Introduction
Non-small cell lung cancer (NSCLC) represents approximately 85% to 90% of lung cancer cases and is the leading cause of cancer-related death worldwide, with a lower than 15% 5-year survival [1, 2]. Since treatment selections have become increasingly related to the biological subtypes of NSCLC, attention has been drawn to tumors harboring epidermal growth factor receptor (EGFR) mutations, which are estimated to exist in 10%-15% of patients with nonsquamous NSCLC [3]. The identification of EGFR mutations has led to the development of targeted therapies, including small molecule tyrosine kinase inhibitors (TKIs) directed at the signal transduction pathway as well as immunotherapies incorporating checkpoint monoclonal antibodies that bind to and inactivate the receptors on cell membranes [4].
As a monotherapy, gefitinib, erlotinib and, more recently, afatinib have been licensed and recommended as first-line treatment regimens for EGFR-mutant NSCLC patients by the European Society for Medical Oncology (ESMO) guidelines. In August 2015, the American Society of Clinical Oncology (ASCO) clinical guidelines recommended two cytotoxic drugs, docetaxel and pemetrexed, and two EGFR-TKIs, erlotinib and gefitinib, to patients who experienced treatment failure with conventional first-line chemotherapy [4]. Nevertheless, several new regimens have been approved by the US FDA, such as the combination of docetaxel and ramucirumab, nivolumab, pembrolizumab, and atezolizumab. At the same time, more than 40 therapeutic options are being assessed in randomized controlled trials (RCTs) [5]. With more clinical trials emerging [4–46], the FLAURA trial [42] has shown that osimertinib has superior efficacy compared with standard EGFR-TKIs in treating advanced EGFR-mutant NSCLC with less serious adverse effects (18.9 months vs 10.2 months for progression-free survival (PFS), P<0.001). The newest National Comprehensive Cancer Network (NCCN) guidelines also regarded osimertinib as category 1 for advanced EGFR-mutant NSCLC.
There is an urgent need to identify complete information on the most effective and latest treatment for advanced EGFR-mutant NSCLC. Conventional meta-analyses have only partially captured the available evidence for treating the intended populations; their outcomes are not comprehensive. This work is a generalized version of a pairwise meta-analysis integrating direct and indirect evidence [4–46] to aid in clinical decision making. Thus, the aim of this article is to comprehensively evaluate the effectiveness and safety of various therapeutics for advanced EGFR-mutant NSCLC.
Results
Study selection and characteristics of the included studies
We identified a total of 1749 records from a database search and 34 records from other available literature; of these, 1721 were excluded based on the selection criteria. Subsequently, 62 potential articles went through full-text review, and 41 studies were ultimately eligible for inclusion (Appendix Figure 1 in the Supplementary Data).
Forty-one RCTs [6–46] consisting of 8430 total participants were included in the analysis. The characteristics and results of the studies are detailed in Appendix Table 2 in the Supplementary Data. The included RCTs encompassed 22 unique treatments and 15 medication classes; there were 39 direct comparisons for PFS (38 trials [6–24, 26–28, 30–40, 42–46]; n= 7670) and 23 direct comparisons for OS (22 trials [6, 7, 9, 11–13, 15, 19–22, 25, 27, 29, 35, 36, 38–42, 45]; n= 3842). Data were extracted from survival plots in 5 studies [9, 11, 18, 22, 35]. Most treatments in eligible trials were first-line setting, the abbreviations for the medication classes and their constituent individual treatments are listed in Table 1. The mean age of the participants ranged from 56.0 to 74.0 years, with a median age of 61.9 years. The follow-up period ranged from 10 to 70 months with a median duration of 27.5 months. Ten studies [14, 26, 31, 34, 35, 38, 39, 42, 43, 46] reported data on metastases, 9 [14, 26, 34, 35, 38, 39, 42, 43, 46] reported data on central nervous system (CNS) metastases, and 5 [14, 26, 31, 34, 42] reported data on visceral, bone or other metastases. Of the 41 total studies, 40 were two-arm trials, 1 [45] was a three-arm trial, 34 studies were phase III clinical trials, and 7 [6, 7, 18, 22, 27, 30, 31] were phase II trials. Of note, 16 studies [10, 14, 22, 23, 28–32, 34, 35, 38, 39, 41, 42, 46] provided EGFR genomic aberration data (19 del and/or 21 L858R), 14 [10, 14, 22, 23, 28, 30–32, 34, 35, 38, 39, 42, 46] for PFS and 2 [29, 41] for OS. There were 12 trials involving Asian patients and 29 trials involving 6408 non-Asian patients (multiple nations or no Asian region).
Table 1. List of medication classes and individual treatments.
| Medication class level | Treatment level | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 1st-gen ET: first generation EGFR-TKI | Gef: gefitinib | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Erlo: erlotinib | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Ico: icotinib | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 2nd-gen ET: second generation EGFR-TKI | Afa: afatinib | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Dac: dacomitinib | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 3rd-gen ET: third generation EGFR-TKI | Osi: osimertinib | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Naq: naquotinib | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ET+aVEGFR: EGFR-TKI+anti-VEGFR | Erlo+Bev: erlotinib+bevacizumab | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sun+Erlo: sunitinib+erlotinib | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| MT+ET: MET-TKI+EGFR-TKI | Ona+Erlo: onartuzumab+erlotinib | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Erlo+Tiv: erlotinib+tivantinib | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ET+CT: EGFR-TKI+cytotoxic therapy | Gef+Peme: gefitinib+pemetrexed | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ET+aVEGFR+Plat: EGFR-TKI+anti-VEGFR + platinum-based therapy | Cil+Cet+Plat: cilengitide+cetuximab+platinum-based therapy | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cet+Bev+Plat: cetuximab+bevacizumab+platinum-based therapy | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ET+Plat: EGFR-TKI+platinum-based therapy | Cet+Plat: cetuximab+platinum-based therapy | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Erlo+Plat: erlotinib+platinum-based therapy | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| aVEGFR+Plat: anti-VEGFR+platinum-based therapy | Mot+Plat: motesanib+platinum-based therapy | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Plat* | Plat: platinum-based therapy | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CT: cytotoxic therapy | Doc: docetaxel | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Vin: vinorelbine | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| WBRT | WBRT: whole-brain radiotherapy | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Placebo | Placebo | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| *In this study, platinum-based therapy contains: pemetrexed + cisplatin/carboplatin, gemcitabine + cisplatin/carboplatin, vinorelbine + cisplatin/carboplatin, paclitaxel + cisplatin/carboplatin, docetaxel + cisplatin/carboplatin, docetaxel + gemcitabine + cisplatin/carboplatin, pemetrexed + gemcitabine+cisplatin/carboplatin. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Risk of bias and quality assessment
In the quality assessment, we found that a vast majority of the included studies had a low risk of bias (Appendix Table 3 in the Supplementary Data). Two [24, 46] studies had “other bias” according to the Cochrane risk of bias tool, and 16 [10, 13, 15, 19, 20, 22, 26–29, 31, 35, 38, 39, 41, 44] had an unclear “other bias”. Most of the risk stemmed from the blinding of participants and personnel and blinding of outcome assessment categories due to the open-label method.
Bayesian NMA at the individual-treatment level
A total of 37 RCTs with 37 arms and 21 unique treatment levels were included in the treatment level analysis for PFS; a study by Reck et al. [45] was omitted for not contributing to a complete loop (Figure 1A). Twenty-one unique nodes were presented in this analysis, with every node representing a unique intervention. The nodes with the most direct interactions were between Plat and Erlo, Plat and Gef, and Osi and Plat (Figure 1B). Erlo+Bev (HR 0.26, 95% CrI: 0.08-0.75), Osi (HR 0.29, 95% CrI: 0.11-0.70), Afa (HR 0.37, 95% CrI: 0.17-0.78), and Erlo (HR 0.46, 95% CrI: 0.21-0.93) showed significant superiority over placebo in terms of PFS. Moreover, the probability for Erlo+Bev ranked first, followed by Osi, Afa, and Erlo; the ranking probabilities were 0.164, 0.164, 0.120, and 0.128, respectively (Figure 1C, 1D). The results of the comparisons among those treatments and all treatments are shown in Figure 1C and Appendix Table 4 in the Supplementary Data. The model fit was good, and there was no significant heterogeneity or loop inconsistency (Table 2).
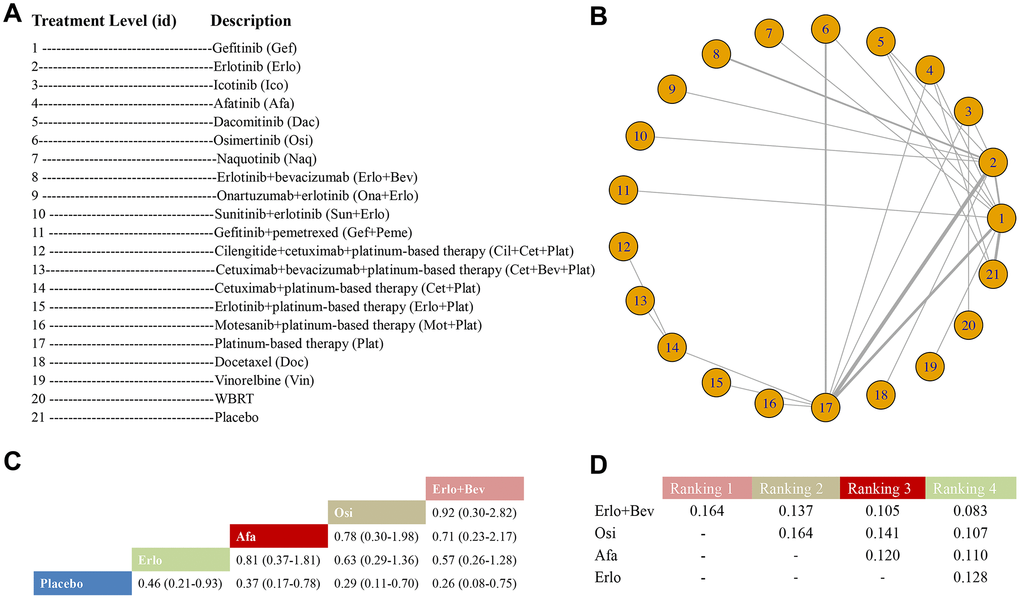
Figure 1. Meta-analysis of efficacy for PFS at the individual-treatment level. (A) Descriptions of the individual treatments included in this analysis. (B) Network plots showing comparisons between nodes (yellow circles), each representing a unique individual treatment. Each line corresponds to direct comparisons between treatments with the width corresponding to the number of direct within-trial comparisons. (C) Comparison results of the most efficacious treatments and placebo (HR (95% CrI)). Each result is a comparison between the column-defining drug and the row-defining treatment. (D) Schematic detailing the most efficacious treatments according to the rankograms.
