Introduction
Glioblastoma (GBM) is the most common primary intracranial malignancy in adults [1, 2]. It is highly lethal, with a median survival of only about 14.4 months [3]. An improved understanding of mechanistic causes of GBM may provide a foundation for the improvement of treatment outcomes [4]. Indeed, significant efforts in recent years have explored the molecular pathogenesis of GBM [5].
With the development of high-through sequencing technology, detailed analyses of genetic associations in the pathology of GBM have accelerated. In the past few years, many disease-associated genetic variations in GBM have been documented. Chromosome 7 amplification, chromosome 10 deletions, EGFR amplification, EGFR mutations (point and vIII mutations), and PTEN deletion are high-frequency mutations in primary GBM [6–8]. The pathogenic mechanism of these mutations in GBM has been extensively studied [9–11]. However, the association between age, a risk factor for many tumors, and glioma have been relatively understudied. Epidemiological studies suggest that nearly 80% of gliomas occur in middle-aged and elderly patients [12, 13]. Therefore, we speculated that gliomas occurred more frequently in elderly patients due to the accumulation of genetic mutations or possibly changes in transcriptomics.
Ribonuclease P protein subunit p30 (RPP30), a component of ribonuclease P (RNase P), generates mature tRNA molecules by cleaving the 5'-end. RNase P is also known as RNA polymerase III and is one of three major nuclear RNA polymerases in human cells. Studies have shown that in addition to the modification of tRNA, RNase P is involved in the regulation of gene transcription and cell cycle [14, 15]. Previous work has reported that RPP30 plays a role in tumorigenesis and malignant progression in breast, ovarian, and lung cancers [16–18]. Furthermore, some studies have studied RNase P as a potential therapy for several cancers [19, 20]. Therefore, we speculated that RPP30 may play an important role in glioma with potential as a novel therapeutic target.
In this article, we studied the relationship between age and gliomagenesis. First, age-related genes were screened in primary GBM and non-tumor brain samples. Functional enrichment analysis found that these genes were closely related to gene transcription. Compared with non-tumor brain samples, we found a loss of post-translational protein modification of RPP30 in GBM. In-depth studies have found that RPP30 expression affected the post-transcriptional modification of tumor pathway genes, which may be one of the causes of primary GBM. Finally, we found that RPP30 was closely associated with the clinical molecular pathological features and transcriptional modification of GBM. In conclusion, we found that RPP30, which is expressed less with age, maybe one of the pathogenic factors leading glioma, and RPP30-targeted therapy could potentially be used for clinical treatment of GBM patients.
Results
RPP30, which decreases with age, was an independent prognostic factor in primary GBM
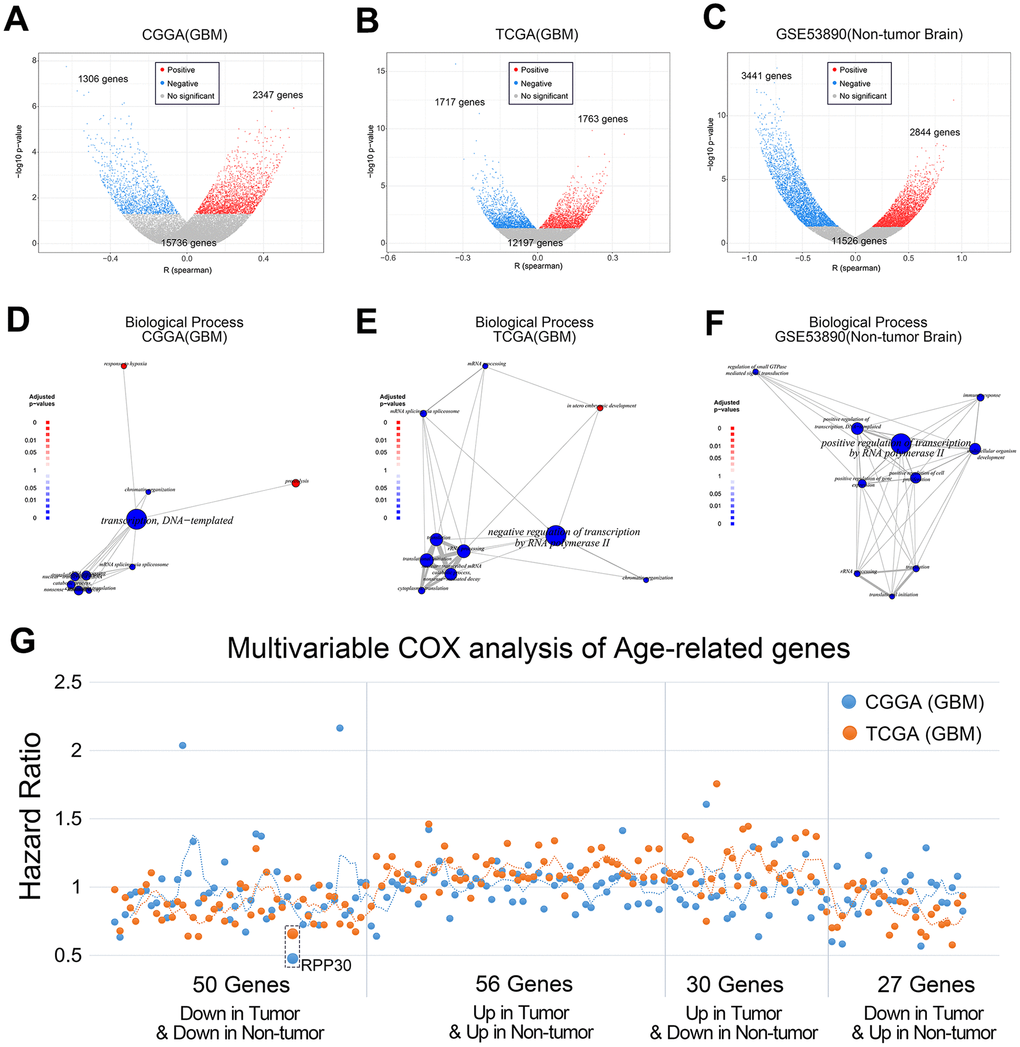
To explore the prognostic significance of age-related genes in primary GBM patients, we performed the following analyses. First, age-related genes from primary GBM and non-tumor brain samples were divided into 4 groups: those whose expression was either up or down in both primary GBM and non-tumor brain samples (56 genes and 50 genes respectively), up in primary GBM but down in non-tumor brain samples (30 genes), or down in primary GBM but up in non-tumor brain samples (27 genes). Subsequently, we performed multivariate COX analysis on these 163 genes including IDH1 mutation status and chemoradiotherapy. Finally, we selected RPP30, an age-related and independent prognostic factor, as the only candidate gene for further study (Figure 1G).
