.918) | P<0.001 | |||
| Chemotherapy | P<0.001 | P<0.001 | |||||||
| Yes | 89.0% | 85.3% | Reference | ||||||
| No/unknown | 94.1% | 82.4% | 1.424 (1.300-1.559) | P<0.001 | |||||
As shown on Figure 6, compared with breast cancer as the primary cancer (PBC), breast cancer as the SBC was associated with shorter BCSS when the age at diagnosis was > 65 years (hazard ratio = 1.36, 95% CI: 1.18–1.81), with grade III and IV tumors (hazard ratio = 1.26, 95% CI: 1.10–1.45), with triple-negative disease (hazard ratio = 1.12, 95% CI: 0.919–1.36), mastectomy (hazard ratio = 1.33, 95% CI: 1.16–1.54), radiotherapy (hazard ratio = 1.36, 95% CI: 1.11–1.50), and chemotherapy (hazard ratio = 1.40, 95% CI: 1.19–1.63) and was associated with shorter OS when the age at diagnosis was > 65 years (hazard ratio = 1.40, 95% CI: 1.29–1.52), with grades III and IV tumors (hazard ratio = 1.33, 95% CI: 1.20–1.49), HER2+/HR- disease (hazard ratio = 1.31, 95% CI: 0.96–1.79), triple-negative breast cancer (hazard ratio = 1.15, 95% CI: 0.99–1.35), and mastectomy (hazard ratio = 1.31, 95% CI: 1.19–1.44). These data suggest that breast cancer as the second primary cancer affects the prognosis of patients who have had a prior cancer.
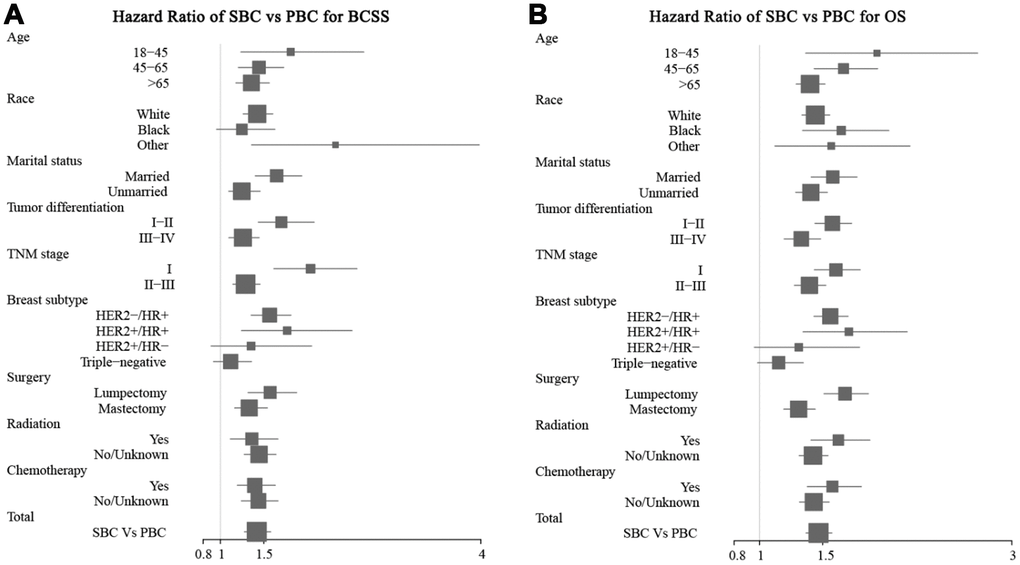
Figure 6. Forest plot of the hazard ratios for survival of patients with breast cancer as the second primary cancer or the prior cancer. Breast cancer as the second primary cancer was associated with short BCSS (A) and OS (B) in most of the subgroups.
Discussion
The cancer survivor population is rapidly growing, and subsequently the number of patients with multiple primary or multi-organ cancers is also increasing [26]. In the US, the cancer survivor population presented a 2% annual increase, and approximately 18% of cancer cases were developed after the prior cancer according to SEER data [27]. The risk of developing an SPM among cancer survivors was reported to be as high as that in the general population [28]. The risk of breast cancer as a second primary cancer is especially high [29]. However, the clinicopathological characteristics and survival of patients with this disease remain largely unknown.