Table 2. Edge-splitting method for direct and indirect evidence relating to PFS, OS, ORR and grade 3 or higher AEs in treatment-level.
| Multiple-treatment | Direct comparison outcome | Indirect comparison outcome | Combined outcome | I2 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PFS (HR and 95% CrI) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Erlo vs Gef | 0.94 (0.42-2.10) | 0.84 (0.41-1.70) | 0.88 (0.53-1.50) | < 50% | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Afa vs Gef | 0.73 (0.23-2.30) | 0.71 (0.28-1.80) | 0.72 (0.36-1.50) | < 50% | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Dac vs Gef | 0.62 (0.20-1.90) | 0.97 (0.38-2.60) | 0.79 (0.40-1.60) | < 50% | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Osi vs Gef | 0.46 (0.15-1.40) | 0.65 (0.23-1.80) | 0.55 (0.27-1.10) | < 50% | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Plat vs Gef | 1.90 (0.94-3.80) | 2.10 (1.10-4.20) | 2.0 (1.30-3.20) | < 50% | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Placebo vs Gef | 2.40 (1.20-5.30) | 1.40 (0.48-3.80) | 1.90 (1.10-3.60) | < 50% | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Ico vs Erlo | 0.78 (0.24-2.50) | 1.60 (0.47-5.30) | 1.10 (0.47-2.50) | < 50% | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Dac vs Erlo | 1.00 (0.30-3.60) | 0.83 (0.30-2.40) | 0.90 (0.43-2.00) | < 50% | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Plat vs Erlo | 2.50 (1.40-4.50) | 2.00 (0.88-4.30) | 2.30 (1.40-3.60) | < 50% | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Plat vs Ico | 1.50 (0.51-4.70) | 3.10 (0.86-11.00) | 2.10 (0.91-4.80) | < 50% | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Plat vs Afa | 3.90 (1.20-12.00) | 2.20 (0.79-5.90) | 2.80 (1.30-5.80) | < 50% | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Plat vs Osi | 3.20 (1.40-7.70) | 4.60 (1.30-16.00) | 3.70 (1.80-7.20) | < 50% | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Placebo vs Afa | 2.00 (0.58-6.70) | 3.50 (1.30-11.00) | 2.70 (1.30-6.00) | < 50% | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Placebo vs Dac | 2.1 (0.65-6.70) | 2.80 (0.96-8.80) | 2.40 (1.10-5.30) | < 50% | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| OS (HR and 95% CrI) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Plat vs Gef | 1.30 (0.25-7.00) | 0.93 (0.32-2.60) | 1.00 (0.44-2.60) | < 50% | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Vin vs Gef | 0.35 (0.06-2.10) | NA | 0.35 (0.09-1.40) | NA | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Placebo vs Gef | 2.30 (0.36-14.00) | 0.91 (0.26-3.20) | 1.20 (0.51-4.00) | < 50% | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Erlo vs Gef | 0.84 (0.17-4.60) | 1.10 (0.43-2.70) | 1.00 (0.48-2.40) | < 50% | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Dac vs Gef | 0.77 (0.15-4.10) | 0.99 (0.37-2.60) | 0.92 (0.44-2.40) | < 50% | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Osi vs Gef | 0.63 (0.12-3.30) | NA | 0.63 (0.20-2.00) | NA | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Erlo vs Sun + Erlo | 0.72 (0.13-4.00) | NA | 0.72 (0.21-2.50) | NA | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cet + Plat vs Cil + Cet +Plat | 1.00 (0.19-5.70) | NA | 1.10 (0.32-3.50) | NA | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cet + Plat vs Cet + Bev +Plat | 1.20 (0.24-6.30) | NA | 1.20 (0.39-3.80) | NA | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Plat vs Cet + Plat | 1.40 (0.26-7.40) | NA | 1.40 (0.44-4.30) | NA | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Erlo vs Plat | 1.20 (0.33-4.10) | 0.88 (0.32-2.50) | 0.99 (0.46-2.20) | < 50% | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Ico vs Plat | 1.00 (0.20-5.30) | NA | 1.00 (0.32-3.30) | NA | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Erlo vs Doc | 0.38 (0.05-2.70) | NA | 0.38 (0.08-1.90) | NA | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Ico vs WBRT | 0.93 (0.17-4.90) | NA | 0.94 (0.29-3.10) | NA | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Afa vs Placebo | 1.60 (0.30-9.20) | NA | 1.60 (0.47-5.80) | NA | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Dac vs Placebo | 0.98 (0.19-5.20) | 0.68 (0.20-2.30) | 0.77 (0.27-1.90) | < 50% | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Dac vs Erlo | 0.94 (0.16-5.80) | 0.90 (0.30-2.70) | 0.91 (0.37-2.40) | < 50% | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Erlo + Bev vs Erlo | 1.20 (0.19-7.20) | NA | 1.2 (0.28-4.80) | NA | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Ona +Erlo vs Erlo | 4.60 (0.54-41.00) | NA | 4.70 (0.71-30.00) | NA | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Erlo + Tiv vs Erlo | 0.72 (0.35-1.50) | NA | 0.72 (0.20-2.60) | NA | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ORR (OR and 95% CrI) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Plat vs Gef | 0.16 (0.03-0.72) | 0.22 (0.06-0.78) | 0.19 (0.07-0.49) | < 50% | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Afa vs Gef | 1.78 (0.39-8.07) | 1.33 (0.39-4.54) | 1.49 (0.56-3.78) | < 50% | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Dac vs Gef | 1.17 (0.26-5.23) | NA | 1.19 (0.37-3.70) | NA | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Gef + Peme vs Gef | 1.16 (0.33-3.51) | NA | 1.16 (0.42-3.00) | NA | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Afa vs Plat | 6.38 (1.43-29.91) | 8.84 (2.60-30.11) | 7.78 (3.02-20.37) | < 50% | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Osi vs Plat | 2.81 (0.89-7.70) | NA | 2.96 (1.16-6.36) | NA | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cet + Plat vs Plat | 2.08 (0.45-9.29) | NA | 2.03 (0.65-6.60) | NA | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Osi vs Erlo | 1.26 (0.27-5.57) | NA | 1.26 (0.41-3.98) | NA | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Naq vs Erlo | 0.53 (0.12-2.39) | NA | 0,53 (0.17-1.67) | NA | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Erlo + Bev vs Erlo | 1.30 (0.44-3.90) | NA | 1.32 (0.55-3.11) | NA | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Grade 3 or higher AEs (OR and 95% CrI) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Plat vs Gef | 0.16 (0.03-0.72) | 0.22 (0.06-0.78) | 0.19 (0.07-0.49) | < 50% | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Afa vs Gef | 1.78 (0.39-8.07) | 1.33 (0.39-4.54) | 1.49 (0.56-3.78) | < 50% | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Dac vs Gef | 1.17 (0.26-5.23) | NA | 1.19 (0.37-3.70) | NA | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Gef + Peme vs Gef | 1.16 (0.33-3.51) | NA | 1.16 (0.42-3.00) | NA | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Afa vs Plat | 6.38 (1.43-29.91) | 8.84 (2.60-30.11) | 7.78 (3.02-20.37) | < 50% | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Osi vs Plat | 2.81 (0.89-7.70) | NA | 2.96 (1.16-6.36) | NA | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cet + Plat vs Plat | 2.08 (0.45-9.29) | NA | 2.03 (0.64-6.60) | NA | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Osi vs Erlo | 1.26 (0.27-5.57) | NA | 1.26 (0.41-3.98) | NA | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Naq vs Erlo | 0.53 (0.12-2.39) | NA | 0.53 (0.17-1.67) | NA | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Erlo + Bev vs Erlo | 1.30 (0.44-3.90) | NA | 1.32 (0.55-3.11) | NA | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Abbreviations: PFS, progression-free survival; OS, overall survival; ORR, objective response rate; AEs, adverse events; NA, not available. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Abbreviations of available treatments could be found in the main body of manuscript. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
A total of 21 RCTs with 21 arms and 18 unique treatment levels were considered in the OS analysis; a study by Reck et al. [45] was excluded from the network (Figure 2A). Eighteen nodes were included in the treatment-level analysis for OS. The most direct interactions were in nodes between Erlo and Plat (Figure 2B). Osi (HR 0.52, 95% CrI: 0.10-2.00), Cet+Bev+Plat (HR 0.51, 95% CrI: 0.06-3.38), Cil+Cet+Plat (HR 0.59, 95% CrI: 0.06-4.30), and Cet+Plat (HR 0.63, 95% CrI: 0.10-3.03) appeared to benefit OS over placebo even though the favorable efficacy was nonsignificant. Osi (ranking probability: 0.183) ranked first, followed by Cet+Bev+Plat (0.157), Cil+Cet+Plat (0.117), and Cet+Plat (0.147) (Figure 2C, 2D). The results of the comparisons among those treatments and all treatments are shown in Figure 2C and Appendix Table 4 in the Supplementary Data. The statistical model was good, and no evidence of heterogeneity or loop inconsistency was observed (Table 2). A total of 13 trials with 10 unique treatment levels were analyzed for ORR after excluding Reck et al. [45] and Yang et al. (2) [38] (Appendix Figure 2 in Supplementary Data). The primary data are shown in Appendix Table 5 in the Supplementary Data. Afa ranked first and had the best ORR (OR 7.67, 95% CrI: 2.93-20.68 vs Plat), followed by Dac (OR 6.10, 95% CrI: 1.34-29.20), Gef (OR 5.14, 95% CrI: 1.96-14.10), and Gef + Peme (OR 6.02, 95% CrI: 1.42-23.13) (Appendix Table 6 in the Supplementary Data). No evidence of heterogeneity was found. A total of 17 trials with 13 unique treatment levels focused on grade 3 or higher AEs (Appendix Figure 3 in the Supplementary Data). The primary data are shown in Appendix Table 7 in the Supplementary Data. Erlo + Bev had the most grade 3 or higher AEs (OR 24.22, 95% CrI: 0.64-NA vs Ico), followed by Gef +Peme (OR 15.28, 95% CrI: 0.43-NA), Plat (OR 12.61, 95% CrI: 0.86-NA) and Dac (OR 11.09, 95% CrI: 0.33-NA) (Appendix Table 8 in the Supplementary Data). No significant heterogeneity was noted (Table 2). The outcome of DoR was not analyzed for insufficient comparisons.