RPP30 behaved differential post-transcriptional modifications in primary GBM brain samples
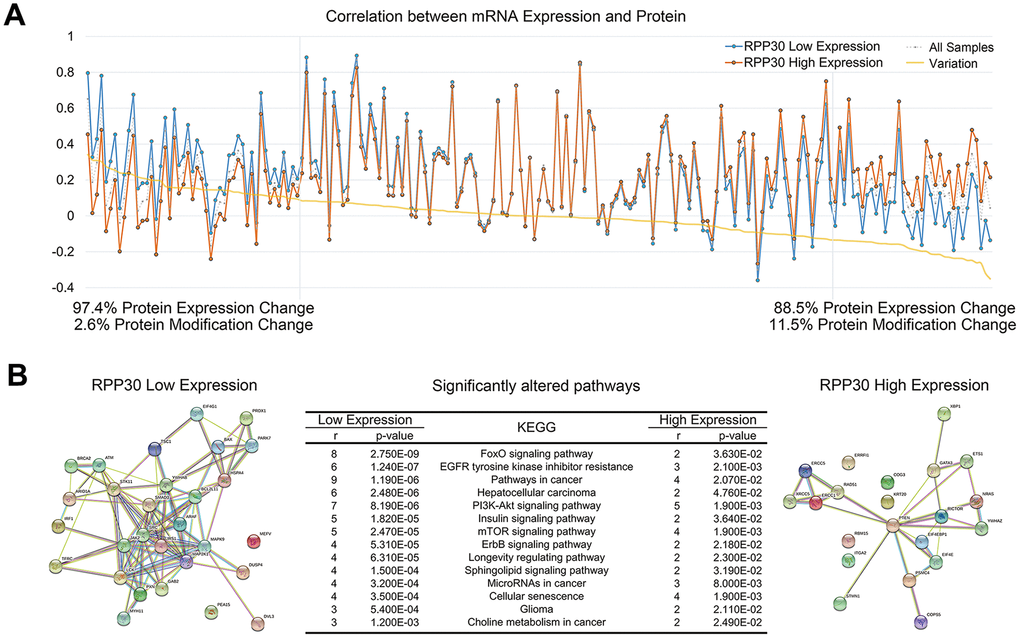
We conducted gene ontology (GO) term enrichment analyses to determine the function of RPP30 in primary GBM and non-tumor brain samples. We found that whereas RPP30 in primary GBM was primarily associated with translation-related functions, it was associated with protein ubiquitination and folding functions in non-tumor brain samples (Figure 2). This result suggested that RPP30 plays a role in different stages of post-transcriptional modifications in primary GBM and non-tumor brain samples. Further, we explored the specific modification functions of RPP30 at each stage of gene expression. Consistent with previous research findings, we found that RPP30 was involved in the modification of RNA, not DNA, in both tumor and non-tumor samples [14, 15, 18]. Surprisingly, we found that RPP30 was closely related to protein modification in non-tumor brain samples, but not in primary GBM samples in both the CGGA and TCGA databases (Figure 2C).
