Introduction
Bisphenol A (BPA) is a high-production-volume chemical extensively used in the production of plastics, epoxy resin linings of food packaging, coatings, and fillings, and now leaches into the environment with over one million pounds each year [1]. Despite its low affinity, BPA could bind classical nuclear estrogen receptor (ER) alpha and beta, as well as membrane-associated GPR30, and may inhibit the function of ER and other nuclear hormone receptors for its similar structure as diethylstilbestrol (DES), a highly potent ER agonist [2].
Owing to the ubiquitous presence of BPA in the environment and its serious endocrine-disrupting effects, coupled with the increased incidence of endocrine-associated cancer, many studies have evaluated the carcinogenesis of BPA in hormone-sensitive organs (prostate, testis, breast and ovary et al.) [3, 4]. The US Food and Drug Administration (FDA) has set the safe reference dose (RfD) at 50 μg/kg/d for humans based on a 1,000-fold reduction of the dose used in the rodent study [5]. Recently, the European Food Safety Authority (EFSA) lowered the RfD to 4 μg/kg/d after reevaluating the toxicological data for BPA [6].
To explore the role of BPA on the reproductive system, scholars have carried out many experiments based on rodent models and achieved preliminary results. However, due to the differences in animal models, dosage, administration methods, and the conflicts of interest behind the experiment, the results of the experiments were controversial. For example, a study sponsored by the Polycarbonate/BPA Global Group, which is an organization promoting the welfare and interests of polycarbonate plastics and BPA manufacturers, stated that, in the SD (Sprague-Dawley) rats model, the cancer risk of any organ system exposed to BPA for a long time did not increase regardless of the exposure dose and period [7]. However, another study evaluating BPA exposure prenatally administered to SD rats suggested that at postnatal day (PND) 180, low dose BPA exposure was sufficient to induce hyperplasia/dysplasia of the prostate (control group: 0% vs. BPA group: 62%) [8].
We performed this study to provide a more rigorous assessment of the existing rodent model by systematically reviewing experimental studies reporting BPA and detrimental outcomes. We reviewed and conducted a meta-analysis of these rodent studies which reported the relationship between prenatal or perinatal (early-life) BPA exposure and the following reproductive-related outcomes: body weight, prostate weight, testis weight, epididymis weight, seminal vesicle weight, sperm motility, daily sperm production (DSP; testis), efficiency of sperm production, and epididymal sperm concentration (Table 1).
Table 1. PECO statement (population, exposure, comparator and outcomes).
| Variable | Description |
| Population | Experimental rodent studies |
| Exposure | Early-life exposure to bisphenol A (prenatal and early postnatal; have a exposure history during gestation period to postnatal day 21) |
| Comparator | Animals exposed to vehicle-only treatment |
| Outcomes | body weight, prostate weight, testis weight, epididymis weight, seminal vesicle weight, sperm motility, daily sperm production (testis), efficiency of sperm production (g/testis/day), and epididymal sperm concentrations |
Results
Characteristics of included studies
The flowchart of the literature search and selection process is shown in Figure 1. After searching PubMed, EMBASE, Toxline databases, and additional references for relevant articles, a total of 2883 articles were found. Of these articles, 103 articles met the criteria for full-text review. Eventually, 31 articles were included for analysis (Table 2) [7, 8, 16–44].
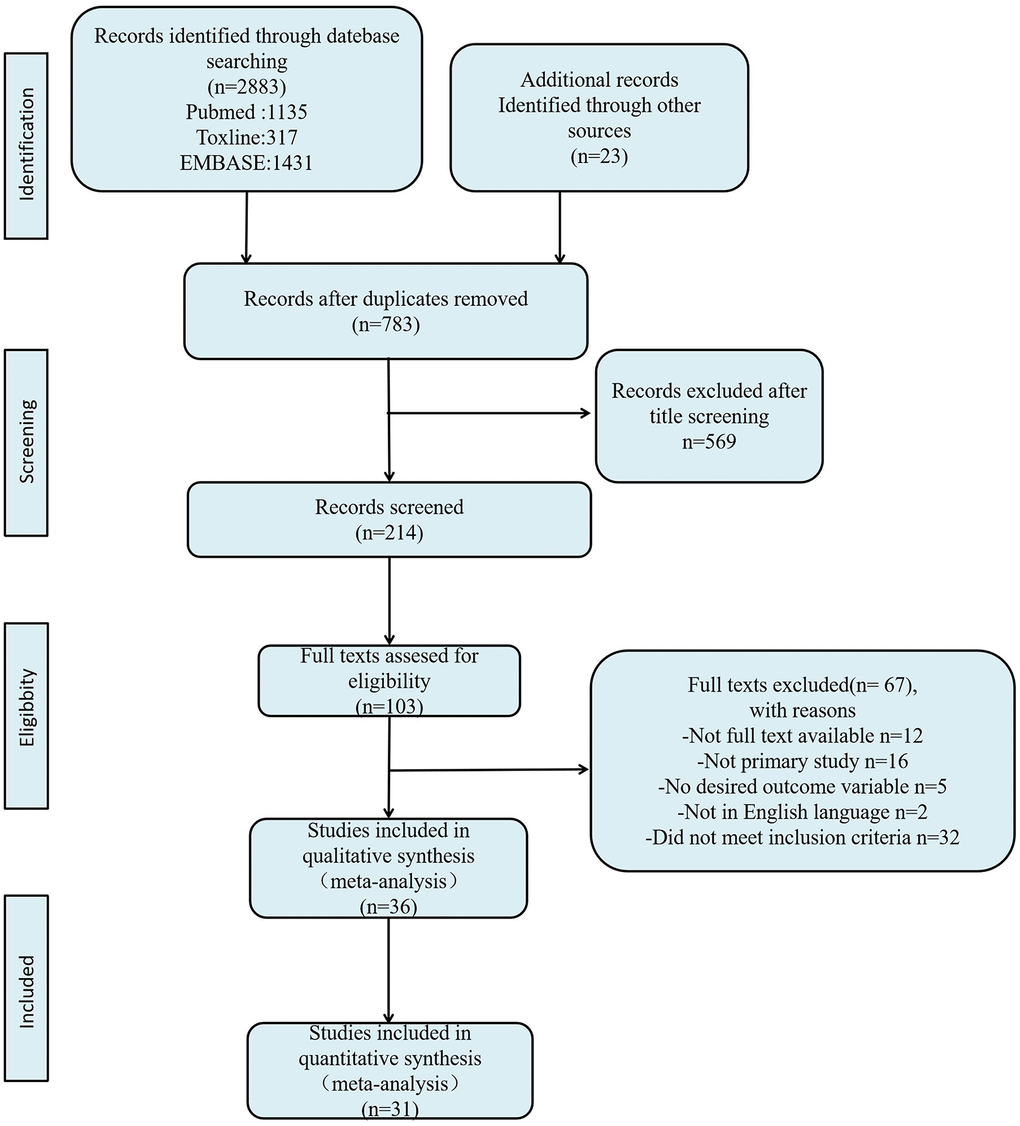
Figure 1. Flow diagram of literature search and selection process.