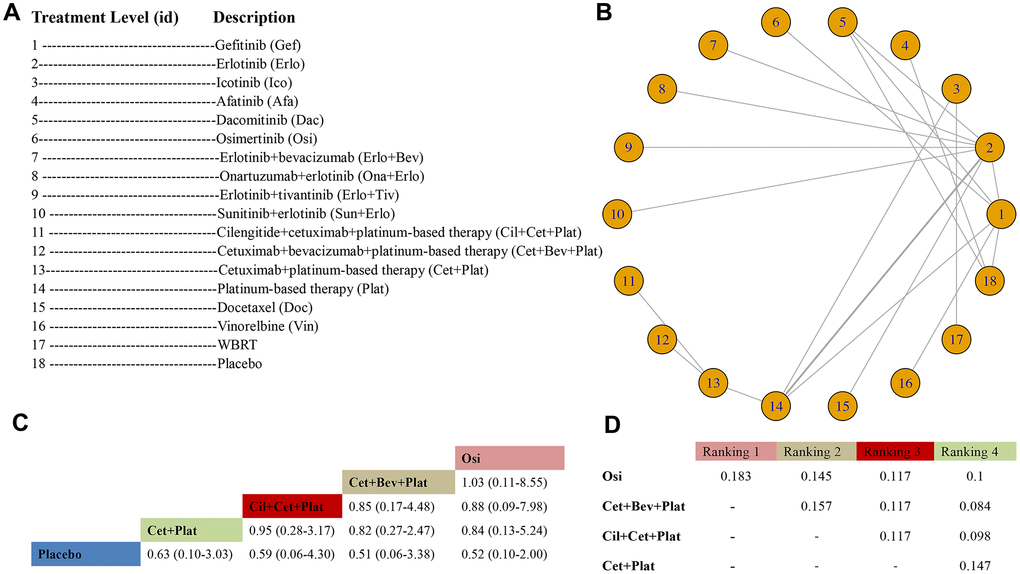
Figure 2. Meta-analysis of efficacy for OS at the individual-treatment level. (A) Descriptions of the individual treatments included in this analysis. (B) Network plots showing comparisons between nodes (yellow circles), each representing a unique individual treatment. Each line corresponds to direct comparisons between treatments, with the width corresponding to the number of direct within-trial comparisons. (C) Comparison results of the most efficacious treatments and placebo (HR (95% CrI)). Each result is a comparison between the column-defining drug and the row-defining treatment. (D) Schematic detailing the most efficacious treatments according to the rankograms.
Bayesian NMA at the medication-class level
There were 35 RCTs with 36 arms and 15 unique class levels included in the class-level analysis for PFS; the studies of Yang et al. [39], Urata et al. [32], and Shi et al. [17] were excluded for having two of the same class interventions. A total of 15 nodes were presented, in which the most direct interventions were between 1st-gen ET and Plat, 1st-gen ET and 2nd-gen ET, and 1st-gen ET and Placebo (Figure 3A and Figure 3B). ET+aVEGFR (HR 0.35, 95% CrI: 0.14-0.85) and 3rd-gen ET (HR 0.39, 95% CrI: 0.16-0.91) were better than placebo, while ET+aVEGFR+Plat (HR 0.40, 95% CrI: 0.11-1.37) and IT+aVEGFR+Plat (HR 0.42, 95% CrI: 0.06-2.63) seemed to be superior than placebo, but the data were not statistically significant (Figure 3C). Our work demonstrated that statistically, ET+aVEGFR ranked first, followed by 3rd-gen ET, ET+aVEGFR+Plat, and IT+aVEGFR+Plat (Figure 3D). The results of the comparisons among those treatments and all treatments are shown in Figure 3C and Appendix Table 9 in the Supplementary Data. The model was good, and no heterogeneity or inconsistency was observed.
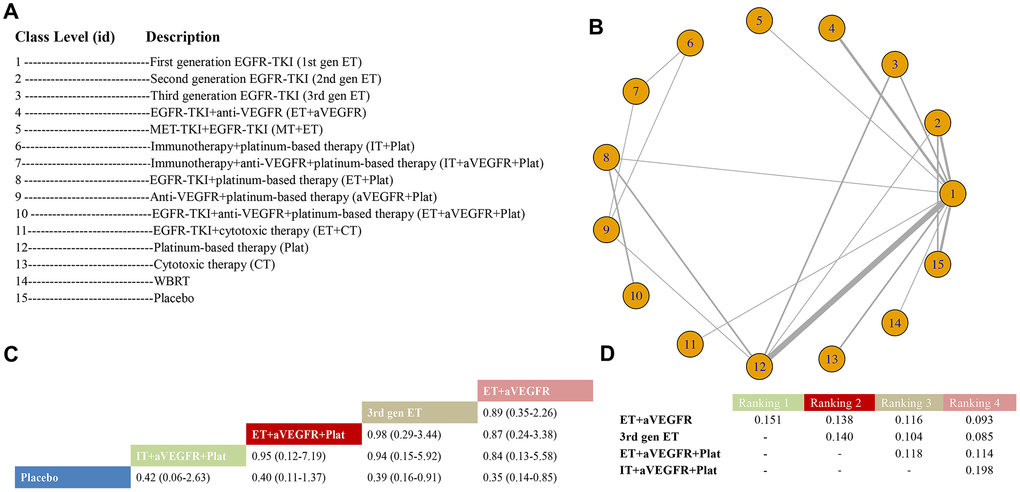
Figure 3. Meta-analysis of efficacy for PFS at the medication-class level. (A) Descriptions of the medication classes included in this analysis. (B) Network plots showing comparisons between nodes (yellow circles), each representing a unique medication class. Each line corresponds to direct comparisons between treatments, with the width corresponding to the number of direct within-trial comparisons. (C) Comparison results of the most efficacious class and placebo (HR (95% CrI)). Each result is a comparison between the column-defining drug and the row-defining class. (D) Schematic detailing the most efficacious medication classes according to the rankograms.
In total, 21 RCTs with 11 arms and 11 unique class levels were selected for the class-level analysis of OS; studies by Yang et al. [39] and Reck et al. [45] were excluded for comparing the same class interventions (Figure 4A). The 11 nodes with the most direct interactions were between 1st-gen ET and Plat (Figure 4B). When compared with placebo, the HR for ET+aVEGFR+Plat was 0.65 (95% CrI: 0.13-2.71), the HR for ET+Plat was 0.76 (95% CrI: 0.17-2.65), and the HR for 3rd-gen ET was 0.66 (95% CrI: 0.16-2.04) (Figure 4C). Bayesian analysis revealed that ET+aVEGFR+Plat was significantly ranked first, followed by ET+Plat, and then 3rd-gen ET (Figure 4D). Additional data on the interactions can be found in Figure 4C and Appendix Table 9 in the Supplementary Data. The fitness for this model was good, and no significant heterogeneity or inconsistencies were observed.
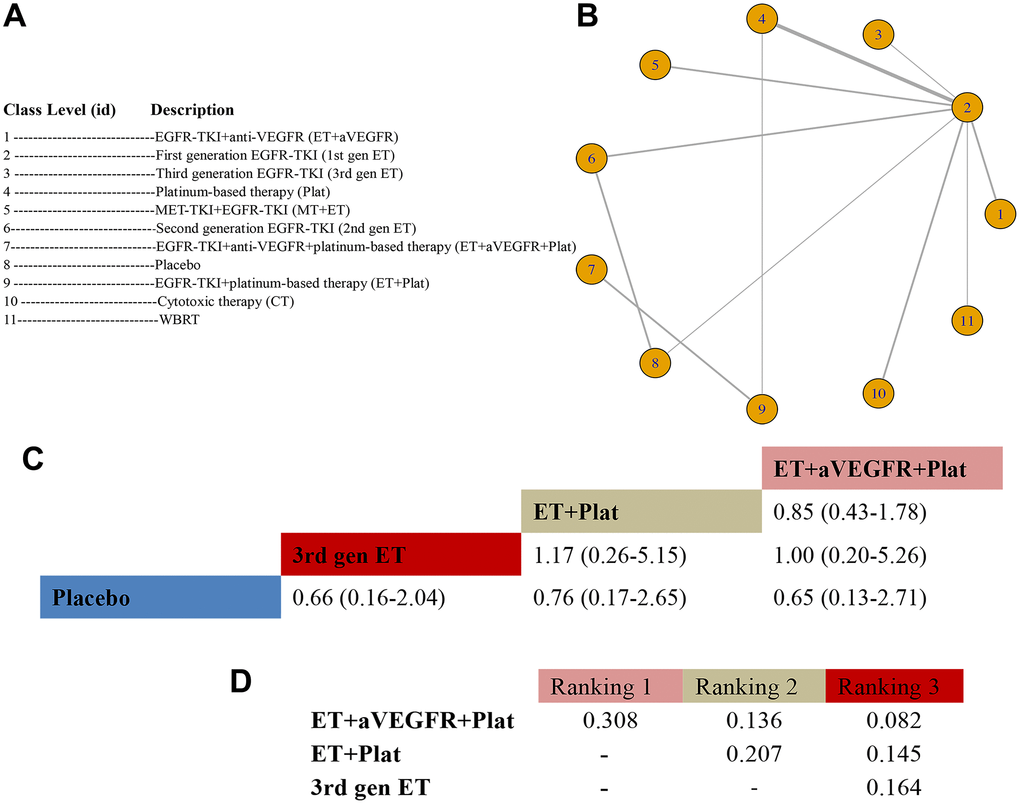
Figure 4. Meta-analysis of efficacy for OS at the medication-class level. (A) Descriptions of the medication classes included in this analysis. (B) Network plots showing comparisons between nodes (yellow circles), each representing a unique medication class. Each line corresponds to direct comparisons between treatments, with the width corresponding to the number of direct within-trial comparisons. (C) Comparison results of the most efficacious class and placebo (HR (95% CrI)). Each result is a comparison between the column-defining drug and the row-defining class. (D) Schematic detailing the most efficacious medication classes according to the rankograms.