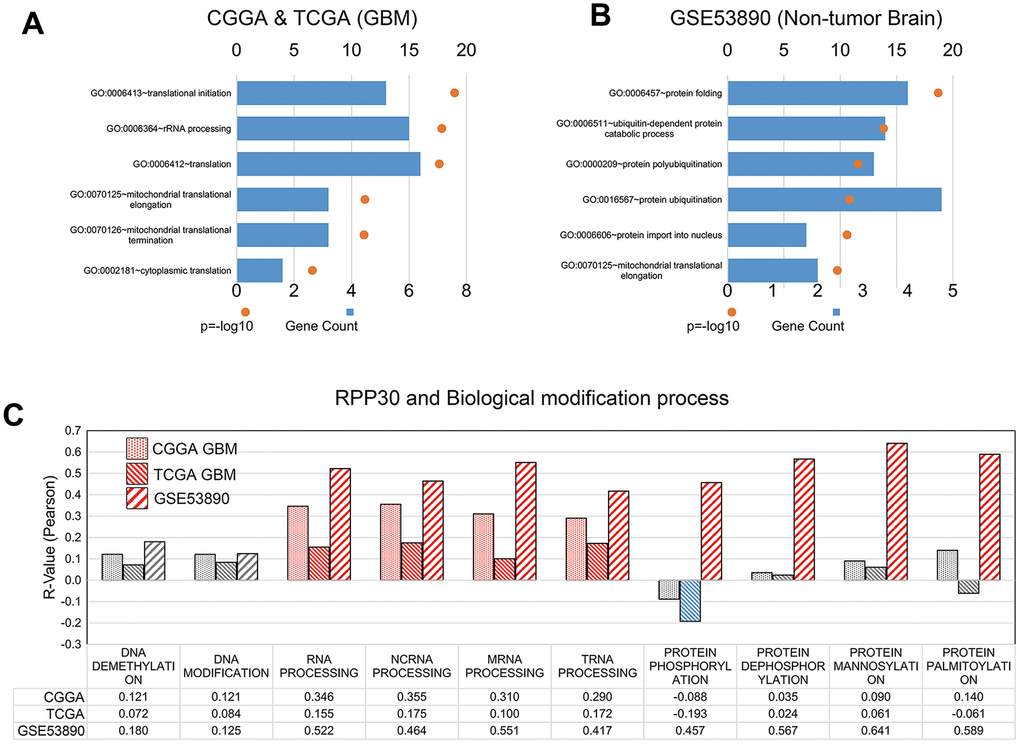
Figure 2. RPP30 involved in the post-translational modification in GBM and non-tumor brain samples. (A) Gene ontology (GO) analyses of RPP30 in GBM. Functional annotation of 500 genes most correlated to age in both CGGA and TCGA databases. (B) Gene ontology (GO) analyses of RPP30 in non-tumor brain samples. Functional annotation of 500 genes most correlated to age in GSE53890. (C) Correlation between RPP30 and GSVA scores of DNA, RNA, and Protein modification in CGGA, TCGA, and GSE53890. Red columns represented significant positive correlation. Blue columns represented significant negative correlation. Gray columns represent no significant correlation. The statistical significance was assessed by Pearson correlation analysis.
RPP30 played a role in transcriptional regulation in primary GBM
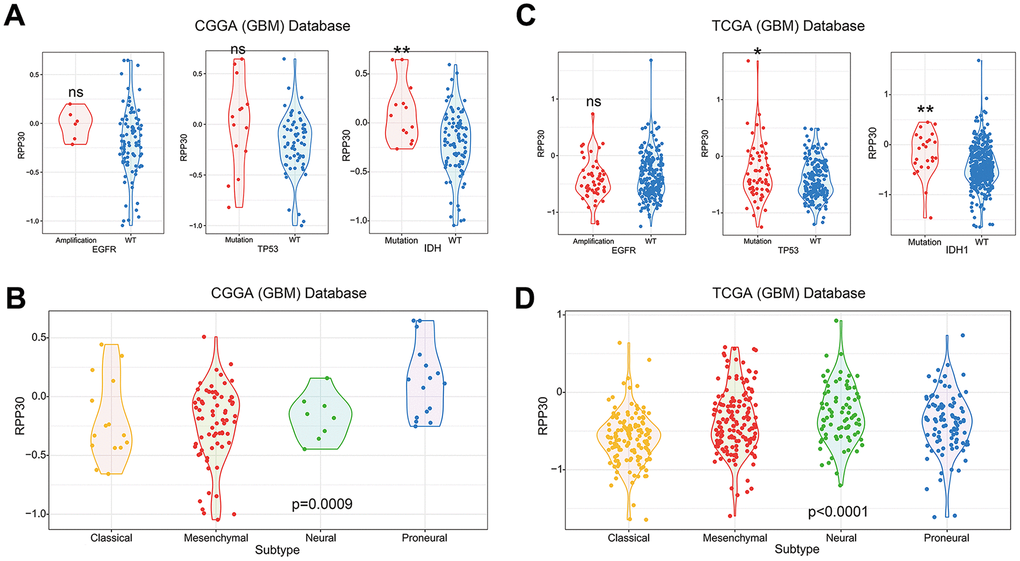
To further illustrate the role of RPP30 in transcriptional regulation, we created heatmaps of RPP30 and transcription-related genes in primary GBM. We found that RPP30 and transcription-related genes shared the same expression pattern in CGGA and TCGA databases (Figure 5A). These genes and their corresponding correlation coefficients are shown in Supplementary Table 3. In addition, we also found that there was no differential expression of RPP30 in male and female patients. This result further suggested that RPP30 plays a role as a transcriptional regulator in primary GBM.