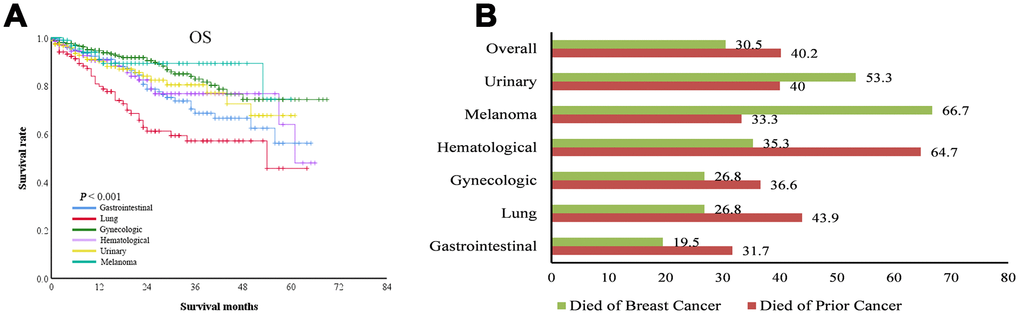
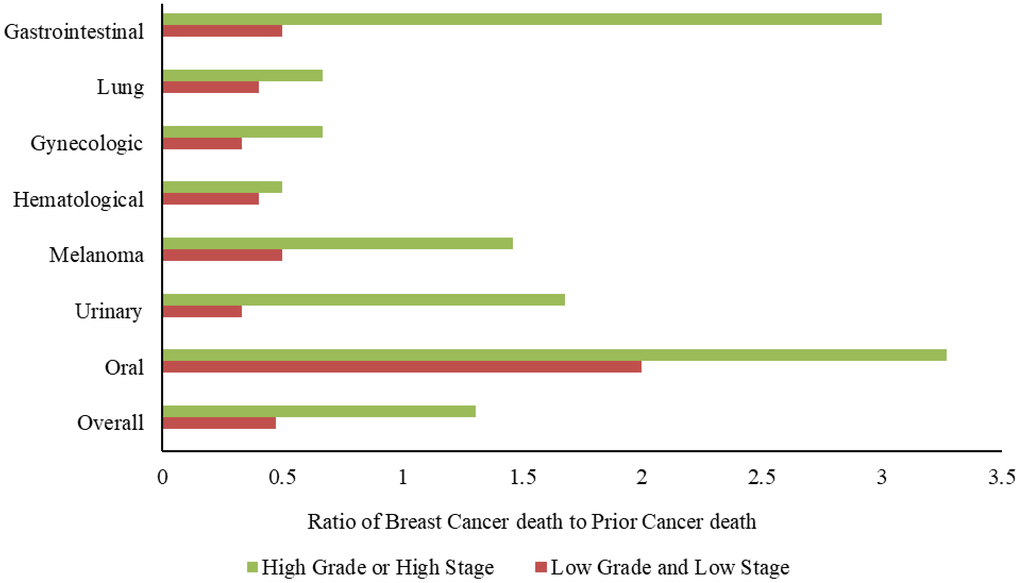
In this population-based study, we found that gynecologic cancers, gastrointestinal cancer, urinary tract cancers, hematological cancers, lung cancer, and melanoma were the most common types of prior cancer. Survivors of gynecologic cancers such as ovarian cancer tended to have a high risk of developing breast cancer as their second cancer. This may be explained by that hereditary breast cancer and ovarian cancer are the most common cancers with genetic predisposition. BRCA1 and/or BRCA2 mutations are frequently detected in both cancer types. The risk of developing breast cancer is increased among patients with prior ovarian cancer harboring BRCA mutations. The 10-year occurrence rates of breast cancer among patients with prior ovarian cancer were 12% in BRCA1 mutation carriers and 2% in BRCA2 mutation carriers [30, 31]. Therefore, annual screening with breast magnetic resonance imaging is recommended for patients with BRCA1 and BRCA2 mutations. In the present study, 30.5% of women with breast cancer as a SPM died of breast cancer, and 40.2% of them died of prior gynecologic cancer. However, among patients with different types of prior cancer, those with prior lung cancer had the shortest OS, those with prior oral cancer were more likely to die of breast cancer regardless of tumor grade and stage, and patients with gastrointestinal cancer, melanoma, and urinary tract cancer were more likely to die of breast cancer when they had high-grade or advanced stage breast cancer. In the whole cohort, 51.8% of patients with high-grade or stage cT3–4/N0/M0 breast cancer died of breast cancer. These data indicate that breast cancer as the second primary cancer remains a main cause of death in women with a prior cancer.