In medication-class level analysis for ORR (Appendix Figure 4 in the Supplementary Data, Appendix Table 5 in the Supplementary Data), 2nd-gen ET was the best (OR 12.52, 95% CrI: 3.38-49.41 vs WBRT), followed by ET + aVEGFR (OR 11.64, 95% CrI: 2.87-49.19) and 1st-gen ET (OR 8.82, 95% CrI: 2.79-29.56) (Appendix Table 10 in the Supplementary Data). Analysis for DoR (Appendix Figure 5 in the Supplementary Data, Appendix Table 11 in the Supplementary Data) revealed that 2nd-gen ET had the longest DoR (MD 5.08, 95% CrI: -11.44 to 21.63 vs ET + aVEGFR), followed by 3rd-gen ET (MD 3.04, 95% CrI: -11.29 to 17.31) and ET + CT (MD 3.87, 95% CrI: -12.47 to 20.38) (Appendix Table 12 in the Supplementary Data). Analysis for grade 3 or higher AEs (Appendix Figure 6 in the Supplementary Data, Appendix Table 7 in the Supplementary Data) revealed that ET + aVEGFR had the most grade 3 or higher AEs (OR 9.01, 95% CrI: 0.99-92.27 vs Placebo), followed by Plat (OR 5.25, 95% CrI: 0.98-29.83) and ET + CT (OR 3.82, 95% CrI: 0.39-37.26) (Appendix Table 13 in the Supplementary Data). There was low to moderate heterogeneity among medication-level comparisons on ORR, DoR and grade 3 or higher AEs.
EGFR genomic status
Multiple treatment-level comparisons (10 treatments) were specifically performed for 19 del and 21 L858R mutations regarding PFS. We found that Erlo+Bev, Osi, Afa, and Erlo were top-ranking alternatives for patients harboring 19 del mutations to prolong PFS; additionally, Erlo+Bev, Osi, Gef+Pem, and Dac were optimal treatments among the available treatments for patients with 21 L858R mutations (Table 3). Overall, Erlo+Bev, Osi, and 2nd-gen ET (including Afa and Dac) were the most viable treatment options, and no significant differences were found in terms of the best strategies for 19 del and 21 L858R mutations. The evidence was insufficient to assess these mutations in terms of OS as well as uncommon mutations.
Table 3. Comparisons of PFS for exon 19 deletion and exon 21 Leu-858Arg mutation according to treatment-level Bayesian analysis.
| Gef | 0.76 (0.26-2.20) | 1.63 (0.27-10.40) | 0.56 (0.13-2.23) | 0.70 (0.22-2.23) | 0.68 (0.22-2.15) | 0.41 (0.09-1.80) | 0.67 (0.16-2.95) | 3.19 (1.10-9.82) | 4.18 (0.41-44.40) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 1.31 (0.45-3.89) | Erlo | 2.15 (0.44-11.80) | 0.73 (0.13-4.30) | 0.92 (0.26-3.40) | 0.90 (0.26-3.40) | 0.54 (0.19-1.54) | 0.88 (0.15-5.69) | 4.21 (1.99-9.59) | 5.49 (0.61-53.60) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 0.61 (0.10-3.67) | 0.47 (0.08-2.26) | Ico | 0.34 (0.03-3.25) | 0.43 (0.07-2.63) | 0.42 (0.06-2.59) | 0.25 (0.03-1.66) | 0.41 (0.04-4.07) | 1.96 (0.46-8.03) | 2.57 (0.56-11.70) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 1.80 (0.45-7.48) | 1.37 (0.23-7.84) | 2.95 (0.31-30.60) | Dac | 1.27 (0.21-7.83) | 1.23 (0.21-7.75) | 0.74 (0.09-5.51) | 1.21 (0.16-9.75) | 5.76 (1.91-34.90) | 7.53 (0.51-125.00) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 1.43 (0.45-4.46) | 1.09 (0.29-3.86) | 2.34 (0.38-15.40) | 0.79 (0.13-4.88) | Afa | 0.97 (0.23-4.14) | 0.58 (0.11-2.97) | 0.95 (0.15-6.19) | 4.57 (1.46-15.10) | 5.99 (0.57-64.80) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 1.47 (0.47-4.50) | 1.12 (0.31-3.86) | 2.41 (0.39-15.90) | 0.81 (0.13-4.85) | 1.03 (0.24-4.31) | Osi | 0.60 (0.11-3.05) | 0.99 (0.15-6.34) | 4.69 (1.50-14.70) | 6.15 (0.57-67.30) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 2.47 (0.56-11.30) | 1.87 (0.65-5.33) | 4.06 (0.60-28.70) | 1.36 (0.18-10.90) | 1.73 (0.34-9.23) | 1.68 (0.33-9.23) | Erlo+Bev | 1.65 (0.21-14.20) | 7.90 (2.24-30.00) | 10.30 (0.92-121.00) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 1.50 (0.34-6.39) | 1.13 (0.18-6.72) | 2.45 (0.25-25.50) | 0.83 (0.10-6.24) | 1.05 (0.16-6.84) | 1.01 (0.16-6.54) | 0.61 (0.07-4.83) | Gef+Peme | 4.79 (0.76-31.20) | 6.24 (0.41-100.00) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 0.31 (0.10-0.91) | 0.24 (0.10-0.50) | 0.51 (0.13-2.19) | 0.17 (0.03-0.99) | 0.22 (0.07-0.69) | 0.21 (0.07-0.67) | 0.13 (0.03-0.45) | 0.21 (0.03-1.31) | Plat | 1.30 (0.16-10.70) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 0.24 (0.02-2.47) | 0.18 (0.02-1.63) | 0.39 (0.09-1.79) | 0.13 (0.01-1.95) | 0.17 (0.02-1.76) | 0.16 (0.01-1.76) | 0.10 (0.01-1.09) | 0.16 (0.01-2.44) | 0.77 (0.09-6.16) | WBRT | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Results for exon 19 deletion are shown in blue-colour cells, results for exon 21 Leu 858Arg mutation are in gray-color cells. Comparisons should be read from left to right and the estimate is in the cell in common between the column-defining drugs and the row-defining treatment. For 19 del and 21 L858R mutations, HRs (and 95% CrI) less than 1 favour the column-defining treatment. To obtain HRs for comparisons in the opposite direction, reciprocals should be taken. Significant results are in bold and underscored. Abbreviations: Gef, gefitinib; Erlo, erlotinib; Ico, icotinib; Dac, dacomitinib; Afa, afatinib; Osi, osimertinib; Erlo+Bev, erlotinib+bevacizumab; Gef+Peme, gefitinib+pemetrexed; Plat, platinum-based therapy. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Sensitivity analysis
Sensitivity analyses helped to confirm the robustness of these findings, which put more weight on main outcomes. The results restricted to phase III trials (trial number = 34 and patient number = 7448) did not show significant deviations compared with the original network Bayesian analyses; however, they showed a more robust status of Osi that had better OS and PFS. Superiority of Erlo + Bev on PFS was also stressed. The results of the primary meta-analysis remained stable across sensitivity analyses by removing Reck et al. [45], (IMpower150) and Soria et al. [42], (FLAURA), respectively. The superiority of Osi was confirmed to be robust. When restricted to Asia (trial number = 12 and patient number = 1977), the superiority of Osi on PFS slightly declined, but Osi was still among the top-ranking treatments. Erlo + Bev and EGFR-TKI + aVEGFR + Plat showed favorable PFS across Asian and non-Asian populations (trial number = 29 and patient number = 6408).
Discussion
With increasing molecular research focused on biomarkers for NSCLC, attention has been drawn to targeted therapies and immunotherapy. The available guidelines have helped improve the level of clinical treatment, but they still need to be updated as more evidence and clinical trials emerge. In the present study, we gathered evidence from 41 RCTs assessing the role of 22 treatment-level options and 22 class-level options for the intended populations. Herein, we found that Erlo+Bev, Osi, Afa, and Erlo are optimal treatment-level options in terms of PFS and that Osi, Cet+Bev+Plat, Cil+Cet+Plat, and Cet+Plat are successively optimal options in terms of OS. For medications at the class level, ET+aVEGFR, 3rd-gen ET, ET+aVEGFR+Plat, and IT+aVEGFR+Plat outperform other treatments in terms of PFS, while ET+aVEGFR+Plat, followed by ET+Plat and 3rd-gen ET, are better alternatives in terms of OS. Additionally, Erlo+Bev and Osi were superior to other treatments for 19 del and 21 L858R mutations in terms of PFS. Although these were modest differences, most ORR and DoR results were consistent with the PFS and OS results. Erlo +Bev elicited the most severe AEs, which should be properly managed during clinical use. Regarding the efficacy, top-ranking treatments for advanced EGFR-mutant NSCLC also include first-line settings such as osimertinib, while their AEs should be considered in clinical medication. These findings provide crucial implications for clinical reference.
ET monotherapy has been established as the standard treatment for patients with EGFR-positive NSCLC, and a meta-analysis involving patients who had not previously received treatment showed a median PFS of 11 months with ET (gefitinib/erlotinib) versus 5.6 months with chemotherapy [56]. However, most patients with lung cancer are diagnosed at an advanced stage, and the prognosis remains poor despite novel therapeutics. To improve PFS, combination treatments with 1st-gen ET and 2nd-gen ET have been evaluated in several clinical trials [57]. Bev inhibits angiogenesis by restricting oxygen and nutrient supplies to suppress tumor growth, and combination chemotherapy with Bev has been shown to be effective against CNS metastasis and pleural effusion [58, 59], but the conclusion is controversial. Several clinical trials (JO25567, NEJ026) [22, 46] included in this study also compared the efficacy of Erlo+Bev with that of Erlo, and the results showed that PFS was significantly prolonged in the Erlo+Bev group. A meta-analysis [60] investigating Erlo+Bev compared with monotherapy for NSCLC found no substantial benefits for the OS or PFS of all patients, but the combination treatment significantly enhanced OS for EGFR-mutant patients. The mechanism by which Erlo+Bev improves PFS is still unclear, but there are some hypotheses. Bev might normalize blood flow, thus improving drug delivery to tumor blood vessels [61, 62]. Autocrine or paracrine signaling by the VEGF receptor might catalyze cancer cell proliferation and produce anti-apoptotic effects, which could be inhibited by Bev to restore apoptosis [63]. Resistance by Erlo+Bev to the VEGF-mediated pathway has been confirmed in basic research [64]. Previous evidence has demonstrated that the response induced by the 21 L858R mutation was inferior to that induced by the 19 del mutation, which could be improved by Erlo+Bev. However, in this study, Erlo+Bev was found to be the preferred treatment for both 21 L858R and 19 del mutations. Although the OS endpoint was or met and was not significantly different, further clinical validation is still needed. When combined with ET, Bev+Plat has shown promising efficacy among patients with liver metastasis harboring EGFR mutations, which was consistent with our findings. Accordingly, Erlo+ET+Plat also offers potential benefits for patients with advanced EGFR-mutant NSCLC.