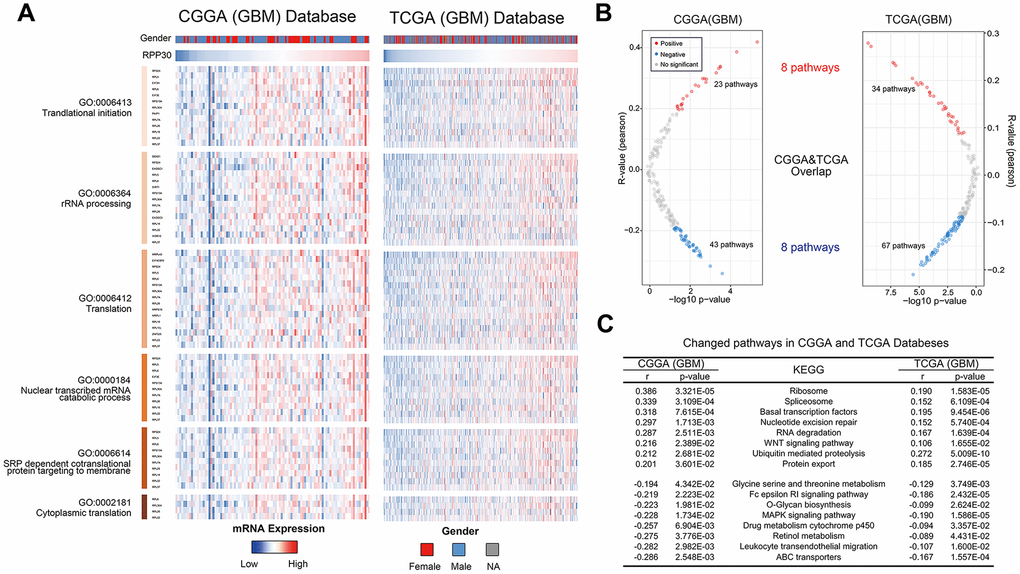
Figure 5. Expression pattern and pathways associated with RPP30 expression in primary GBM of CGGA and TCGA databases. (A) The heatmap showed the expression pattern of RPP30 and transcription-related genes in GBM. Transcription-related genes were obtained from the AmiGO 2 Web portals. Besides, there was no difference in RPP30 expression between different genders of GBM. (B) The scatter plot showed the pathways closely related to RPP30 in CGGA and TCGA databases. There were 8 pathways positively and 8 pathways negatively correlated with RPP30 expression in both CGGA and TCGA databases. (C) The correlation coefficient and p-value of the above 16 pathways with RPP30 were shown in the table. The statistical significance was assessed by Pearson correlation analysis.
RPP30 activated cancer and drug metabolism pathways
To further explore the biological functions of RPP30, we performed a correlation analysis between RPP30 and 186 KEGG pathways in primary GBM. We found 8 positively and 8 negatively correlated pathways associated with RPP30 in the CGGA and TCGA databases (Figure 5B). In addition to transcriptional modification, RPP30 was positively correlated with the WNT signaling pathway (pro-cancer) and negatively correlated with several drug metabolism pathways (cytochrome p450 and ABC transporters) (Figure 5C). Meanwhile, the expression level of RPP30 was significantly correlated with the expression of genes in the cancer-related pathways (Pathway in cancer, WNT Pathway, MAPK Pathway, and WNT Pathway) in both CGGA and TCGA databases (Supplementary Figure 4). These results suggested possible differential biochemical and functional outcomes triggered by RPP30 that may finally result in poorer prognosis in older patients.
RPP30 affected cell proliferation and pathway activation in vitro
To verify the function of RPP30, we performed in vitro experiments. We found that knockdown of RPP30 mRNA expression in HA cells led to the activation of the STAT3 and NF-κB pathways (Figure 6A). Further functional experiments found that knocking down RPP30 increased the proliferation of HA cells (Figure 6B, 6C). In contrast, the activation of tumor-related pathways and proliferation ability of HA cells were impaired by the over-expressed RPP30 (Supplementary Figure 5). Further, we measured the relative expression of RPP30 in non-tumor and GBM samples via qRT-PCR. We found that RPP30 was lowly-expressed in GBM samples (Figure 6D). Together, these results indicate that RPP30 may contribute to the pathogenesis of GBM.
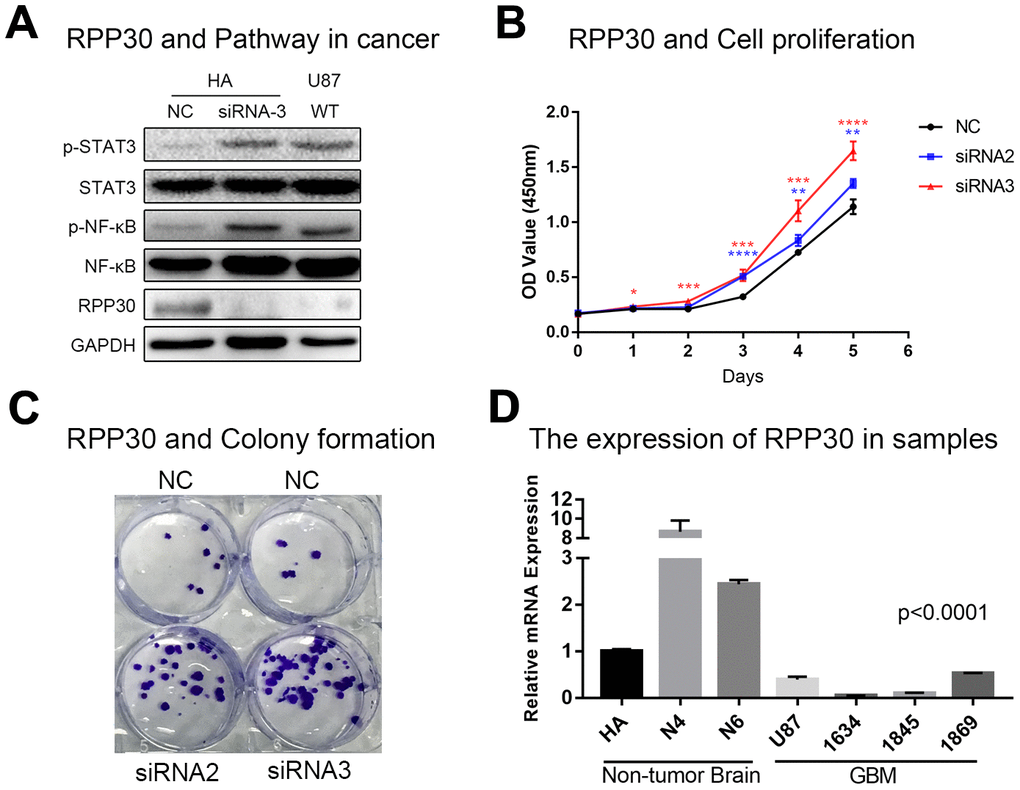
Figure 6. RPP30 regulated protein activation and cell proliferation in vitro. (A) Western blot showed knockdown of RPP30 led to increased expression of p-STAT3 and p-NF-κB in HA cells. (B, C) Cell proliferation ability increased significantly after knocking down RPP30 in HA cells. (D) RPP30 was lowly-expressed in GBM samples by qRT-PCR.