Our results showed that breast cancer as the second primary cancer was associated with short survival. The 5-year BCSS and OS rates were significantly lower in patients with breast cancer as the second primary cancer than in those with breast cancer as the prior cancer in both the standard and PSM analyses, these results are similar to those observed by Zhou et al. [32]. A subgroup survival analysis also showed that HER2−/HR+ and HER2+/HR+ patients with breast cancer as the second primary cancer had shorter BCSS and OS than other subgroup. As such, breast cancer patients with prior cancers must be carefully considered for clinical trials. Univariate and multivariate analyses indicated that breast cancer as the second primary cancer was an independent risk factor for BCSS and OS. The high occurrence and death rates of breast cancer in cancer survivors may likely be attributed to BRCA1 and BRCA2 mutations as well as chemotherapy and radiotherapy for prior cancer. Patients who received underwent chemotherapy for prior cancer may not respond well to systemic treatment for their second primary cancer due to decreased efficacy and tolerability after prior systemic treatment [33, 34]. Interestingly, Fine and Gray’s regression analysis showed that breast cancer treatment was associated with a decreased risk of BCSM. The ratio of breast cancer deaths to prior cancer deaths was significantly higher in patients with diagnoses interval ≥ 3 years than in those with the interval < 3 years. Although patients’ breast cancer as the second primary cancer had short survival, breast cancer treatment can still prolong survival.
Some limitations in the present study need to be mentioned. First, as a retrospective study, selection bias was inevitable. Although we performed PSM to minimize such bias, a PSM analysis is also vulnerable to hidden biases, and residual confounding factors could not be entirely ruled out. Second, as the data were retrieved from the SEER database, we could not control treatment-related factors of the prior cancer, which may alter findings related to second cancer incidence and survival. Therefore, further studies with more data are required to validate our findings.
Conclusions
Survivors of gynecologic cancers such as ovarian cancer tend to have a high risk of breast cancer. Breast cancer as a second primary cancer, especially locally advanced or high-grade breast cancer, is a significant cause of death in patients with a prior cancer. However, breast cancer treatment decreased the risk of BCSM in these patients. Considering the increases in cancer survivors and deaths related to breast cancer as the second primary cancer, effective detection and treatment strategies is warranted to be investigated in this population.
Materials and Methods
The SEER database is a population-based cancer registry sponsored by the US National Cancer Institute (NCI). The SEER program collects data regarding patient demographic characteristics, cancer incidence, treatment, and survival. The current study included 2 independent patient cohorts derived from the SEER database for separate analyses.
Primary cohort
The principal analysis of this study used the multiple primary-standard incidence ratio function of SEER*Stat version 8.1.5 software. Using the software to select patients from the registry that covered the 9 regions of the US recognized by the SEER program (covering 9.4% of the US population), we identified 13,011 patients with breast cancer as SPM diagnosed between 2010 and 2015. After excluding patients with bilateral breast cancer, missing information, non-malignant prior neoplasm, multiple malignancies, and diagnosis at autopsy or by death certificate only, 1,310 patients were included in the analysis. The 6 most common types of prior cancer in these 1,310 patients were gynecologic cancer (26.7%) (including ovarian and uterus cancers), gastrointestinal cancer (16.0%) (including gastric, colon, and rectal cancers), urinary tract cancer (11.5%) (including kidney, ureteral, bladder cancers), hematological cancer (10.4%) (including non-Hodgkin lymphoma and leukemia), lung cancer (9.5%), and melanoma (7.9%).