Table 2. Characteristics of the studies included in the meta-analysis.
| Reference | Year | Species (strain) | Dose in μg/kg bw/day | Exposure period | Exposure route | Timing of outcome measurement | Effects on male reproduction extracted in the analysis | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Dabeer et al. [16] | 2020 | Rat (Wistar) | 10000 | 180 days prebreed-PND35 | Drinking water | PND35 | Organ weight, Sperm parameters | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Shi et al. [17] | 2018 | Mice (CD-1) | 0.5/20/50 | GD11-PND1 | Oral | PND60, PND120 | Body weight, Organ weight, Sperm parameters | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Prints et al. [18] | 2018 | Rat (Sprague-Dawley) | 2.5/25/250/2500/ 25000 | Grp1:GD6-PND365; Grp2:GD6- PND21 | Gavage | PND365 | Body weight | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Spörndly-Nees et al. [19] | 2018 | Rat (F344) | 0.5/50 | GD3.5-GD22 | Drinking water | PND35, PND360 | Body weight, Organ weight | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Dere et al. [20] | 2018 | Rat (Sprague-Dawley) | 2.5/25/250/2500/25000/250000 | GD6-PND90 | Gavage | PND90 | Body weight, Organ weight | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Rahman et al. [21] | 2017 | Mice (CD-1) | 50/5000/50000 | GD7-GD14 | Gavage | PND120 | Sperm parameters | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Bernardo et al. [22] | 2015 | Rat (Sprague-Dawley) | 25/250 | GD10-GD21 | Gavage | PND21, PND180 | Body weight, Organ weight | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Rodrıguez et al. [23] | 2015 | Gerbil | 40 | GD17-24 | Oral | PND1, PND90 | Body weight, Organ weight | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hass et al. [24] | 2015 | Rat (Wistar) | 25/250/5000/50000 | GD7-PND22 | Gavage | PND90 | Body weight, Organ weight | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Delclos et al. [25] | 2014 | Rat (Sprague-Dawley) | 2.5/8/25/80/260/840/2700/100000/300000 | GD6-PND90 | Diet | PND90 | Organ weight | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Brandt et al. [8] | 2014 | Rat (Sprague-Dawley) | 25/250 | GD10-GD21 | Gavage | PND21, PND180 | Body weight, Organ weight | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Gámez et al. [26] | 2014 | Rat (Wistar) | 3 | GD1-PND21 | Drinking water | PND35 | Body weight, Organ weight | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Vilela et al. [27] | 2013 | Mice (Vesper) | 40/80/200 | GD5-PND1 | Gavage | PND70 | Sperm parameters | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Kendig et al. [28] | 2012 | Mice (CD-1) | 30/300/3000/30000/300000 | GD1-PND90 | Oral | PND90 | Organ weight, Sperm parameters | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Kobayashi et al. [29] | 2012 | Rat (Sprague-Dawley) | 330/3300/33000 | GD6-PND21 | Diet | PND35, PND90 | Body weight, Organ weight, Sperm parameters | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LaRocca et al. [30] | 2011 | Mice (C57/Bl6) | 50/1000 | GD10-GD16 | Gavage | PND56 | Body weight, Organ weight | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Salian et al. [31] | 2009a | Rat (Holtzman) | 1.2/2.4 | GD12-PND21 | Gavage | PND125 | Body weight, Organ weight, Sperm parameters | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Salian et al. [32] | 2009b | Rat (Holtzman) | 100/200/400/830/1660 | PND1-PND5 | Injection | PND135 | Sperm parameters | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Tyl et al. [33] | 2007 | Mice (CD-1) | 3/30/300/5000/50000/600000 | 40 days prebreed-PND140 | Diet | PND140 | Body weight, Organ weight, Sperm parameters | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Howdeshell et al. [34] | 2007 | Rat (Long Evans) | 2/20/200 | GD7-PND1 | Gavage | PND150 | Body weight, Organ weight, Sperm parameters | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Kato et al. [35] | 2006 | Rat (Sprague-Dawley) | 2/11/56/277/97000 | PND1-PND9 | Injection | PND10, PND35, PND150 | Body weight, Organ weight, Sperm parameters | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Ichihara et al. [36] | 2003 | Rat (F344) | 50/7500/12000 | 3 weeks prebreed-PND22 | Gavage | PND455 | Body weight, Organ weight | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Tyl et al. [7] | 2002 | Rat (Sprague-Dawley) | 1/20/300/5000/50000/500000 | 10 weeks prebreed- PND21 | Diet | PND161 | Body