Discussion
Due to our limited understanding of the pathogenesis of GBM, current postoperative treatment is mainly untargeted adjuvant therapy, which is largely ineffective for most patients and has poor prognoses [1, 22]. However, the launch of the TCGA and CGGA projects have catapulted GBM research into a new era. In just the past decade, there have been thousands of high-throughput sequencing studies in GBM. Many genomic alterations in GBM have been identified, including EGFR amplification, EGFR mutations (point and vIII mutations), PTEN deletion, and others [23–26]. However, the primary causes of GBM progression remain unknown. Therefore, individualized targeted therapy is only able to benefit a small number of GBM patients. Therefore, the clinical community is in urgent need of an improved understanding of fundamental pathogenic processes that could serve as therapeutic targets. Advancing age is considered to be the pathogenic factor of many cancers [27, 28]. Previous studies have suggested that age-related epigenetic alterations, accumulated mutation, cell metabolic reprogramming, and changes to the tumor microenvironment play an important role in tumorigenesis [29–37]. This study was designed to explore the mechanistic relationship between age and the pathogenesis of primary GBM.
Unexpectedly, the number of gene mutations did not increase with age in primary GBM. Therefore, we studied the relationship between transcriptome and age. Analysis of transcriptome sequencing data of GBM and non-tumor brain samples revealed that transcription of 163 genes is closely related to age. Importantly, these genes were primarily found to be involved in gene transcription in both tumor and non-tumor brain samples. This suggests that differential epigenetic regulation of the transcriptome is closely related to age in tumor and non-tumor brain samples. Further survival analysis found that only one particular gene - RPP30 - was an independent prognostic factor for primary GBM. Previous studies reported that RPP30 played an important role in gene transcription, with a primary role in making post-translational modifications [38]. Our study found that RPP30 was mainly involved in the post-translational modification in both tumor and non-tumor samples. However, we also found that the specific post-transcriptional modifications made by RPP30 were different in tumor and non-tumor brain samples. Our data suggested that RPP30 was associated with RNA and protein modification in non-tumors but associated with RNA modification in GBM tissue. Indeed, this hypothesis was corroborated by the finding that RPP30 could regulate protein expression, rather than protein modification, by post-transcriptional modification in GBM. The protein regulation role of RPP30 may be an important potential pathogenic factor in GBM. Functional enrichment of RPP30-related proteins showed that these proteins were mainly enriched in cancer-related pathways. In addition, we found that the downregulation of RPP30 can increase phosphorylation/activation of cancer pathway-associated proteins in vitro. Whereas overexpression of RPP30 has the opposite biological functions. Since there is no anti-RPP30 antibody available for immunohistochemistry, we measured the relative expression of RPP30 in non-tumor and GBM brain samples via qRT-PCR. Our findings suggested that RPP30 was lowly expressed in GBM samples. These results suggest that RPP30 might act as a pathogenic factor in GBM by carrying out post-translational modifications of cancer pathway-related proteins. However, significant further research of how these findings may inform a therapeutic strategy is required before translation to the clinic.
Herein, we reported for the first time an analysis of the biological functions of RPP30 in glioma. We found a relationship between RPP30 and specific hotspot mutations and molecular subtypes in GBM. Further, we analyzed RPP30-related transcriptional regulatory genes and molecular pathways. Of interest, the RPP family has previously been studied as a therapeutic target in the treatment of tumors [19]. Our results from this work validate the possibility that RPP30 may be an important pathogenic pathway in the development of GBM. We found that RPP30 likely acts via post-transcriptional editing of select oncogenes. With follow-up developmental studies, we believe that RPP30-targeted therapy might benefit GBM patients.
Materials and Methods
Sample and database
In the CGGA database, we have collected transcriptome data from 109 primary GBM samples, originally generated by the Agilent Whole Human Genome Array platform. Overall survival (OS) was calculated from the data diagnosed as the time of death or last follow-up. Written informed consents were obtained from the patients (or their families) for the CGGA project. We downloaded transcriptome microarray data from 507 samples of primary GBM from the official website of TCGA (https://cancergenome.nih.gov). Transcriptome microarray data from non-tumor brain samples were obtained from GSE53890.
Cell culture
We obtained GBM cell line U87 and astrocytic cell line HA from the Institute of Biochemistry and Cell Biology, Chinese Academy of Science. U87 cells were cultured in DMEM (Gibco) supplemented with 10% fetal bovine serum (Gibco). HA cells were cultured in Astrocyte Medium (Gibco). All cell lines were cultured at 37 °C and 5% CO2.