Demographic and clinicopathological data extracted from the SEER database included age, race, marital status, tumor grade, TNM stage, surgical type, and administration of radiotherapy and chemotherapy. Race was divided into white, black, and others. Patients were classified as married or unmarried. The SEER program uses the 7th edition of the American Joint Committee on Cancer (AJCC) TNM staging system. Survival data in the database are presented in the unit of months. We calculated the interval between diagnoses of the prior cancer and breast cancer and the interval from breast cancer diagnosis to death or last follow-up. Each patient was categorized as alive, dead of breast cancer, dead of the prior cancer, or dead of other causes.
Demographic, clinical, and pathological characteristics were compared between patients who died of breast cancer and those who died of the prior cancer using the Student t test, and rank-sum and chi-square analyses. The ratio of the percentage of breast cancer deaths to the percentage of prior cancer deaths was calculated for each prior cancer type, stratified by breast cancer grade and TNM stage. Fine and Gray’s competing risk regression model was used to assess the association of breast cancer treatment with breast cancer-specific mortality (BCSM), with death from prior cancer and other causes setting as competing risks. All 2-sided P values < 0.05 were considered significant. Statistical analyses were conducted using R version 3.2.2 software (R Foundation for Statistical Computing, Vienna, Austria).
Secondary cohort
The secondary cohort included 73,716 patients with stage I-III, histologically confirmed breast cancer identified in the 2010–2011 SEER database, which covered 18 regions of the US (covering 27.8% of the US population). Each patient was categorized as having breast cancer as her only malignancy (primary breast cancer, 63,761 women, 86.5%) or having breast cancer as an SPM (9845 women, 13.5%). Data extracted included age, race, marital status, tumor differentiation, TNM stage, and breast cancer subtype. Patients were assigned to low-, intermediate-, and high-risk groups according to the risk categories of St. Gallen 2007 [35, 36].
The propensity score matching (PSM) method with a ratio of 1:1 was performed to balance the baseline characteristics of patients with breast cancer as the second primary cancer and as the prior cancer [37]. Breast cancer-specific survival (BCSS) was defined as the duration from the diagnosis of breast cancer to death due to breast cancer. Overall survival (OS) was defined as the duration from the diagnosis of breast cancer to death from any cause. Survival between groups was compared using the Kaplan-Meier method. Logistic regression analyses were used to examine the relations between clinicopathological characteristics. A 2-sided P value of < 0.05 was considered significant.
Author Contributions
F.J. and C.Q.Y. conceived and designed the study; M.Y. and L.L.Z. analyzed the data; F.J. drafted the paper; T.Z., M.Y.C., Q.Q.X., S.Y.W., A.L.Z., K.W. reviewed the manuscript. All authors interpreted the data, edited or commented, and approved the final manuscript.
Conflicts of Interest
All the authors declare that they have no conflicts of interest.
Funding