Osi is an oral 3rd-gen ET that selectively inhibits ET sensitivity and EGFR T790M resistance (present in almost 60% of patients) and has been approved for the treatment of patients with metastatic T790M-positive NSCLC. Preclinical and clinical data (AURA3) [34, 65] support the ability of Osi to cross the blood-brain barrier and penetrate the CNS; the PFS of advanced EGFR-mutant NSCLC in the FLAURA trial [42] was significantly enhanced in the Osi group. Moreover, the OS of the Osi group was also significantly enhanced (38.6 months vs 31.8 months of standard ET; HR 0.48, 95% CI, 0.26-0.86), as reported by an abstract presented at the 2019 ESMO conference. Patients with T790M resistance mutations who receive Osi may still eventually exhibit progression, and thus, they require better treatment options. Reck et al. [45] noted improved survival with immunotherapy+Bev+Plat compared with Bev+Plat, which suggested the potential priority of immunotherapy in the intended populations. The randomized phase III trial CheckMate 227 [66] demonstrated that nivolumab+ipilimumab resulted in a longer OS duration than chemotherapy, regardless of the programmed cell death-ligand 1 (PD-L1) expression level. Before the further application of immunotherapy in real clinical practice, we should understand the role of tumor mutational burden (TMB) as a biomarker and note safety concerns.
This study has several clinical implications and strengths that should be mentioned. To the best of our knowledge, this study is the most comprehensive work comparing treatment effectiveness for advanced EGFR-mutant NSCLC to date. Current national and international guidelines are mostly based on the results of single RCTs, as well as standard meta-analyses dedicated to the pairwise comparisons of two or three treatments. Moreover, with the large number of treatment options, meta-analyses of direct comparisons are inevitably limited by the relatively small number of studies assessing a particular pair of treatments. Meta-analyses on multiple treatments reduce this issue by creating indirect comparisons and allowing data synthesis that helps to identify the best treatment. This study is the first to address the efficacy of therapeutics for advanced EGFR-mutant NSCLC with state-of-the art Bayesian methods. The conclusions are also strengthened by the highest level of evidence. Finally, this study is based on multivariable, time-varying HRs that assumed proportional hazards, examined the relative treatment efficacy based on parameters of survival plots (shape and scale), and considered the influence of time [67]. Network studies regarding relative risks or odds ratios do not have these strengths.
There are also several limitations that should be acknowledged. First, one or two studies were omitted from the treatment-level and medication-level analyses for not contributing to a complete network loop. Although the credibility of our conclusion might not be maximized, the omitted studies had little impact on our final results. Second, the results on ORR and DoR might not be as well reported because of the limited study number. Additionally, potential AEs could influence our judgment on the preferred treatment. Third, the OS outcome was not as comprehensively reported by the primary studies as PFS, and the results based on statistical data still need clinical validation.
Conclusions
This is the first Bayesian NMA to show promising, significant efficacy and safety for Erlo+Bev, Erlo+Bev+Plat and Osi over other available treatments for advanced EGFR-mutant NSCLC, considering the balance between PFS and OS. Erlo+Bev and Osi are the top-ranked regimens for patients with either 19 del or 21 L858R mutations. Accordingly, the three strategies can be reasonably recommended to these intended patients based on their effectiveness. However, their AEs should also be determined in real clinical practice. IT+aVEGFR+Plat is a potential superior regimen but still needs to be confirmed by more clinical trials. The current study not only provides evidence for the use of Erlo+Bev but also addresses the landscape of the use of Osi in advanced EGFR-mutant NSCLC. In the future, more evidence is needed to develop novel therapies and to identify the best treatment options for patients according to their NSCLC subtype and for those with site-specific metastases.
Materials and Methods
This article was based on the Preferred Reporting Items for Systematic Reviews and Meta-Analyses extension statement for network meta-analyses of health care interventions (Appendix Table 1 in the Supplementary Data). The protocol was registered with PROSPERO (CRD42019137033).
Search strategy
Related published trials were identified after a rigorous literature search in PubMed, EMBASE, Cochrane Library and Clinical Trials.gov from their inception to September 2019. The key terms used were “EGFR mutant”, “non-small cell lung cancer”, “NSCLC”, and “randomized controlled trials” (Appendix Material and Methods in the Supplementary Data). No language restrictions were applied. Reference lists were searched manually for additional records.
Selection criteria
All published RCTs involving adult patients (≥18 years) whose Eastern Cooperative Oncology Group (ECOG) performance status was 0 or 1 and comparing any systematic interventions (pharmaceutical, surgical, radiological, combinations, etc.) for histologically or cytologically confirmed advanced (stage III/IV/recurrent/distant metastasis (brain, liver, bone, etc.)) EGFR-mutant NSCLC was identified. There were no mandatory restrictions on first-line treatment settings or other-line settings. The included patients within the selected trials must have positive and clear advanced EGFR-mutant cancer diagnoses. The duration period of the eligible trials was no less than 6 months. No further restrictions were applied on other individual-level (i.e., age, sex) or program-level characteristics (i.e., start year, follow-up period). If a multi-arm trial compared one treatment to two or more different treatments, we extracted every arm/comparison. The most recent and informative publication was selected to avoid duplications. We excluded trials comparing different administration schemes with the same drug or combinations. Dose-expansion trials, reviews, and fundamental experiments were also excluded.
Definitions of outcomes and treatment arms
In this study, the primary outcomes were PFS and OS according to the Response Evaluation Criteria in Solid Tumors (RECIST version 1.1). The secondary outcomes were objective response rate (ORR), duration of response (DoR, month) and grade 3 or higher adverse events (AEs) (severe AEs). Eligible studies should report at least one of both clinical outcomes. EGFR mutations included exon 19 deletion (19 del), exon 21 Leu858Arg mutation (21 L858R) and other uncommon mutations (19 del and 21 L858R were the main focus) [47].
To organize the current treatment options in clinical trials into clinically meaningful arms, we used general prespecified criteria, as shown in Table 1. Cilengitide (Cil) and cetuximab (Cet) are seldom used in NSCLC, and for statistical convenience and network simplification, Cil is categorized in the aVEGFR class, and Cet is categorized in the ET class [48].
Data extraction and quality assessment
Relevant data were independently extracted by two investigators following our prespecified protocol. Any discrepancies were resolved by discussion with a third investigator. The extracted information included characteristics of the eligible trials (publication year, first author, trial name, follow-up period, number of arms, etc.), characteristics of the populations (mean age, number of enrolled patients, etc.), and characteristics of the program (types of systematic interventions, outcomes of intended endpoints, registration information, etc.). Outcome estimates were extracted using fully adjusted models. Additionally, we contacted the authors if there were any missing data. If we received no response, the analysis was performed without these data. Intent-to-treat data were used when available.
The risk of bias of the included RCTs was assessed using the modified Cochrane Collaboration’s risk of bias tool [49]. The two coauthors performed a quality assessment on all the included RCTs. In the case of disagreements, the two authors rechecked the original articles, and a consensus was achieved after a discussion.
Statistical analysis
For PFS and OS, the hazard ratios (HRs) and confidence intervals (CIs) were directly extracted from the original studies or were calculated by methods provided by Tierney et al. [50]. We also tried to contact the authors if the study provided only figures without exact data. If the authors did not respond, the program Engauge Digitizer 4.1 (http://digitizer.sourceforge.net) was run to extract the exact data from the figures. Odd ratios (ORs) for ORR and grade 3 or higher AEs were manually calculated based on extracted information.
A Bayesian network meta-analysis (NMA) was performed with a random effects model to estimate the HR and 95% credible interval (95% CrI) for direct and indirect evidence on advanced EGFR-mutant NSCLC by combining multiple systematic arms across studies with all the information regarding PFS and OS. In the case of multi-arm trials (trials with three or more interventions), adjustments were made to preserve randomization and correlation within the multi-arm trials by converting log-HRs to log-hazards. ORs and 95% CrI in the random effects model were prepared for ORR and grade 3 or higher AEs for direct and indirect evidence; the mean difference (MD) and 95% CrI in random effects was conducted for DoR because DoR was regarded as a continuous variable. Following the Cochrane Handbook [49], the standard deviation (SD) was roughly computed by the (Xmax-Xmin)/range difference for further analysis.
The Markov chain Monte Carlo (MCMC) method was used to estimate the posterior distribution of each parameter, and the fit of the random effects model was assessed by the deviance information criteria (DIC) [51, 52]. A hierarchical Bayesian model synthesizes comparisons between the treatment pairs and simultaneously summarizes all outcomes of interest by assuming a common heterogeneity parameter (a derived I2 statistic > 50% or a P value for Cochran Q chi-square test <0.1 was regarded as indicating significant heterogeneity) [53, 54]; the inconsistency of this model was evaluated by the edge-splitting method based on all direct and indirect evidence [54]. To confirm the robustness of our findings, sensitivity analyses were performed with studies restricted to phase III trials, studies excluding Reck et al. [45] and Soria et al. [42], respectively and Asian and non-Asian studies. The relative treatment rankings were graphically displayed with rankograms [55].
In the Bayesian context, the statistical significance of HRs and ORs was established when the 95% CrI did not contain 1, and that of MDs was established when it did not contain 0. Calculations were performed in R version 3.5.3 (www.r-project.org) using the gemtc and rjags packages, which are publicly available. The detailed statistical methods are provided in the Appendix Material and Methods in the Supplementary Data.
Data availability statement
Data sharing is not applicable to this article as no new data were created or analyzed in this study.
Supplementary Materials
Author Contributions
All authors designed and conducted this review. Xinmin Zeng, and Binghao Zhao wrote the paper. Xinmin Zeng and Qinghua Zeng helped the study design. Wenxiong Zhang revised the statistical methodology. Qinghua Zeng and Binghao Zhao had primary responsibility for the final content. All authors read and approved the final manuscript.
Acknowledgments
The authors thank professor Wenbin Ma, MD (Peking Union Medical College Hospital, Chinese Academy of Medical Sciences and Peking Union Medical College) for his advice and professor Jichun Liu, MD, PhD (The second affiliated hospital of Nanchang University) for his data collection. The authors also thank the support from National Natural Science Foundation of China (NSFC).
Conflicts of Interest
The authors declare no conflict of interest.