Screening of candidate genes
Age-related genes were screened out by pearson correlation analysis in primary GBM (from CGGA and TCGA databases) and non-tumor brain samples (from GSE53890). We found 163 genes to be significantly associated with age in both primary GBM and non-tumor brain samples and included those for further analysis (Supplementary Table 1). To explore the prognostic significance of 163 genes in primary GBM, we performed multivariate COX analysis in CGGA and TCGA databases. Finally, we selected RPP30, an age-related gene and prognostic factor of primary GBM, as the candidate gene of focus for the rest of our studies.
Functional enrichment analysis
In this study, we used three functional enrichment methods. We performed functional enrichment of age-related genes by the HTSanalyzeR package in R. The p-value cutoff was 0.01, permutations were 100, and minGeneSetSize was 20. Gene ontology (GO) analyses of RPP30 in GBM and non-tumor brain samples were performed in DAVID (https://david.ncifcrf.gov/). The functional enrichment of RPP30-related proteins was performed in STRING (https://string-db.org/).
Gene Set Variation Analysis (GSVA) analysis
GSVA analysis was performed using the gsva package in R. Analysis was performed using the default parameters. The Gene Ontology (GO) gene set was downloaded from AmiGO 2 Web portals (http://amigo.geneontology.org/amigo/landing). The correlation between genes and biological functions were analyzed by pearson analysis.
Cell proliferation test and colony formation
We conducted a cell proliferation test using the Cell Counting Kit-8 Kit (Dojindo). For the colony formation assay, we plated dissociated single cells at a density of 1 cell/μl and counted the number of colonies that formed after 14 days.
Quantitative Real-time PCR
Expression levels of mRNA were analyzed using the ABI 7500 Real-time PCR System. We calculated the relative mRNA expression levels of RPP30 using the 2–ΔΔCt method. Transcript levels of the GAPDH gene were used for normalization. The primer sequences for various human genes used in this study are listed in Supplementary Table 2.
Statistical analysis
Statistical analyses and figures were executed using R (https://www.r-project.org/, v3.5.0), SPSS software (IBM, v25.0), and Microsoft office 2016. We used SPSS for multivariable COX analysis and the results were depicted using Microsoft office 2016. Other statistical computations and figures were generated using R packages. A p-value less than 0.05 was considered statistically significant. All statistical tests were two-tailed.
Ethics statement
This study was approved by Beijing Tiantan Hospital institutional review board (IRB). All patients provided written informed consent for the publication of all associated data in this study.
Availability of data and materials
The sequencing data of the CGGA database has been published on the CGGA portal website (http://www.cgga.org.cn/). The sequencing data of the TCGA database could be downloaded from the official website of TCGA (https://cancergenome.nih.gov). Transcriptome microarray data from non-tumor brain samples were obtained from GSE53890 (https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE53890). All data analyzed during the study are available from the corresponding author on reasonable request.
Author Contributions
G.Z.L.: data analysis and editing the manuscript. Y.Z., H.J.L., Z.L.W.: data collection and organization of CGGA database. R.Y.H., H.Y.J., Y.M.F., Y.H.C.: data collection and organization of TCGA database. F.W., F.Z.: data collection and organization of GSE53890 database. T.J., W.Z.: conception, supervision, and design of this article.
Acknowledgments
We thank Ms. Shuqing Sun and Hua Huang for tissue sample collection and clinical data retrieval.
Conflicts of Interest
The authors declare no potential conflicts of interest.
Funding
This work was supported by grants from National Natural Science Foundation of China (No. 81672479, 81802994), National Natural Science Foundation of China (NSFC)/Research Grants Council (RGC) Joint Research Scheme (81761168038), Beijing Municipal Administration of Hospitals’ Mission Plan (SML20180501).
References
- 1. Stupp R, Mason WP, van den Bent MJ, Weller M, Fisher B, Taphoorn MJ, Belanger K, Brandes AA, Marosi C, Bogdahn U, Curschmann J, Janzer RC, Ludwin SK, et al, European Organisation for Research and Treatment of Cancer Brain Tumor and Radiotherapy Groups, and National Cancer Institute of Canada Clinical Trials Group. Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med. 2005; 352:987–96. https://doi.org/10.1056/NEJMoa043330 [PubMed]