weight, Organ weight, Sperm parameters | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Tinwell et al. [37] | 2002 | Rat (Wistar) | 20/100/50000 | GD 6-21 | Gavage | PND90 | Body weight, Organ weight, Sperm parameters | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Yoshino et al. [38] | 2002 | Rat (F344) | 7500/12000 | 3 weeks prebreed-PND21 | Gavage | PND90 | Body weight, Organ weight, Sperm parameters | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Nagao et al. [39] | 2002 | Mice (C57BL/6N) | 2/20/200 | GD11-GD18 | Diet | PND84 | Body weight, Organ weight, Sperm parameters | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Ema et al. [40] | 2001 | Rat (Sprague-Dawley) | 0.2/2/20/200 | G1-PND21 | Diet | PND120 | Body weight, Organ weight | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Kwon et al. [41] | 2000 | Rat (Sprague-Dawley) | 3200/32000/320000 | GD11-PND20 | Oral | PND180 | Body weight, Organ weight | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cagen et al. [42] | 1999 | Mice (CF-1) | 0.2/20/200 | GD 11-17 | Oral | PND90 | Body weight, Organ weight, Sperm parameters | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Ashby et al. [43] | 1999 | Mice (CF-1) | 20-Feb | GD11-17 | Oral | PND183-187 | Body weight, Organ weight, Sperm parameters | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Vom Saal et al. [44] | 1998 | Mice (CF-1) | 20-Feb | GD11-17 | Drinking water | PND180 | Body weight, Organ weight, Sperm parameters | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Notes: PND, postnatal day; GD, gestation day. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
The dose of BPA in studies ranged from 0.1 ug/kg/day to 600 mg/kg/day, which was divided by four ascending intervals: 0-60ug/kg/day (low); 100-600ug/kg/day (medium); 1-50mg/kg/day (high); >50mg/kg/day (extremely high). For the timing of data collection, outcomes were measured between PND0 and PND455 and divided by three subgroups:
Rob evaluation
The assessment results of Rob and methodological quality were shown in Supplementary Table 2 and Figure 2, with many items receiving an “unclear” rating, resulting in an unknown Rob. For selection bias (Q1-Q3), 13 studies reported the sequence generation process (Q1); 12 studies reported on the adjustment of confounding factors and baseline characteristics of two groups (BPA group and control group) (Q2); none of study reported the allocation concealment (Q3). Regarding performance bias (Q4, Q5), the random housing and blinding of caregivers were not reported at all. On the aspect of detection bias (Q6, Q7), outcome assessor was described blinded in 7 studies (Q7), whereas no study reported the randomization of outcome measure (Q6). For attrition bias (Q8), 2 studies adequately addressed incomplete outcome data. All studies had an unclear risk of reporting bias (Q9). Besides, one study (Nagao 2002) was scored with a high risk of other bias for cesarean section (Q10). With respect of methodological quality (Q11-Q14), 19 studies reported randomization at any level of the experiment (Q11); only 8 studies reported the blinding (Q12); 13 studies stated that there was no conflict of interest (Q13); and the funding source was provided in 25 studies (Q14).
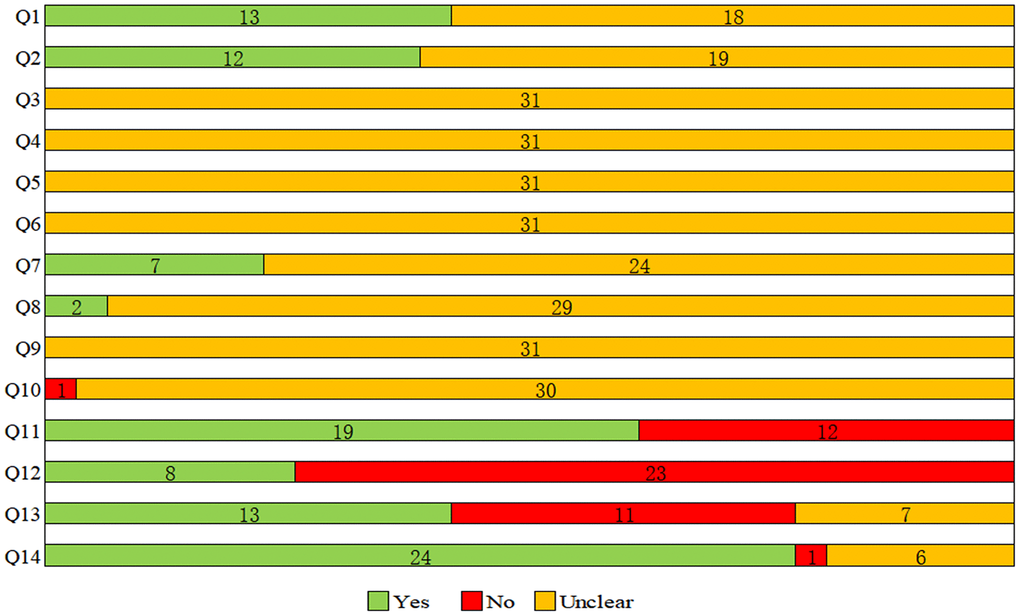
Figure 2. Results of the risk of bias and methodological quality indicators for all included studies. Notes: Q, question. Q1: Was the allocation sequence adequately generated and applied?; Q2: Were the groups similar at baseline or were they adjusted for confounders in the analysis?; Q3: Was the allocation to the different groups adequately concealed?; Q4: Were the animals randomly housed during the experiment?; Q5: Were the caregivers and/or investigators blinded from knowledge which intervention each animal received during the experiment?; Q6: Were animals selected at random for outcome assessment?; Q7: Was the outcome assessor blinded?; Q8: Were incomplete outcome data adequately addressed?; Q9: Are reports of the study free of selective outcome reporting?; Q10: Was the study apparently free of other problems that could result in high risk of bias?; Q11: Was it stated that the experiment was randomized at any level?; Q12: Was it stated that the experiment was blinded at any level?; Q13: Was it stated that there was no conflict of interest?; Q14: Was the funding source of the study provided?