Funding
The work is supported by Chinese Academy of Medical Sciences Innovation Fund for Medical Sciences (Grant number: 2016-I2M-2-001), National Natural Science Foundation of China (NSFC) (Grant number: 81560345, 81860379), Natural Science Foundation of Jiangxi Province (Grant number: 20181BBG78023) and by Science and technology project of Jiangxi health and Health Commission (Grant number: 20197015).
References
- 1. Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A. Global cancer statistics, 2012. CA Cancer J Clin. 2015; 65:87–108. https://doi.org/10.3322/caac.21262 [PubMed]
- 2. Siegel RL, Miller KD, Jemal A. Cancer statistics, 2019. CA Cancer J Clin. 2019; 69:7–34. https://doi.org/10.3322/caac.21551 [PubMed]
- 3. Barlesi F, Mazieres J, Merlio JP, Debieuvre D, Mosser J, Lena H, Ouafik L, Besse B, Rouquette I, Westeel V, Escande F, Monnet I, Lemoine A, et al, and Biomarkers France contributors. Routine molecular profiling of patients with advanced non-small-cell lung cancer: results of a 1-year nationwide programme of the French Cooperative Thoracic Intergroup (IFCT). Lancet. 2016; 387:1415–26. https://doi.org/10.1016/S0140-6736(16)00004-0 [PubMed]
- 4. Hanna N, Johnson D, Temin S, Baker S
Jr , Brahmer J, Ellis PM, Giaccone G, Hesketh PJ, Jaiyesimi I, Leighl NB, Riely GJ, Schiller JH, Schneider BJ, et al. Systemic Therapy for Stage IV Non-Small-Cell Lung Cancer: American Society of Clinical Oncology Clinical Practice Guideline Update. J Clin Oncol. 2017; 35:3484–515. https://doi.org/10.1200/JCO.2017.74.6065 [PubMed] - 5. Créquit P, Trinquart L, Yavchitz A, Ravaud P. Wasted research when systematic reviews fail to provide a complete and up-to-date evidence synthesis: the example of lung cancer. BMC Med. 2016; 14:8. https://doi.org/10.1186/s12916-016-0555-0 [PubMed]
- 6. Crinò L, Cappuzzo F, Zatloukal P, Reck M, Pesek M, Thompson JC, Ford HE, Hirsch FR, Varella-Garcia M, Ghiorghiu S, Duffield EL, Armour AA, Speake G, Cullen M. Gefitinib versus vinorelbine in chemotherapy-naive elderly patients with advanced non-small-cell lung cancer (INVITE): a randomized, phase II study. J Clin Oncol. 2008; 26:4253–60. https://doi.org/10.1200/JCO.2007.15.0672 [PubMed]
- 7. Goss G, Ferry D, Wierzbicki R, Laurie SA, Thompson J, Biesma B, Hirsch FR, Varella-Garcia M, Duffield E, Ataman OU, Zarenda M, Armour AA. Randomized phase II study of gefitinib compared with placebo in chemotherapy-naive patients with advanced non-small-cell lung cancer and poor performance status. J Clin Oncol. 2009; 27:2253–60. https://doi.org/10.1200/JCO.2008.18.4408 [PubMed]
- 8. Mok TS, Wu YL, Thongprasert S, Yang CH, Chu DT, Saijo N, Sunpaweravong P, Han B, Margono B, Ichinose Y, Nishiwaki Y, Ohe Y, Yang JJ, et al. Gefitinib or carboplatin-paclitaxel in pulmonary adenocarcinoma. N Engl J Med. 2009; 361:947–57. https://doi.org/10.1056/NEJMoa0810699 [PubMed]
- 9. Maemondo M, Inoue A, Kobayashi K, Sugawara S, Oizumi S, Isobe H, Gemma A, Harada M, Yoshizawa H, Kinoshita I, Fujita Y, Okinaga S, Hirano H, et al, and North-East Japan Study Group. Gefitinib or chemotherapy for non-small-cell lung cancer with mutated EGFR. N Engl J Med. 2010; 362:2380–88. https://doi.org/10.1056/NEJMoa0909530 [PubMed]
- 10. Zhou C, Wu YL, Chen G, Feng J, Liu XQ, Wang C, Zhang S, Wang J, Zhou S, Ren S, Lu S, Zhang L, Hu C, et al. Erlotinib versus chemotherapy as first-line treatment for patients with advanced EGFR mutation-positive non-small-cell lung cancer (OPTIMAL, CTONG-0802): a multicentre, open-label, randomised, phase 3 study. Lancet Oncol. 2011; 12:735–42. https://doi.org/10.1016/S1470-2045(11)70184-X [PubMed]
- 11. Gridelli C, Ciardiello F, Gallo C, Feld R, Butts C, Gebbia V, Maione P, Morgillo F, Genestreti G, Favaretto A, Leighl N, Wierzbicki R, Cinieri S, et al. First-line erlotinib followed by second-line cisplatin-gemcitabine chemotherapy in advanced non-small-cell lung cancer: the TORCH randomized trial. J Clin Oncol. 2012; 30:3002–11. https://doi.org/10.1200/JCO.2011.41.2056 [PubMed]
- 12. Miller VA, Hirsh V, Cadranel J, Chen YM, Park K, Kim SW, Zhou C, Su WC, Wang M, Sun Y, Heo DS, Crino L, Tan EH, et al. Afatinib versus placebo for patients with advanced, metastatic non-small-cell lung cancer after failure of erlotinib, gefitinib, or both, and one or two lines of chemotherapy (LUX-Lung 1): a phase 2b/3 randomised trial. Lancet Oncol. 2012; 13:528–38. https://doi.org/10.1016/S1470-2045(12)70087-6 [PubMed]
- 13. Pirker R, Pereira JR, von Pawel J, Krzakowski M, Ramlau R, Park K, de Marinis F, Eberhardt WE, Paz-Ares L, Störkel S, Schumacher KM, von Heydebreck A, Celik I, O’Byrne KJ. EGFR expression as a predictor of survival for first-line chemotherapy plus cetuximab in patients with advanced non-small-cell lung cancer: analysis of data from the phase 3 FLEX study. Lancet Oncol. 2012; 13:33–42. https://doi.org/10.1016/S1470-2045(11)70318-7 [PubMed]
- 14. Rosell R, Carcereny E, Gervais R, Vergnenegre A, Massuti B, Felip E, Palmero R, Garcia-Gomez R, Pallares C, Sanchez JM, Porta R, Cobo M, Garrido P, et al, and Spanish Lung Cancer Group in collaboration with Groupe Français de Pneumo-Cancérologie and Associazione Italiana Oncologia Toracica. Erlotinib versus standard chemotherapy as first-line treatment for European patients with advanced EGFR mutation-positive non-small-cell lung cancer (EURTAC): a multicentre, open-label, randomised phase 3 trial. Lancet Oncol. 2012; 13:239–46. https://doi.org/10.1016/S1470-2045(11)70393-X [PubMed]
- 15. Scagliotti GV, Krzakowski M, Szczesna A, Strausz J, Makhson A, Reck M, Wierzbicki RF, Albert I, Thomas M, Miziara JE, Papai ZS, Karaseva N, Thongprasert S, et al. Sunitinib plus erlotinib versus placebo plus erlotinib in patients with previously treated advanced non-small-cell lung cancer: a phase III trial. J Clin Oncol. 2012; 30:2070–78. https://doi.org/10.1200/JCO.2011.39.2993 [PubMed]
- 16. Zhang L, Ma S, Song X, Han B, Cheng Y, Huang C, Yang S, Liu X, Liu Y, Lu S, Wang J, Zhang S, Zhou C, et al, and INFORM investigators. Gefitinib versus placebo as maintenance therapy in patients with locally advanced or metastatic non-small-cell lung cancer (INFORM; C-TONG 0804): a multicentre, double-blind randomised phase 3 trial. Lancet Oncol. 2012; 13:466–75. https://doi.org/10.1016/S1470-2045(12)70117-1 [PubMed]
- 17. Shi Y, Zhang L, Liu X, Zhou C, Zhang L, Zhang S, Wang D, Li Q, Qin S, Hu C, Zhang Y, Chen J, Cheng Y, et al. Icotinib versus gefitinib in previously treated advanced non-small-cell lung cancer (ICOGEN): a randomised, double-blind phase 3 non-inferiority trial. Lancet Oncol. 2013; 14:953–61. https://doi.org/10.1016/S1470-2045(13)70355-3 [PubMed]
- 18. Wu YL, Lee JS, Thongprasert S, Yu CJ, Zhang L, Ladrera G, Srimuninnimit V, Sriuranpong V, Sandoval-Tan J, Zhu Y, Liao M, Zhou C, Pan H, et al. Intercalated combination of chemotherapy and erlotinib for patients with advanced stage non-small-cell lung cancer (FASTACT-2): a randomised, double-blind trial. Lancet Oncol. 2013; 14:777–86. https://doi.org/10.1016/S1470-2045(13)70254-7 [PubMed]
- 19. Ellis PM, Shepherd FA, Millward M, Perrone F, Seymour L, Liu G, Sun S, Cho BC, Morabito A, Leighl NB, Stockler MR, Lee CW, Wierzbicki R, et al, and NCIC CTG, and Australasian Lung Cancer Trials Group, and NCI Naples Clinical Trials Unit. Dacomitinib compared with placebo in pretreated patients with advanced or metastatic non-small-cell lung cancer (NCIC CTG BR.26): a double-blind, randomised, phase 3 trial. Lancet Oncol. 2014; 15:1379–88. https://doi.org/10.1016/S1470-2045(14)70472-3 [PubMed]
- 20. Kawaguchi T, Ando M, Asami K, Okano Y, Fukuda M, Nakagawa H, Ibata H, Kozuki T, Endo T, Tamura A, Kamimura M, Sakamoto K, Yoshimi M, et al. Randomized phase III trial of erlotinib versus docetaxel as second- or third-line therapy in patients with advanced non-small-cell lung cancer: Docetaxel and Erlotinib Lung Cancer Trial (DELTA). J Clin Oncol. 2014; 32:1902–08. https://doi.org/10.1200/JCO.2013.52.4694 [PubMed]