This study was funded by grants from Natural Science Foundation of Guangdong Province (grant number 2017A030313882), Guangdong Basic and Applied Basic Research Foundation (grant number 2020A1515010346) and Guangdong Medical Science and Technology Research Fund (grant number A2019494, A2019252). The Doctor Launch Fund of Guangdong Provincial People's hospital (grant number 2018bq04). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1. de Moor JS, Mariotto AB, Parry C, Alfano CM, Padgett L, Kent EE, Forsythe L, Scoppa S, Hachey M, Rowland JH. Cancer survivors in the United States: prevalence across the survivorship trajectory and implications for care. Cancer Epidemiol Biomarkers Prev. 2013; 22:561–70. https://doi.org/10.1158/1055-9965.EPI-12-1356 [PubMed]
- 2. Donin N, Filson C, Drakaki A, Tan HJ, Castillo A, Kwan L, Litwin M, Chamie K. Risk of second primary malignancies among cancer survivors in the United States, 1992 through 2008. Cancer. 2016; 122:3075–86. https://doi.org/10.1002/cncr.30164 [PubMed]
- 3. Miller KD, Siegel RL, Lin CC, Mariotto AB, Kramer JL, Rowland JH, Stein KD, Alteri R, Jemal A. Cancer treatment and survivorship statistics, 2016. CA Cancer J Clin. 2016; 66:271–89. https://doi.org/10.3322/caac.21349 [PubMed]
- 4. Wood ME, Vogel V, Ng A, Foxhall L, Goodwin P, Travis LB. Second malignant neoplasms: assessment and strategies for risk reduction. J Clin Oncol. 2012; 30:3734–45. https://doi.org/10.1200/JCO.2012.41.8681 [PubMed]
- 5. Travis LB, Curtis RE, Storm H, Hall P, Holowaty E, Van Leeuwen FE, Kohler BA, Pukkala E, Lynch CF, Andersson M, Bergfeldt K, Clarke EA, Wiklund T, et al. Risk of second malignant neoplasms among long-term survivors of testicular cancer. J Natl Cancer Inst. 1997; 89:1429–39. https://doi.org/10.1093/jnci/89.19.1429 [PubMed]
- 6. Henderson TO, Amsterdam A, Bhatia S, Hudson MM, Meadows AT, Neglia JP, Diller LR, Constine LS, Smith RA, Mahoney MC, Morris EA, Montgomery LL, Landier W, et al. Systematic review: surveillance for breast cancer in women treated with chest radiation for childhood, adolescent, or young adult cancer. Ann Intern Med. 2010; 152:444–55; W144–54. https://doi.org/10.7326/0003-4819-152-7-201004060-00009 [PubMed]
- 7. Park SM, Lim MK, Jung KW, Shin SA, Yoo KY, Yun YH, Huh BY. Prediagnosis smoking, obesity, insulin resistance, and second primary cancer risk in male cancer survivors: National Health Insurance Corporation Study. J Clin Oncol. 2007; 25:4835–43. https://doi.org/10.1200/JCO.2006.10.3416 [PubMed]
- 8. Youlden DR, Baade PD. The relative risk of second primary cancers in Queensland, Australia: a retrospective cohort study. BMC Cancer. 2011; 11:83. https://doi.org/10.1186/1471-2407-11-83 [PubMed]
- 9. Murphy CC, Gerber DE, Pruitt SL. Prevalence of prior cancer among persons newly diagnosed with cancer: an initial report from the surveillance, epidemiology, and end results program. JAMA Oncol. 2018; 4:832–36. https://doi.org/10.1001/jamaoncol.2017.3605 [PubMed]
- 10. Lin C, Wu J, Ding S, Goh C, Andriani L, Shen K, Zhu L. Impact of Prior Cancer History on the Clinical Outcomes in Advanced Breast Cancer: A Propensity Score-Adjusted, Population-Based Study. Cancer Res Treat. 2019. [Epub ahead of print]. https://doi.org/10.4143/crt.2019.210 [PubMed]
- 11. Tiwari AK, Roy HK, Lynch HT. Lynch syndrome in the 21st century: clinical perspectives. QJM. 2016; 109:151–58. https://doi.org/10.1093/qjmed/hcv137 [PubMed]
- 12. Marcheselli R, Marcheselli L, Cortesi L, Bari A, Cirilli C, Pozzi S, Ferri P, Napolitano M, Federico M, Sacchi S. Risk of second primary malignancy in breast cancer survivors: a nested population-based case-control study. J Breast Cancer. 2015; 18:378–85. https://doi.org/10.4048/jbc.2015.18.4.378 [PubMed]