BPA at low dose (0-60ug/kg/day)
Out of 31 studies, 27 studies have applied low-dose BPA intervention in one or more experimental groups. Regardless of the period, no significant difference was found with 52 comparisons investigating the low-dose BPA on body weight (SMD: 0.08; 95% Cl: -0.13, 0.29). For organ weight, it seem that the exposure of BPA at low dose may increase the weight of prostate at any period (especially in the period of
On the aspect of sperm parameters, the results revealed that the exposure of low-dose BPA exhibited a significantly decreased sperm motility (Total: SMD: -0.72; 95% Cl: -1.08, -0.35;
BPA at medium dose (100-600ug/kg/day)
In total, 100 independent comparisons in 18 studies applied medium-dose BPA intervention. As with low-dose exposure, medium-dose BPA exposure had no significant effect on body weight (SMD: 0.25; 95% Cl: -0.05, 0.26; I2 = 75.3%), yet had sufficient effect on prostate weight, especially at the period of
To be noted, at the period of PND60-PND180, BPA exposure at medium dose had a significant negative association with all the sperm parameters, which include sperm motility (SMD: -0.59; 95% Cl: -0.85, -0.32; I2 = 47.6%), daily sperm production (SMD: -0.29; 95% Cl: -0.50, -0.08; I2 = 5.4%), efficiency of sperm production (SMD: -0.45; 95% Cl: -0.66, -0.23; I2 =30.2%) and epididymal sperm count (SMD: -2.48; 95% Cl: -3.45, -1.51; I2 = 94.0%).
BPA at high dose (1-50mg/kg/day)
A total of 17 studies applied high-dose BPA intervention in one or more experimental groups. When the dose increased to a high level, the negative effect of BPA on body weight seemed to begin to show (Total: SMD: -0.23; 95% Cl: -0.39, -0.06; I2 = 27.8%;
For sperm parameters, high-dose exposure could inhibit daily sperm production (SMD: -0.28; 95% Cl: -0.47, -0.09; I2 = 41.1%), efficiency of sperm production (SMD: -0.49; 95% Cl: -0.96, -0.02; I2 = 82.7%) and epididymal sperm count (SMD: -2.43; 95% Cl: -3.27, -1.59; I2 = 90.5%), except for sperm motility (SMD: -0.11; 95% Cl: -0.47, 0.25; I2 = 71.5%).
BPA at extremely high dose (>50mg/kg/day)
Out of 31 studies, 8 studies have applied extremely high-dose BPA intervention in one or more experimental groups. The result indicated that extremely high-dose BPA exposure was significantly associated with decreased weight of body (SMD: -1.49; 95% Cl: -2.22, -0.75; I2 = 82.6%) and reproductive-related organ (Prostate weight: SMD: -4.21; 95% Cl: -5.97, -2.44; I2 = 91.1%; Testis weight; SMD: -1.92; 95% Cl: -2.61, -1.23; I2 = 81.4%; Epididymis weight; SMD: -2.16; 95% Cl: -3.47, -0.86; I2 = 95.2%), as well as negative sperm parameters (Daily sperm production; SMD: -1.90; 95% Cl: -3.27, -0.53; I2 = 93.3%; Epididymal sperm count; SMD: -3.42; 95% Cl: -3.87, -2.97; I2 = 14.8%).
Publication bias
Funnel plot (Supplementary Figure 1) and Egger’s test (Supplementary Table 4) were used to assess publication bias. The funnel plot indicates no significant publication bias, which were confirmed by Egger’s test, respectively.
Sensitivity analyses
Sensitivity analysis was utilized to detect the influence of each study by repeating the meta-analysis while omitting one study each time. The result was shown in Supplementary Table 5, and no change in direction of the association was found after sensitivity analyses.
Confidence ratings
Owing to many items of Rob were scored with “unclear”, the initial high confidence for all outcome measures was downgraded (Table 4). By reason of varying point estimates, minimal or no overlap of confidence intervals between studies, and substantial heterogeneity (I2 > 75%), the confidence rating of body weight and seminal vesicle weight were downgraded for unexplained inconsistency. The confidence rating of testis weight, epididymis weight and epididymal sperm concentration were downgraded for varying point estimates and moderate heterogeneity (I2 > 50%), whereas the sperm motility was downgraded because of varying point estimates and minimal overlap of confidence intervals between studies. All the outcome measures were not upgraded for the factors of magnitude, dose response, residual confounding and consistency species.