- 21. Ramalingam SS, Jänne PA, Mok T, O’Byrne K, Boyer MJ, Von Pawel J, Pluzanski A, Shtivelband M, Docampo LI, Bennouna J, Zhang H, Liang JQ, Doherty JP, et al. Dacomitinib versus erlotinib in patients with advanced-stage, previously treated non-small-cell lung cancer (ARCHER 1009): a randomised, double-blind, phase 3 trial. Lancet Oncol. 2014; 15:1369–78. https://doi.org/10.1016/S1470-2045(14)70452-8 [PubMed]
- 22. Seto T, Kato T, Nishio M, Goto K, Atagi S, Hosomi Y, Yamamoto N, Hida T, Maemondo M, Nakagawa K, Nagase S, Okamoto I, Yamanaka T, et al. Erlotinib alone or with bevacizumab as first-line therapy in patients with advanced non-squamous non-small-cell lung cancer harbouring EGFR mutations (JO25567): an open-label, randomised, multicentre, phase 2 study. Lancet Oncol. 2014; 15:1236–44. https://doi.org/10.1016/S1470-2045(14)70381-X [PubMed]
- 23. Wu YL, Zhou C, Hu CP, Feng J, Lu S, Huang Y, Li W, Hou M, Shi JH, Lee KY, Xu CR, Massey D, Kim M, et al. Afatinib versus cisplatin plus gemcitabine for first-line treatment of Asian patients with advanced non-small-cell lung cancer harbouring EGFR mutations (LUX-Lung 6): an open-label, randomised phase 3 trial. Lancet Oncol. 2014; 15:213–22. https://doi.org/10.1016/S1470-2045(13)70604-1 [PubMed]
- 24. Yang JC, Kang JH, Mok T, Ahn MJ, Srimuninnimit V, Lin CC, Kim DW, Tsai CM, Barraclough H, Altug S, Orlando M, Park K. First-line pemetrexed plus cisplatin followed by gefitinib maintenance therapy versus gefitinib monotherapy in East Asian patients with locally advanced or metastatic non-squamous non-small cell lung cancer: a randomised, phase 3 trial. Eur J Cancer. 2014; 50:2219–30. https://doi.org/10.1016/j.ejca.2014.05.011 [PubMed]
- 25. Scagliotti G, von Pawel J, Novello S, Ramlau R, Favaretto A, Barlesi F, Akerley W, Orlov S, Santoro A, Spigel D, Hirsh V, Shepherd FA, Sequist LV, et al. Phase III Multinational, Randomized, Double-Blind, Placebo-Controlled Study of Tivantinib (ARQ 197) Plus Erlotinib Versus Erlotinib Alone in Previously Treated Patients With Locally Advanced or Metastatic Nonsquamous Non-Small-Cell Lung Cancer. J Clin Oncol. 2015; 33:2667–74. https://doi.org/10.1200/JCO.2014.60.7317 [PubMed]
- 26. Soria JC, Wu YL, Nakagawa K, Kim SW, Yang JJ, Ahn MJ, Wang J, Yang JC, Lu Y, Atagi S, Ponce S, Lee DH, Liu Y, et al. Gefitinib plus chemotherapy versus placebo plus chemotherapy in EGFR-mutation-positive non-small-cell lung cancer after progression on first-line gefitinib (IMPRESS): a phase 3 randomised trial. Lancet Oncol. 2015; 16:990–98. https://doi.org/10.1016/S1470-2045(15)00121-7 [PubMed]
- 27. Vansteenkiste J, Barlesi F, Waller CF, Bennouna J, Gridelli C, Goekkurt E, Verhoeven D, Szczesna A, Feurer M, Milanowski J, Germonpre P, Lena H, Atanackovic D, et al. Cilengitide combined with cetuximab and platinum-based chemotherapy as first-line treatment in advanced non-small-cell lung cancer (NSCLC) patients: results of an open-label, randomized, controlled phase II study (CERTO). Ann Oncol. 2015; 26:1734–40. https://doi.org/10.1093/annonc/mdv219 [PubMed]
- 28. Wu YL, Zhou C, Liam CK, Wu G, Liu X, Zhong Z, Lu S, Cheng Y, Han B, Chen L, Huang C, Qin S, Zhu Y, et al. First-line erlotinib versus gemcitabine/cisplatin in patients with advanced EGFR mutation-positive non-small-cell lung cancer: analyses from the phase III, randomized, open-label, ENSURE study. Ann Oncol. 2015; 26:1883–89. https://doi.org/10.1093/annonc/mdv270 [PubMed]
- 29. Zhou C, Wu YL, Chen G, Feng J, Liu XQ, Wang C, Zhang S, Wang J, Zhou S, Ren S, Lu S, Zhang L, Hu C, et al. Final overall survival results from a randomised, phase III study of erlotinib versus chemotherapy as first-line treatment of EGFR mutation-positive advanced non-small-cell lung cancer (OPTIMAL, CTONG-0802). Ann Oncol. 2015; 26:1877–83. https://doi.org/10.1093/annonc/mdv276 [PubMed]
- 30. Cheng Y, Murakami H, Yang PC, He J, Nakagawa K, Kang JH, Kim JH, Wang X, Enatsu S, Puri T, Orlando M, Yang JC. Randomized Phase II Trial of Gefitinib With and Without Pemetrexed as First-Line Therapy in Patients With Advanced Nonsquamous Non-Small-Cell Lung Cancer With Activating Epidermal Growth Factor Receptor Mutations. J Clin Oncol. 2016; 34:3258–66. https://doi.org/10.1200/JCO.2016.66.9218 [PubMed]
- 31. Park K, Tan EH, O’Byrne K, Zhang L, Boyer M, Mok T, Hirsh V, Yang JC, Lee KH, Lu S, Shi Y, Kim SW, Laskin J, et al. Afatinib versus gefitinib as first-line treatment of patients with EGFR mutation-positive non-small-cell lung cancer (LUX-Lung 7): a phase 2B, open-label, randomised controlled trial. Lancet Oncol. 2016; 17:577–89. https://doi.org/10.1016/S1470-2045(16)30033-X [PubMed]
- 32. Urata Y, Katakami N, Morita S, Kaji R, Yoshioka H, Seto T, Satouchi M, Iwamoto Y, Kanehara M, Fujimoto D, Ikeda N, Murakami H, Daga H, et al. Randomized Phase III Study Comparing Gefitinib With Erlotinib in Patients With Previously Treated Advanced Lung Adenocarcinoma: WJOG 5108L. J Clin Oncol. 2016; 34:3248–57. https://doi.org/10.1200/JCO.2015.63.4154 [PubMed]
- 33. Kubota K, Yoshioka H, Oshita F, Hida T, Yoh K, Hayashi H, Kato T, Kaneda H, Yamada K, Tanaka H, Ichinose Y, Park K, Cho EK, et al. Phase III, Randomized, Placebo-Controlled, Double-Blind Trial of Motesanib (AMG-706) in Combination With Paclitaxel and Carboplatin in East Asian Patients With Advanced Nonsquamous Non-Small-Cell Lung Cancer. J Clin Oncol. 2017; 35:3662–70. https://doi.org/10.1200/JCO.2017.72.7297 [PubMed]
- 34. Mok TS, Wu YL, Ahn MJ, Garassino MC, Kim HR, Ramalingam SS, Shepherd FA, He Y, Akamatsu H, Theelen WS, Lee CK, Sebastian M, Templeton A, et al, and AURA3 Investigators. Osimertinib or Platinum-Pemetrexed in EGFR T790M-Positive Lung Cancer. N Engl J Med. 2017; 376:629–40. https://doi.org/10.1056/NEJMoa1612674 [PubMed]
- 35. Shi YK, Wang L, Han BH, Li W, Yu P, Liu YP, Ding CM, Song X, Ma ZY, Ren XL, Feng JF, Zhang HL, Chen GY, et al. First-line icotinib versus cisplatin/pemetrexed plus pemetrexed maintenance therapy for patients with advanced EGFR mutation-positive lung adenocarcinoma (CONVINCE): a phase 3, open-label, randomized study. Ann Oncol. 2017; 28:2443–50. https://doi.org/10.1093/annonc/mdx359 [PubMed]
- 36. Spigel DR, Edelman MJ, O’Byrne K, Paz-Ares L, Mocci S, Phan S, Shames DS, Smith D, Yu W, Paton VE, Mok T. Results From the Phase III Randomized Trial of Onartuzumab Plus Erlotinib Versus Erlotinib in Previously Treated Stage IIIB or IV Non-Small-Cell Lung Cancer: METLung. J Clin Oncol. 2017; 35:412–20. https://doi.org/10.1200/JCO.2016.69.2160 [PubMed]
- 37. Wu YL, Cheng Y, Zhou X, Lee KH, Nakagawa K, Niho S, Tsuji F, Linke R, Rosell R, Corral J, Migliorino MR, Pluzanski A, Sbar EI, et al. Dacomitinib versus gefitinib as first-line treatment for patients with EGFR-mutation-positive non-small-cell lung cancer (ARCHER 1050): a randomised, open-label, phase 3 trial. Lancet Oncol. 2017; 18:1454–66. https://doi.org/10.1016/S1470-2045(17)30608-3 [PubMed]
- 38. Yang JJ, Zhou C, Huang Y, Feng J, Lu S, Song Y, Huang C, Wu G, Zhang L, Cheng Y, Hu C, Chen G, Zhang L, et al. Icotinib versus whole-brain irradiation in patients with EGFR-mutant non-small-cell lung cancer and multiple brain metastases (BRAIN): a multicentre, phase 3, open-label, parallel, randomised controlled trial. Lancet Respir Med. 2017; 5:707–16. https://doi.org/10.1016/S2213-2600(17)30262-X [PubMed]
- 39. Yang JJ, Zhou Q, Yan HH, Zhang XC, Chen HJ, Tu HY, Wang Z, Xu CR, Su J, Wang BC, Jiang BY, Bai XY, Zhong WZ, et al. A phase III randomised controlled trial of erlotinib vs gefitinib in advanced non-small cell lung cancer with EGFR mutations. Br J Cancer. 2017; 116:568–74. https://doi.org/10.1038/bjc.2016.456 [PubMed]
- 40. Herbst RS, Redman MW, Kim ES, Semrad TJ, Bazhenova L, Masters G, Oettel K, Guaglianone P, Reynolds C, Karnad A, Arnold SM, Varella-Garcia M, Moon J, et al. Cetuximab plus carboplatin and paclitaxel with or without bevacizumab versus carboplatin and paclitaxel with or without bevacizumab in advanced NSCLC (SWOG S0819): a randomised, phase 3 study. Lancet Oncol. 2018; 19:101–14. https://doi.org/10.1016/S1470-2045(17)30694-0 [PubMed]