- 2. Omuro A, DeAngelis LM. Glioblastoma and other Malignant gliomas: a clinical review. JAMA. 2013; 310:1842–50. https://doi.org/10.1001/jama.2013.280319 [PubMed]
- 3. Jiang T, Mao Y, Ma W, Mao Q, You Y, Yang X, Jiang C, Kang C, Li X, Chen L, Qiu X, Wang W, Li W, et al, and Chinese Glioma Cooperative Group (CGCG). CGCG clinical practice guidelines for the management of adult diffuse gliomas. Cancer Lett. 2016; 375:263–73. https://doi.org/10.1016/j.canlet.2016.01.024 [PubMed]
- 4. Hu H, Mu Q, Bao Z, Chen Y, Liu Y, Chen J, Wang K, Wang Z, Nam Y, Jiang B, Sa JK, Cho HJ, Her NG, et al. Mutational landscape of secondary glioblastoma guides MET-targeted trial in brain tumor. Cell. 2018; 175:1665–78.e18. https://doi.org/10.1016/j.cell.2018.09.038 [PubMed]
- 5. Sanai N, Alvarez-Buylla A, Berger MS. Neural stem cells and the origin of gliomas. N Engl J Med. 2005; 353:811–22. https://doi.org/10.1056/NEJMra043666 [PubMed]
- 6. Verhaak RG, Hoadley KA, Purdom E, Wang V, Qi Y, Wilkerson MD, Miller CR, Ding L, Golub T, Mesirov JP, Alexe G, Lawrence M, O'Kelly M, et al. Integrated genomic analysis identifies clinically relevant subtypes of glioblastoma characterized by abnormalities in PDGFRA, IDH1, EGFR, and NF1. Cancer Cell. 2010; 17:98–110. https://doi.org/10.1016/j.ccr.2009.12.020 [PubMed]
- 7. Yan H, Parsons DW, Jin G, McLendon R, Rasheed BA, Yuan W, Kos I, Batinic-Haberle I, Jones S, Riggins GJ, Friedman H, Friedman A, Reardon D, et al. IDH1 and IDH2 mutations in gliomas. N Engl J Med. 2009; 360:765–73. https://doi.org/10.1056/NEJMoa0808710 [PubMed]
- 8. Parsons DW, Jones S, Zhang X, Lin JC, Leary RJ, Angenendt P, Mankoo P, Carter H, Siu IM, Gallia GL, Olivi A, McLendon R, Rasheed BA, et al. An integrated genomic analysis of human glioblastoma multiforme. Science. 2008; 321:1807–12. https://doi.org/10.1126/science.1164382 [PubMed]
- 9. Yin J, Park G, Kim TH, Hong JH, Kim YJ, Jin X, Kang S, Jung JE, Kim JY, Yun H, Lee JE, Kim M, Chung J, et al. Pigment Epithelium-Derived Factor (PEDF) Expression Induced by EGFRvIII Promotes Self-renewal and Tumor Progression of Glioma Stem Cells. PLoS Biol. 2015; 13:e1002152. https://doi.org/10.1371/journal.pbio.1002152 [PubMed]
- 10. Turcan S, Rohle D, Goenka A, Walsh LA, Fang F, Yilmaz E, Campos C, Fabius AW, Lu C, Ward PS, Thompson CB, Kaufman A, Guryanova O, et al. IDH1 mutation is sufficient to establish the glioma hypermethylator phenotype. Nature. 2012; 483:479–83. https://doi.org/10.1038/nature10866 [PubMed]
- 11. Fan QW, Cheng CK, Gustafson WC, Charron E, Zipper P, Wong RA, Chen J, Lau J, Knobbe-Thomsen C, Weller M, Jura N, Reifenberger G, Shokat KM, Weiss WA. EGFR phosphorylates tumor-derived EGFRvIII driving STAT3/5 and progression in glioblastoma. Cancer Cell. 2013; 24:438–49. https://doi.org/10.1016/j.ccr.2013.09.004 [PubMed]
- 12. Ostrom QT, Cote DJ, Ascha M, Kruchko C, Barnholtz-Sloan JS. Adult glioma incidence and survival by race or ethnicity in the United States from 2000 to 2014. JAMA Oncol. 2018; 4:1254–62. https://doi.org/10.1001/jamaoncol.2018.1789 [PubMed]
- 13. Ricard D, Idbaih A, Ducray F, Lahutte M, Hoang-Xuan K, Delattre JY. Primary brain tumours in adults. Lancet. 2012; 379:1984–96. https://doi.org/10.1016/S0140-6736(11)61346-9 [PubMed]
- 14. Mancini C, Messana E, Turco E, Brussino A, Brusco A. Gene-targeted embryonic stem cells: real-time PCR assay for estimation of the number of neomycin selection cassettes. Biol Proced Online. 2011; 13:10. https://doi.org/10.1186/1480-9222-13-10 [PubMed]
- 15. Dyavar SR, Ye Z, Byrareddy SN, Scarsi KK, Winchester LC, Weinhold JA, Fletcher CV, Podany AT. Normalization of cell associated antiretroviral drug concentrations with a novel RPP30 droplet digital PCR assay. Sci Rep. 2018; 8:3626. https://doi.org/10.1038/s41598-018-21882-0 [PubMed]
- 16. Wang J, Ramakrishnan R, Tang Z, Fan W, Kluge A, Dowlati A, Jones RC, Ma PC. Quantifying EGFR alterations in the lung cancer genome with nanofluidic digital PCR arrays. Clin Chem. 2010; 56:623–32. https://doi.org/10.1373/clinchem.2009.134973 [PubMed]
- 17. Chamizo C, Rojo F, Madoz-Gúrpide J. Determination of true ERBB2 gene amplification in breast cancer by quantitative PCR using a reference and a novel control gene. Appl Immunohistochem Mol Morphol. 2016; 24:179–87. https://doi.org/10.1097/PAI.0000000000000160 [PubMed]
- 18. Oscorbin I, Kechin A, Boyarskikh U, Filipenko M. Multiplex ddPCR assay for screening copy number variations in BRCA1 gene. Breast Cancer Res Treat. 2019; 178:545–55. https://doi.org/10.1007/s10549-019-05425-3 [PubMed]
- 19. Cobaleda C, Sánchez-García I. In vivo inhibition by a site-specific catalytic RNA subunit of RNase P designed against the BCR-ABL oncogenic products: a novel approach for cancer treatment. Blood. 2000; 95:731–37. [PubMed]