Table 4. Quality of the evidence of the overall effects of bisphenol A on the reproductive-related outcome measures using the office of health assessment and translation confidence rating methodology (NTP 2015).
| Outcome measure | Body of evidence (animal studies) | Factors for downgrading | Factors for upgrading | Final confidence rating | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Risk of bias | Unexplained inconsistency | Indirectness | Imprecision | Publication bias | Magnitude | Dose response | Residual confounding | Consistency species | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Body weight | Initial high confidence | ↓f | ↓↓c | – | – | –d | – | –e | – | – | Low | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Prostate weight | Initial high confidence | ↓f | – | – | – | –d | – | –e | – | – | Moderate | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Testis weight | Initial high confidence | ↓f | ↓h | – | – | –d | – | –e | – | – | Low to Moderate | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Epididymis weight | Initial high confidence | ↓f | ↓h | – | – | –d | – | –e | – | – | Low | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Seminal vesicle weight | Initial high confidence | ↓f | ↓↓c | – | – | –d | – | –e | – | – | Low | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sperm motility | Initial high confidence | ↓f | ↓g | – | – | –d | – | –e | – | – | Low to Moderate | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Daily sperm production (testis) | Initial high confidence | ↓f | – | – | – | –d | – | –e | – | – | Moderate | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Efficiency of sperm production | Initial high confidence | ↓f | – | – | – | –d | – | –e | – | – | Moderate | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Epididymal sperm concentration | Initial high confidence | ↓f | ↓h | – | – | –d | – | –e | – | – | Low to Moderate | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Note: –, no concern, or not present; ↓, serious concern; ↑, sufficient to upgrade evidence. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| a The factors “large effect magnitude” and “residual confounding” were not assessed in this study and consequently were not used to upgrade the evidence. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| b Serious concern because of many “unclear” scores and a change in direction of the association after sensitivity analyses. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| c Serious concern because of varying point estimates, minimal or no overlap of confidence intervals between studies, and substantial heterogeneity (I2 > 75%). | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| d No strongly suspected publication bias observed. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| e Indications for dose–response effects either within or across studies, but the consistency of these indications was not considered sufficient to upgrade the confidence. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| f Serious concern because of many “unclear” scores. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| g Serious concern because of varying point estimates and minimal overlap of confidence intervals between studies. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| h Serious concern because of varying point estimates and moderate heterogeneity (I2 > 50%). | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| i Serious concern because of varying point estimates. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| j No subgroup differences were estimated across species and strains. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| k Body of evidence was already downgraded for unexplained inconsistency and additional downgrading for imprecision was not considered appropriate (NTP 2015). | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Finally, based on this stringent confidence rating, the evidence of prostate weight, daily sperm production (testis) and efficiency of sperm production were rated as moderate; the evidence of testis weight, sperm motility and epididymal sperm concentration were rated as low-to-moderate; the evidence of body weight, epididymis weight and seminal vesicle weight were rated as low.
Discussion
As a common-used chemical with weak estrogenic properties, BPA may influence the developmental plasticity during early life, and injury the hormone-sensitive organs (prostate, testis, breast, ovary et al.) [45]. In fact, despite the endocrine interference effect, its prospective promotion on the carcinogenesis and reproductive damage is still controversial.
To our knowledge, this meta-analysis is the first study that focuses on the effect of BPA exposure (low, medium, high, extremely high) on reproductive-related outcomes in male rodents (
Our analysis on body weight showed that high (1-50mg/kg/day) and extremely high (>50mg/kg/day) dose of BPA could decrease the weight of body. However, when the dose was reduced to the level of low (0-60ug/kg/day) and medium (100-600ug/kg/day), this negative association became insignificance. Another meta-analysis conducted by Wassenaar et al. [46], estimating the effect of early-life BPA exposure on body weight, revealed that the dose of >50ug/kg/day might lead to a decreased body weight, which was not absolutely aligned with our result. We supposed that it may be related to the different BPA concentrations and selection bias.
For the prostate, its correlation with estrogen has been studied in vivo, in vitro, and clinically [47, 48]. Due to the limited data of enrolled studies, we only analyzed the association between BPA and prostate weight. Interestingly, the result revealed that BPA could increase prostate weight at the dose of <50mg/kg/day, yet decrease at the dose of >50mg/kg/day. This trend of bi-phasic dose responses is consistent with previous findings conducted by Huang et al. [49], which manifested that 0.01-1 nM BPA promoted cell growth, but 10-1,000 nM elicited growth inhibition. This effect may be explained by several possible mechanisms. First, relatively high-dose BPA could induce cell death for its cytotoxic effects, and then decrease the weight of prostate [50]. Second, with a different affinity of BPA and ERα or ERβ, the combination of BPA and ER (ERα and ERβ) could promote cell proliferation or inhibition [51]. Generally, ERα is considered to promote the proliferation of prostatic epithelial cells, while ERβ has an anti-proliferative effect [52]. Third, evidence showed that the interaction of BPA and androgen receptors (AR) may produce an impact on the prostate [53].
From our result, the susceptiveness of the testis, epididymis, and seminal vesicle to BPA seemed to be inferior to the prostate. But interestingly, we found that puberty and prepuberty male rodents (
The analysis of sperm parameters showed a strong correlation. Early-life BPA exposure, at extremely high level (>50mg/kg/day), is an independent factor for impaired spermatogenesis and motility at any period of life. Despite the fact that the combined result of sperm motility was not significantly changed at the dose of higher than 1mg/kg/day, the negative pooled SMD (-0.11) still showed this trend. As an AR antagonist, BPA could block the normal binding activity and its interaction between AR and endogenous androgen, and thus impaired the normal spermatogenesis [55]. Concomitantly, BPA may have a direct adverse impact on spermatogenesis by targeting Sertoli cells and interrupting the meiotic progression of germ cells [56]. Also, some experi-mental studies stated that BPA could inhibit the sperm mobility, which might be mediated by means of compromising mitochondrial functions (increase the mitochondrial ROS or reduce the high mitochondrial inner transmembrane potential) and decreasing ATP levels in spermatozoa [57, 58]. In fact, the damage to sperm parameters was observed in our pooled analysis at the dose of far below 50mg/kg/day (RfD formulated by FDA for rodents) and 4mg/kg/day (RfD formulated by EFSA for rodents), which might be considered insignificant in a single study.