- 13. Berrington de Gonzalez A, Curtis RE, Kry SF, Gilbert E, Lamart S, Berg CD, Stovall M, Ron E. Proportion of second cancers attributable to radiotherapy treatment in adults: a cohort study in the US SEER cancer registries. Lancet Oncol. 2011; 12:353–60. https://doi.org/10.1016/S1470-2045(11)70061-4 [PubMed]
- 14. Do KA, Johnson MM, Doherty DA, Lee JJ, Wu XF, Dong Q, Hong WK, Khuri FR, Fu KK, Spitz MR. Second primary tumors in patients with upper aerodigestive tract cancers: joint effects of smoking and alcohol (United States). Cancer Causes Control. 2003; 14:131–38. https://doi.org/10.1023/A:1023060315781 [PubMed]
- 15. van Leeuwen FE, Klokman WJ, Stovall M, Hagenbeek A, van den Belt-Dusebout AW, Noyon R, Boice JD
Jr , Burgers JM, Somers R. Roles of radiotherapy and smoking in lung cancer following Hodgkin’s disease. J Natl Cancer Inst. 1995; 87:1530–37. https://doi.org/10.1093/jnci/87.20.1530 [PubMed] - 16. Tabuchi T, Ito Y, Ioka A, Nakayama T, Miyashiro I, Tsukuma H. Tobacco smoking and the risk of subsequent primary cancer among cancer survivors: a retrospective cohort study. Ann Oncol. 2013; 24:2699–704. https://doi.org/10.1093/annonc/mdt279 [PubMed]
- 17. Druesne-Pecollo N, Keita Y, Touvier M, Chan DS, Norat T, Hercberg S, Latino-Martel P. Alcohol drinking and second primary cancer risk in patients with upper aerodigestive tract cancers: a systematic review and meta-analysis of observational studies. Cancer Epidemiol Biomarkers Prev. 2014; 23:324–31. https://doi.org/10.1158/1055-9965.EPI-13-0779 [PubMed]
- 18. Knight JA, Bernstein L, Largent J, Capanu M, Begg CB, Mellemkjaer L, Lynch CF, Malone KE, Reiner AS, Liang X, Haile RW, Boice JD
Jr , Bernstein JL, and WECARE Study Collaborative Group. Alcohol intake and cigarette smoking and risk of a contralateral breast cancer: The Women’s Environmental Cancer and Radiation Epidemiology Study. Am J Epidemiol. 2009; 169:962–68. https://doi.org/10.1093/aje/kwn422 [PubMed] - 19. Travis LB, Fosså SD, Schonfeld SJ, McMaster ML, Lynch CF, Storm H, Hall P, Holowaty E, Andersen A, Pukkala E, Andersson M, Kaijser M, Gospodarowicz M, et al. Second cancers among 40,576 testicular cancer patients: focus on long-term survivors. J Natl Cancer Inst. 2005; 97:1354–65. https://doi.org/10.1093/jnci/dji278 [PubMed]
- 20. Lu CH, Lee KD, Chen PT, Chen CC, Kuan FC, Huang CE, Chen MF, Chen MC. Second primary malignancies following thyroid cancer: a population-based study in Taiwan. Eur J Endocrinol. 2013; 169:577–85. https://doi.org/10.1530/EJE-13-0309 [PubMed]
- 21. Montero-Miranda PH, Ganly I. Survivorship—competing mortalities, morbidities, and second malignancies. Otolaryngol Clin North Am. 2013; 46:681–710. https://doi.org/10.1016/j.otc.2013.04.008 [PubMed]
- 22. Laccetti AL, Pruitt SL, Xuan L, Halm EA, Gerber DE. Effect of prior cancer on outcomes in advanced lung cancer: implications for clinical trial eligibility and accrual. J Natl Cancer Inst. 2015; 107:djv002. https://doi.org/10.1093/jnci/djv002 [PubMed]
- 23. Shukla A, Shukla S, Osowo A, Mashtare T, Bhutani MS, Guha S. Risk of colorectal adenomas in women with prior breast cancer. Dig Dis Sci. 2012; 57:3240–45. https://doi.org/10.1007/s10620-012-2432-9 [PubMed]
- 24. Garg R, Zahurak ML, Trimble EL, Armstrong DK, Bristow RE. Abdominal carcinomatosis in women with a history of breast cancer. Gynecol Oncol. 2005; 99:65–70. https://doi.org/10.1016/j.ygyno.2005.05.013 [PubMed]