- 20. Trang P, Kim K, Liu F. Developing RNase P ribozymes for gene-targeting and antiviral therapy. Cell Microbiol. 2004; 6:499–508. https://doi.org/10.1111/j.1462-5822.2004.00398.x [PubMed]
- 21. Gill T, Cai T, Aulds J, Wierzbicki S, Schmitt ME. RNase MRP cleaves the CLB2 mRNA to promote cell cycle progression: novel method of mRNA degradation. Mol Cell Biol. 2004; 24:945–53. https://doi.org/10.1128/mcb.24.3.945-953.2004 [PubMed]
- 22. Stupp R, Hegi ME, Gorlia T, Erridge SC, Perry J, Hong YK, Aldape KD, Lhermitte B, Pietsch T, Grujicic D, Steinbach JP, Wick W, Tarnawski R, et al. Cilengitide combined with standard treatment for patients with newly diagnosed glioblastoma with methylated MGMT promoter (CENTRIC EORTC 26071-22072 study): a multicentre, randomised, open-label, phase 3 trial. Lancet Oncol. 2014; 15:1100–8. https://doi.org/10.1016/S1470-2045(14)70379-1 [PubMed]
- 23. Cancer Genome Atlas Research Network. Comprehensive genomic characterization defines human glioblastoma genes and core pathways. Nature. 2008; 455:1061–68. https://doi.org/10.1038/nature07385 [PubMed]
- 24. Kim H, Zheng S, Amini SS, Virk SM, Mikkelsen T, Brat DJ, Grimsby J, Sougnez C, Muller F, Hu J, Sloan AE, Cohen ML, Van Meir EG, et al. Whole-genome and multisector exome sequencing of primary and post-treatment glioblastoma reveals patterns of tumor evolution. Genome Res. 2015; 25:316–27. https://doi.org/10.1101/gr.180612.114 [PubMed]
- 25. Zadeh G, Bhat KP, Aldape K. EGFR and EGFRvIII in glioblastoma: partners in crime. Cancer Cell. 2013; 24:403–04. https://doi.org/10.1016/j.ccr.2013.09.017 [PubMed]
- 26. Smith JS, Tachibana I, Passe SM, Huntley BK, Borell TJ, Iturria N, O’Fallon JR, Schaefer PL, Scheithauer BW, James CD, Buckner JC, Jenkins RB. PTEN mutation, EGFR amplification, and outcome in patients with anaplastic astrocytoma and glioblastoma multiforme. J Natl Cancer Inst. 2001; 93:1246–56. https://doi.org/10.1093/jnci/93.16.1246 [PubMed]
- 27. Graus F, Bruna J, Pardo J, Escudero D, Vilas D, Barceló I, Brell M, Pascual C, Crespo JA, Erro E, García-Romero JC, Estela J, Martino J, et al. Patterns of care and outcome for patients with glioblastoma diagnosed during 2008-2010 in Spain. Neuro Oncol. 2013; 15:797–805. https://doi.org/10.1093/neuonc/not013 [PubMed]
- 28. Ringel F, Pape H, Sabel M, Krex D, Bock HC, Misch M, Weyerbrock A, Westermaier T, Senft C, Schucht P, Meyer B, Simon M, and SN1 study group. Clinical benefit from resection of recurrent glioblastomas: results of a multicenter study including 503 patients with recurrent glioblastomas undergoing surgical resection. Neuro Oncol. 2016; 18:96–104. https://doi.org/10.1093/neuonc/nov145 [PubMed]
- 29. DePinho RA. The age of cancer. Nature. 2000; 408:248–54. https://doi.org/10.1038/35041694 [PubMed]
- 30. Sperka T, Wang J, Rudolph KL. DNA damage checkpoints in stem cells, ageing and cancer. Nat Rev Mol Cell Biol. 2012; 13:579–90. https://doi.org/10.1038/nrm3420 [PubMed]
- 31. Kroemer G, Pouyssegur J. Tumor cell metabolism: cancer’s achilles’ heel. Cancer Cell. 2008; 13:472–82. https://doi.org/10.1016/j.ccr.2008.05.005 [PubMed]
- 32. Vander Heiden MG, Cantley LC, Thompson CB. Understanding the warburg effect: the metabolic requirements of cell proliferation. Science. 2009; 324:1029–33. https://doi.org/10.1126/science.1160809 [PubMed]
- 33. Cassim S, Raymond VA, Dehbidi-Assadzadeh L, Lapierre P, Bilodeau M. Metabolic reprogramming enables hepatocarcinoma cells to efficiently adapt and survive to a nutrient-restricted microenvironment. Cell Cycle. 2018; 17:903–16. https://doi.org/10.1080/15384101.2018.1460023 [PubMed]
- 34. Cassim S, Raymond VA, Lacoste B, Lapierre P, Bilodeau M. Metabolite profiling identifies a signature of tumorigenicity in hepatocellular carcinoma. Oncotarget. 2018; 9:26868–83. https://doi.org/10.18632/oncotarget.25525 [PubMed]
- 35. Cassim S, Vučetić M, Ždralević M, Pouyssegur J. Warburg and beyond: the power of mitochondrial metabolism to collaborate or replace fermentative glycolysis in cancer. Cancers (Basel). 2020; 12:1119. https://doi.org/10.3390/cancers12051119 [PubMed]
- 36. Cassim S, Pouyssegur J. Tumor microenvironment: a metabolic player that shapes the immune response. Int J Mol Sci. 2019; 21:157. https://doi.org/10.3390/ijms21010157 [PubMed]
- 37. Miranda-Gonçalves V, Lameirinhas A, Henrique R, Jerónimo C. Metabolism and epigenetic interplay in cancer: regulation and putative therapeutic targets. Front Genet. 2018; 9:427. https://doi.org/10.3389/fgene.2018.00427 [PubMed]
- 38. Reiner R, Ben-Asouli Y, Krilovetzky I, Jarrous N. A role for the catalytic ribonucleoprotein RNase P in RNA polymerase III transcription. Genes Dev. 2006; 20:1621–35. https://doi.org/10.1101/gad.386706 [PubMed]