For male rodents, as expected, early-life exposure to doses higher than 50mg/kg/day showed a negative association with body weight, organ weight and sperm parameters. However, when we reviewed the full data (Table 3), we found a non-monotonic association (or non-monotonic dose response curves; NMDRCs) for body weight and organ weight. The mathematical definition of NMDRCs is that the slope of curve changes from positive to negative (or vice versa) at somewhere along the range of doses examined [59]. Actually, with traditional toxicology dogma “the dose makes the poison”, people always straightly believe that there is no or little toxicity under so-called “safe” dose for most endocrine-disrupting chemical (EDC) [60]. If there is a monotonic relationship between dose and effect, the assumption that a dose below NOAEL (unobserved level of adverse reactions) is a “safe” dose seems to be reliable. Yet the NMDRCs are common in the BPA in vitro experiments [61], and thus we deem it flawed for the premise of high dose testing to extrapolate to low “safe” doses.
Furthermore, despite we strictly followed the guidelines of SYRCLE (specifically designed to evaluate animal studies) and got robust statistical evidence through this study, some limitations have been identified. First, most studies in our analysis have an unknown Rob for many items receiving an “unclear” rating in the assessments and methodological quality. In fact, it is common for most rodent studies. To rectify this situation, in the future, scholars could use some checklists to improve the quality, for example, the ARRIVE guidelines et al. [62]. Second and unavoidingly considerable heterogeneity existed in our study, which was not effectively reduced after subgroup analysis, indicating that the heterogeneity may be attributed to the diversity of experimental design and quality. Thus, the result should be interpreted with caution. Third, owing to the limitation of data, direct evidence of damage, such as pathology, was not analyzed. Consequently, if more experiments are present in the future, the results would be more accurate.
Conclusions
In conclusion, the result of our meta-analysis suggested a significant negative association between early-life BPA exposure and reproductive-related outcomes, especially at the dose of >50mg/kg/day. Meanwhile, sperm parameters seem to be more sensitive to BPA - the adverse effects occurred at any dose level. Moreover, we found that the NMDRCs of BPA for organ weight and body weight, which may challenge the existing “safe dose” theory. Consequently, we believe that, with more studies focused on the effect of BPA in the future, the understanding of BPA toxicity will be more limpid.
Materials and Methods
This meta-analysis followed the Systematic Review Centre for Laboratory Animal Experimentation (SYRCLE) [9] and PRISMA guidelines [10].
Search strategy
We searched PubMed, EMBASE, and Toxline databases from inception until December 2019 for relevant studies on the effects of developmental exposure to BPA on the reproductive system. The search strategy in PubMed, EMBASE and Toxline database included the following domains of Medical Subject Heading (MeSH) terms: “bisphenol A”, “reproductive”, “sperm” and “rodents”. These terms were combined with “AND” or “OR”. Comprehensive search strategies were shown in Supplementary Table 1. Besides, the reference lists of included studies and related comments were manually filtered for new studies that may be relevant.
Study screening
Two authors (RX and ZT) independently reviewed the title and abstract of the primary selection and then conducted a full-text screening when necessary. The inclusion criteria were as follows: (1) experimental rodent study; (2) exposure to BPA; (3) have an exposure history during gestation and/or lactation; (4) complete and interested outcome indicators and (5) outcomes measured in F1 males.
The exclusion criteria were as follows: (1) did not contain a control group; (2) non-English article; (3) not a rodent study; (4) no outcomes; (5) not early-life exposure; (6) exposure to a chemical other than BPA, (7) outcomes not measured in F1 generation and (8) disunity of administration unit (for example: ug/kg and ppm)
Data extraction
The data of bibliography (journal, year and authors), animal model (species, strain, and sex), BPA exposure (time, period, dose, and frequency), study design (number of animals, duration of follow-up, and timing of data collection), and outcome measures (body weight, prostate weight, ventral prostate (VP) weight, testis weight, epididymis weight, seminal vesicle weight, sperm motility, DSP, efficiency of sperm production, and epididymal sperm concentrations) were extracted from each study. Engauge Digitizer 4.1 software (markmitch, Boston, MA, USA) was used to extract data from figures [11].
Risk of bias (RoB)
We used SYRCLE’s RoB tool, adapted from the Cochrane RoB tool and adjusted for aspects of bias that play a specific role in animal experiments, to assess the risk of bias in the studies we included [12]. Following the instructions of SYRCLE’s RoB tool, two authors (RX and ZT) independently assessed the risk of bias, and disagreements were resolved by discussion. This RoB tool consists of 10 items, which could evaluate 6 types of bias: selection, performance, reporting, detection, attrition and other bias [12]. Moreover, we further assessed the quality of the enrolled studies from four aspects: random, blind, conflict and fund.
Statistical analysis
Stata 16.0 software (Stata Corporation, College Station, TX, USA) was used to perform analyses. All P-values were two-sided, and P < 0.05 was regarded as significant. The results of individual study were summarized. Considering the different scale of reported data and time of data collection, as well as the average value of vast differences, standardized mean differences (SMDs) was used to calculate the combined estimates. The estimates were calculated using fixed-effects or random-effects models according to the heterogeneity, which was reported using the Cochrane Q-test [13] and the inconsistency index value [14] (Higgins et al. 2003). Funnel plot and Egger’s test were used to judge the publication bias.
Confidence rating
The OHAT method (NTP 2015), which is based on the Grading of Recommendations, Assessment, Development and Evaluation (GRADE) method [15], was used to rate the quality of evidence. Upon OHAT method, the initial evidence was divided into four grades (high quality, moderate quality, low quality, and very low quality) following rating the features of experiment design: a) controlled exposure; b) exposure before outcome development; c) outcome assessment on the individual level; and d) inclusion of a comparison group. Based on such evaluation, four factors could increase the confidence rating (large magnitude of effect, dose-response relationship, residual confounding, and consistency across study designs) and five factors could decrease the confidence rating (risk of bias, unexplained inconsistency for the outcomes, indirectness or reduced applicability in the results, imprecision, and publication bias).