- 25. Pierce SR, Stine JE, Gehrig PA, Havrilesky LJ, Secord AA, Nakayama J, Snavely AC, Moore DT, Kim KH. Prior breast cancer and tamoxifen exposure does not influence outcomes in women with uterine papillary serous carcinoma. Gynecol Oncol. 2017; 144:531–35. https://doi.org/10.1016/j.ygyno.2016.12.024 [PubMed]
- 26. Jung SY, Kim YA, Jo M, Park SM, Won YJ, Ghang H, Kong SY, Jung KW, Lee ES. Prediagnosis obesity and secondary primary cancer risk in female cancer survivors: A national cohort study. Cancer Med. 2019; 8:824–38. https://doi.org/10.1002/cam4.1959 [PubMed]
- 27. Smyth EC, Tarazona N, Peckitt C, Armstrong E, Mansukhani S, Cunningham D, Chau I. Exclusion of Gastrointestinal Cancer Patients With Prior Cancer From Clinical Trials: Is This Justified? Clin Colorectal Cancer. 2016; 15:e53–59. https://doi.org/10.1016/j.clcc.2015.11.003 [PubMed]
- 28. Park SM, Yun YH, Kim YA, Jo M, Won YJ, Back JH, Lee ES. Prediagnosis Body Mass Index and Risk of Secondary Primary Cancer in Male Cancer Survivors: A Large Cohort Study. J Clin Oncol. 2016; 34:4116–24. https://doi.org/10.1200/JCO.2016.66.4920 [PubMed]
- 29. Corkum M, Hayden JA, Kephart G, Urquhart R, Schlievert C, Porter G. Screening for new primary cancers in cancer survivors compared to non-cancer controls: a systematic review and meta-analysis. J Cancer Surviv. 2013; 7:455–63. https://doi.org/10.1007/s11764-013-0278-6 [PubMed]
- 30. Domchek SM, Jhaveri K, Patil S, Stopfer JE, Hudis C, Powers J, Stadler Z, Goldstein L, Kauff N, Khasraw M, Offit K, Nathanson KL, Robson M. Risk of metachronous breast cancer after BRCA mutation-associated ovarian cancer. Cancer. 2013; 119:1344–48. https://doi.org/10.1002/cncr.27842 [PubMed]
- 31. Travis LB, Demark Wahnefried W, Allan JM, Wood ME, Ng AK. Aetiology, genetics and prevention of secondary neoplasms in adult cancer survivors. Nat Rev Clin Oncol. 2013; 10:289–301. https://doi.org/10.1038/nrclinonc.2013.41 [PubMed]
- 32. Zhou H, Huang Y, Qiu Z, Zhao H, Fang W, Yang Y, Zhao Y, Hou X, Ma Y, Hong S, Zhou T, Zhang Y, Zhang L. Impact of prior cancer history on the overall survival of patients newly diagnosed with cancer: A pan-cancer analysis of the SEER database. Int J Cancer. 2018; 143:1569–77. https://doi.org/10.1002/ijc.31543 [PubMed]
- 33. Li Z, Wang K, Shi Y, Zhang X, Wen J. Incidence of second primary malignancy after breast cancer and related risk factors-Is breast-conserving surgery safe? A nested case-control study. Int J Cancer. 2020; 146:352–62. https://doi.org/10.1002/ijc.32259 [PubMed]
- 34. Molina-Montes E, Requena M, Sánchez-Cantalejo E, Fernández MF, Arroyo-Morales M, Espín J, Arrebola JP, Sánchez MJ. Risk of second cancers cancer after a first primary breast cancer: a systematic review and meta-analysis. Gynecol Oncol. 2015; 136:158–71. https://doi.org/10.1016/j.ygyno.2014.10.029 [PubMed]
- 35. Zhang Z. Propensity score method: a non-parametric technique to reduce model dependence. Ann Transl Med. 2017; 5:7. https://doi.org/10.21037/atm.2016.08.57 [PubMed]
- 36. Austin PC. An Introduction to Propensity Score Methods for Reducing the Effects of Confounding in Observational Studies. Multivariate Behav Res. 2011; 46:399–424. https://doi.org/10.1080/00273171.2011.568786 [PubMed]
- 37. Goldhirsch A, Wood WC, Gelber RD, Coates AS, Thürlimann B, Senn HJ, and 10th St. Gallen conference. Progress and promise: highlights of the international expert consensus on the primary therapy of early breast cancer 2007. Ann Oncol. 2007; 18:1133–44. https://doi.org/10.1093/annonc/mdm271 [PubMed]