- 41. Mok TS, Cheng Y, Zhou X, Lee KH, Nakagawa K, Niho S, Lee M, Linke R, Rosell R, Corral J, Migliorino MR, Pluzanski A, Sbar EI, et al. Improvement in Overall Survival in a Randomized Study That Compared Dacomitinib With Gefitinib in Patients With Advanced Non-Small-Cell Lung Cancer and EGFR-Activating Mutations. J Clin Oncol. 2018; 36:2244–50. https://doi.org/10.1200/JCO.2018.78.7994 [PubMed]
- 42. Soria JC, Ohe Y, Vansteenkiste J, Reungwetwattana T, Chewaskulyong B, Lee KH, Dechaphunkul A, Imamura F, Nogami N, Kurata T, Okamoto I, Zhou C, Cho BC, et al, and FLAURA Investigators. Osimertinib in Untreated EGFR-Mutated Advanced Non-Small-Cell Lung Cancer. N Engl J Med. 2018; 378:113–25. https://doi.org/10.1056/NEJMoa1713137 [PubMed]
- 43. Wu YL, Ahn MJ, Garassino MC, Han JY, Katakami N, Kim HR, Hodge R, Kaur P, Brown AP, Ghiorghiu D, Papadimitrakopoulou VA, Mok TS. CNS Efficacy of Osimertinib in Patients With T790M-Positive Advanced Non-Small-Cell Lung Cancer: Data From a Randomized Phase III Trial (AURA3). J Clin Oncol. 2018; 36:2702–09. https://doi.org/10.1200/JCO.2018.77.9363 [PubMed]
- 44. Kelly RJ, Shepherd FA, Krivoshik A, Jie F, Horn L. A phase III, randomized, open-label study of ASP8273 versus erlotinib or gefitinib in patients with advanced stage IIIB/IV non-small-cell lung cancer. Ann Oncol. 2019; 30:1127–33. https://doi.org/10.1093/annonc/mdz128 [PubMed]
- 45. Reck M, Mok TS, Nishio M, Jotte RM, Cappuzzo F, Orlandi F, Stroyakovskiy D, Nogami N, Rodríguez-Abreu D, Moro-Sibilot D, Thomas CA, Barlesi F, Finley G, et al, and IMpower150 Study Group. Atezolizumab plus bevacizumab and chemotherapy in non-small-cell lung cancer (IMpower150): key subgroup analyses of patients with EGFR mutations or baseline liver metastases in a randomised, open-label phase 3 trial. Lancet Respir Med. 2019; 7:387–401. https://doi.org/10.1016/S2213-2600(19)30084-0 [PubMed]
- 46. Saito H, Fukuhara T, Furuya N, Watanabe K, Sugawara S, Iwasawa S, Tsunezuka Y, Yamaguchi O, Okada M, Yoshimori K, Nakachi I, Gemma A, Azuma K, et al. Erlotinib plus bevacizumab versus erlotinib alone in patients with EGFR-positive advanced non-squamous non-small-cell lung cancer (NEJ026): interim analysis of an open-label, randomised, multicentre, phase 3 trial. Lancet Oncol. 2019; 20:625–35. https://doi.org/10.1016/S1470-2045(19)30035-X [PubMed]
- 47. Herbst RS, Morgensztern D, Boshoff C. The biology and management of non-small cell lung cancer. Nature. 2018; 553:446–54. https://doi.org/10.1038/nature25183 [PubMed]
- 48. Cedra S, Wiegand S, Kolb M, Dietz A, Wichmann G. Reduced Cytokine Release in Ex Vivo Response to Cilengitide and Cetuximab Is a Marker for Improved Survival of Head and Neck Cancer Patients. Cancers (Basel). 2017; 9:E117. https://doi.org/10.3390/cancers9090117 [PubMed]
- 49. Higgins JP, Altman DG, Gøtzsche PC, Jüni P, Moher D, Oxman AD, Savovic J, Schulz KF, Weeks L, Sterne JA, and Cochrane Bias Methods Group, and Cochrane Statistical Methods Group. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ. 2011; 343:d5928. https://doi.org/10.1136/bmj.d5928 [PubMed]
- 50. Tierney JF, Stewart LA, Ghersi D, Burdett S, Sydes MR. Practical methods for incorporating summary time-to-event data into meta-analysis. Trials. 2007; 8:16. https://doi.org/10.1186/1745-6215-8-16 [PubMed]
- 51. Caldwell DM, Ades AE, Higgins JP. Simultaneous comparison of multiple treatments: combining direct and indirect evidence. BMJ. 2005; 331:897–900. https://doi.org/10.1136/bmj.331.7521.897 [PubMed]
- 52. Mavridis D, Salanti G. A practical introduction to multivariate meta-analysis. Stat Methods Med Res. 2013; 22:133–58. https://doi.org/10.1177/0962280211432219 [PubMed]
- 53. Lu G, Ades AE. Combination of direct and indirect evidence in mixed treatment comparisons. Stat Med. 2004; 23:3105–24. https://doi.org/10.1002/sim.1875 [PubMed]
- 54. Higgins JP, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ. 2003; 327:557–60. https://doi.org/10.1136/bmj.327.7414.557 [PubMed]
- 55. Salanti G, Ades AE, Ioannidis JP. Graphical methods and numerical summaries for presenting results from multiple-treatment meta-analysis: an overview and tutorial. J Clin Epidemiol. 2011; 64:163–71. https://doi.org/10.1016/j.jclinepi.2010.03.016 [PubMed]
- 56. Lee CK, Davies L, Wu YL, Mitsudomi T, Inoue A, Rosell R, Zhou C, Nakagawa K, Thongprasert S, Fukuoka M, Lord S, Marschner I, Tu YK, et al. Gefitinib or Erlotinib vs Chemotherapy for EGFR Mutation-Positive Lung Cancer: Individual Patient Data Meta-Analysis of Overall Survival. J Natl Cancer Inst. 2017; 109. https://doi.org/10.1093/jnci/djw279 [PubMed]
- 57. Watanabe S, Yamaguchi OU, Masumoto AI, Maeno Y, Kawashima Y, Ishimoto O, Sugawara S, Yoshizawa H, Kikuchi T, Nukiwa T, Kobayashi K. Phase I Study Evaluating the Combination of Afatinib with Carboplatin and Pemetrexed After First-line EGFR-TKIs. Anticancer Res. 2018; 38:4699–704. https://doi.org/10.21873/anticanres.12776 [PubMed]
- 58. Ilhan-Mutlu A, Osswald M, Liao Y, Gömmel M, Reck M, Miles D, Mariani P, Gianni L, Lutiger B, Nendel V, Srock S, Perez-Moreno P, Thorsen F, et al. Bevacizumab Prevents Brain Metastases Formation in Lung Adenocarcinoma. Mol Cancer Ther. 2016; 15:702–10. https://doi.org/10.1158/1535-7163.MCT-15-0582 [PubMed]
- 59. Kitamura K, Kubota K, Ando M, Takahashi S, Nishijima N, Sugano T, Toyokawa M, Miwa K, Kosaihira S, Noro R, Minegishi Y, Seike M, Yoshimura A, Gemma A. Bevacizumab plus chemotherapy for advanced non-squamous non-small-cell lung cancer with malignant pleural effusion. Cancer Chemother Pharmacol. 2013; 71:457–61. https://doi.org/10.1007/s00280-012-2026-4 [PubMed]
- 60. Zhao B, Zhang W, Yu D, Xu J, Wei Y. Erlotinib in combination with bevacizumab has potential benefit in non-small cell lung cancer: A systematic review and meta-analysis of randomized clinical trials. Lung Cancer. 2018; 122:10–21. https://doi.org/10.1016/j.lungcan.2018.05.011 [PubMed]
- 61. Wildiers H, Guetens G, De Boeck G, Verbeken E, Landuyt B, Landuyt W, de Bruijn EA, van Oosterom AT. Effect of antivascular endothelial growth factor treatment on the intratumoral uptake of CPT-11. Br J Cancer. 2003; 88:1979–86. https://doi.org/10.1038/sj.bjc.6601005 [PubMed]
- 62. Dickson PV, Hamner JB, Sims TL, Fraga CH, Ng CY, Rajasekeran S, Hagedorn NL, McCarville MB, Stewart CF, Davidoff AM. Bevacizumab-induced transient remodeling of the vasculature in neuroblastoma xenografts results in improved delivery and efficacy of systemically administered chemotherapy. Clin Cancer Res. 2007; 13:3942–50. https://doi.org/10.1158/1078-0432.CCR-07-0278 [PubMed]
- 63. Masuda C, Yanagisawa M, Yorozu K, Kurasawa M, Furugaki K, Ishikura N, Iwai T, Sugimoto M, Yamamoto K. Bevacizumab counteracts VEGF-dependent resistance to erlotinib in an EGFR-mutated NSCLC xenograft model. Int J Oncol. 2017; 51:425–34. https://doi.org/10.3892/ijo.2017.4036 [PubMed]
- 64. Furugaki K, Fukumura J, Iwai T, Yorozu K, Kurasawa M, Yanagisawa M, Moriya Y, Yamamoto K, Suda K, Mizuuchi H, Mitsudomi T, Harada N. Impact of bevacizumab in combination with erlotinib on EGFR-mutated non-small cell lung cancer xenograft models with T790M mutation or MET amplification. Int J Cancer. 2016; 138:1024–32. https://doi.org/10.1002/ijc.29848 [PubMed]
- 65. Ballard P, Yates JW, Yang Z, Kim DW, Yang JC, Cantarini M, Pickup K, Jordan A, Hickey M, Grist M, Box M, Johnström P, Varnäs K, et al. Preclinical Comparison of Osimertinib with Other EGFR-TKIs in EGFR-Mutant NSCLC Brain Metastases Models, and Early Evidence of Clinical Brain Metastases Activity. Clin Cancer Res. 2016; 22:5130–40. https://doi.org/10.1158/1078-0432.CCR-16-0399 [PubMed]
- 66. Hellmann MD, Paz-Ares L, Bernabe Caro R, Zurawski B, Kim SW, Carcereny Costa E, Park K, Alexandru A, Lupinacci L, de la Mora Jimenez E, Sakai H, Albert I, Vergnenegre A, et al. Nivolumab plus Ipilimumab in Advanced Non-Small-Cell Lung Cancer. N Engl J Med. 2019; 381:2020–31. https://doi.org/10.1056/NEJMoa1910231 [PubMed]
- 67. Woods BS, Hawkins N, Scott DA. Network meta-analysis on the log-hazard scale, combining count and hazard ratio statistics accounting for multi-arm trials: a tutorial. BMC Med Res Methodol. 2010; 10:54. https://doi.org/10.1186/1471-2288-10-54 [PubMed]