Supplementary Materials
Author Contributions
RX and ZT collected the data and performed the meta-analysis. RX wrote the manuscript. All the authors participated in the data analysis and approved the final version of the manuscript.
Conflicts of Interest
The author reports no conflicts of interest in this work.
Funding
This work was supported by National Natural Science Foundation (81800587).
References
- 1. Jalal N, Surendranath AR, Pathak JL, Yu S, Chung CY. Bisphenol a (BPA) the mighty and the mutagenic. Toxicol Rep. 2017; 5:76–84. https://doi.org/10.1016/j.toxrep.2017.12.013 [PubMed]
- 2. Sonavane M, Gassman NR. Bisphenol a co-exposure effects: a key factor in understanding BPA’s complex mechanism and health outcomes. Crit Rev Toxicol. 2019; 49:371–86. https://doi.org/10.1080/10408444.2019.1621263 [PubMed]
- 3. Renaud L, Huff M, da Silveira WA, Angert M, Haas M, Hardiman G. Genome-wide analysis of low dose bisphenol-a (BPA) exposure in human prostate cells. Curr Genomics. 2019; 20:260–74. https://doi.org/10.2174/1389202920666190603123040 [PubMed]
- 4. Huff J. Does exposure to bisphenol a represent a human health risk? Regul Toxicol Pharmacol. 2003; 37:407–08. https://doi.org/10.1016/s0273-2300(03)00009-6 [PubMed]
- 5. U.S. Environmental Protection Agency. 1988. Integrated Risk Information System: Bisphenol A. https://cfpub.epa.gov/ncea/iris/iris_documents/documents/subst/0356_summary.pdf.
- 6. European Food Safety Authority. 2015. Factsheet: Scientific opinion on bisphenol A. http://www.efsa.europa.eu/sites/default/files/corporate_publications/files/factsheetbpa150121.pdf.
- 7. Tyl RW, Myers CB, Marr MC, Thomas BF, Keimowitz AR, Brine DR, Veselica MM, Fail PA, Chang TY, Seely JC, Joiner RL, Butala JH, Dimond SS, et al. Three-generation reproductive toxicity study of dietary bisphenol a in CD sprague-dawley rats. Toxicol Sci. 2002; 68:121–46. https://doi.org/10.1093/toxsci/68.1.121 [PubMed]
- 8. Brandt JZ, Silveira LT, Grassi TF, Anselmo-Franci JA, Fávaro WJ, Felisbino SL, Barbisan LF, Scarano WR. Indole-3-carbinol attenuates the deleterious gestational effects of bisphenol a exposure on the prostate gland of male F1 rats. Reprod Toxicol. 2014; 43:56–66. https://doi.org/10.1016/j.reprotox.2013.11.001 [PubMed]
- 9. de Vries RBM, Hooijmans CR, Langendam MW, Leenaars M, Ritskes-Hoitinga M, Wever KE. A protocol format for the preparation, registration and publication of systematic reviews of animal intervention studies. Evid Based Preclin Med. 2015; 2:1–9. https://doi.org/10.1002/ebm2.7
- 10. Moher D, Liberati A, Tetzlaff J, Altman DG, and PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med. 2009; 6:e1000097. https://doi.org/10.1371/journal.pmed.1000097 [PubMed]
- 11. Tierney JF, Stewart LA, Ghersi D, Burdett S, Sydes MR. Practical methods for incorporating summary time-to-event data into meta-analysis. Trials. 2007; 8:16. https://doi.org/10.1186/1745-6215-8-16 [PubMed]
- 12. Hooijmans CR, Rovers MM, de Vries RB, Leenaars M, Ritskes-Hoitinga M, Langendam MW. SYRCLE’s risk of bias tool for animal studies. BMC Med Res Methodol. 2014; 14:43. https://doi.org/10.1186/1471-2288-14-43 [PubMed]
- 13. Lau J, Ioannidis JP, Schmid CH. Quantitative synthesis in systematic reviews. Ann Intern Med. 1997; 127:820–26. https://doi.org/10.7326/0003-4819-127-9-199711010-00008 [PubMed]
- 14. Higgins JP, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ. 2003; 327:557–60. https://doi.org/10.1136/bmj.327.7414.557 [PubMed]
- 15. Rooney AA, Boyles AL, Wolfe MS, Bucher JR, Thayer KA. Systematic review and evidence integration for literature-based environmental health science assessments. Environ Health Perspect. 2014; 122:711–18. https://doi.org/10.1289/ehp.1307972 [PubMed]
- 16. Dabeer S, Afjal MA, Ahmad S, Fatima M, Habib H, Parvez S, Raisuddin S. Transgenerational effect of parental obesity and chronic parental bisphenol a exposure on hormonal profile and reproductive organs of preadolescent wistar rats of F1 generation: a one-generation study. Hum Exp Toxicol. 2020; 39:59–76. https://doi.org/10.1177/0960327119873017 [PubMed]
- 17. Shi M, Sekulovski N, MacLean JA
2nd , Hayashi K. Prenatal exposure to bisphenol a analogues on male reproductive functions in mice. Toxicol Sci. 2018; 163:620–31. https://doi.org/10.1093/toxsci/kfy061 [PubMed] - 18. Prins GS, Hu WY, Xie L, Shi GB, Hu DP, Birch L, Bosland MC. Evaluation of bisphenol a (BPA) exposures on prostate stem cell homeostasis and prostate cancer risk in the NCTR-sprague-dawley rat: an NIEHS/FDA CLARITY-BPA consortium study. Environ Health Perspect. 2018; 126:117001. https://doi.org/10.1289/EHP3953 [PubMed]
- 19. Spörndly-Nees E, Boberg J, Ekstedt E, Holm L, Fakhrzadeh A, Dunder L, Kushnir MM, Lejonklou MH, Lind PM. Low-dose exposure to bisphenol a during development has limited effects on male reproduction in midpubertal and aging fischer 344 rats. Reprod Toxicol. 2018; 81:196–206. https://doi.org/10.1016/j.reprotox.2018.08.007 [PubMed]
- 20. Dere E, Anderson LM, Huse SM, Spade DJ, McDonnell-Clark E, Madnick SJ, Hall SJ, Camacho L, Lewis SM, Vanlandingham MM, Boekelheide K. Effects of continuous bisphenol a exposure from early gestation on 90 day old rat testes function and sperm molecular profiles: a CLARITY-BPA consortium study. Toxicol Appl Pharmacol. 2018; 347:1–9. https://doi.org/10.1016/j.taap.2018.03.021 [PubMed]
- 21. Rahman MS, Kwon WS, Karmakar PC, Yoon SJ, Ryu BY, Pang MG. Gestational exposure to bisphenol a affects the function and proteome profile of F1 spermatozoa in adult mice. Environ Health Perspect. 2017; 125:238–45. https://doi.org/10.1289/EHP378 [PubMed]
- 22. Bernardo BD, Brandt JZ, Grassi TF, Silveira LT, Scarano WR, Barbisan LF. Genistein reduces the noxious effects of in utero bisphenol a exposure on the rat prostate gland at weaning and in adulthood. Food Chem Toxicol. 2015; 84:64–73. https://doi.org/10.1016/j.fct.2015.07.011 [PubMed]
- 23. Rodríguez DA, de Lima RF, Campos MS, Costa JR, Biancardi MF, Marques MR, Taboga SR, Santos FC. Intrauterine exposure to bisphenol a promotes different effects in both neonatal and adult prostate of male and female gerbils (meriones unguiculatus). Environ Toxicol. 2016; 31:1740–50. https://doi.org/10.1002/tox.22176 [PubMed]
- 24. Hass U, Christiansen S, Boberg J, Rasmussen MG, Mandrup K, Axelstad M. Low-dose effect of developmental bisphenol a exposure on sperm count and behaviour in rats. Andrology. 2016; 4:594–607. https://doi.org/10.1111/andr.12176 [PubMed]
- 25. Delclos KB, Camacho L, Lewis SM, Vanlandingham MM, Latendresse JR, Olson GR, Davis KJ, Patton RE, Gamboa da Costa G, Woodling KA, Bryant MS, Chidambaram M, Trbojevich R, et al. Toxicity evaluation of bisphenol a administered by gavage to sprague dawley rats from gestation day 6 through postnatal day 90. Toxicol Sci. 2014; 139:174–97. https://doi.org/10.1093/toxsci/kfu022 [PubMed]
- 26. Gámez JM, Penalba R, Cardoso N, Ponzo O, Carbone S, Pandolfi M, Scacchi P, Reynoso R. Low dose of bisphenol a impairs the reproductive axis of prepuberal male rats. J Physiol Biochem. 2014; 70:239–46. https://doi.org/10.1007/s13105-013-0298-8 [PubMed]
- 27. Vilela J, Hartmann A, Silva EF, Cardoso T, Corcini CD, Varela-Junior AS, Martinez PE, Colares EP. Sperm impairments in adult vesper mice (calomys laucha) caused by in utero exposure to bisphenol a. Andrologia. 2014; 46:971–78. https://doi.org/10.1111/and.12182 [PubMed]
- 28. Kendig EL, Buesing DR, Christie SM, Cookman CJ, Gear RB, Hugo ER, Kasper SN, Kendziorski JA, Ungi KR, Williams K, Belcher SM. Estrogen-like disruptive effects of dietary exposure to bisphenol a or 17α-ethinyl estradiol in CD1 mice. Int J Toxicol. 2012; 31:537–50. https://doi.org/10.1177/1091581812463254 [PubMed]
- 29. Kobayashi K, Kubota H, Ohtani K, Hojo R, Miyagawa M. Lack of effects for dietary exposure of bisphenol a during in utero and lactational periods on reproductive development in rat offspring. J Toxicol Sci. 2012; 37:565–73. https://doi.org/10.2131/jts.37.565 [PubMed]
- 30. LaRocca J, Boyajian A, Brown C, Smith SD, Hixon M. Effects of in utero exposure to bisphenol a or diethylstilbestrol on the adult male reproductive system. Birth Defects Res B Dev Reprod Toxicol. 2011; 92:526–33. https://doi.org/10.1002/bdrb.20336 [PubMed]
- 31. Salian S, Doshi T, Vanage G. Neonatal exposure of male rats to bisphenol a impairs fertility and expression of sertoli cell junctional proteins in the testis. Toxicology. 2009; 265:56–67. https://doi.org/10.1016/j.tox.2009.09.012 [PubMed]
- 32. Salian S, Doshi T, Vanage G. Perinatal exposure of rats to bisphenol a affects the fertility of male offspring. Life Sci. 2009; 85:742–52. https://doi.org/10.1016/j.lfs.2009.10.004 [PubMed]
- 33. Tyl RW, Myers CB, Marr MC, Sloan CS, Castillo NP, Veselica MM, Seely JC, Dimond SS, Van Miller JP, Shiotsuka RN, Beyer D, Hentges SG, Waechter JM
Jr . Two-generation reproductive toxicity study of dietary bisphenol a in CD-1 (Swiss) mice. Toxicol Sci. 2008; 104:362–84. https://doi.org/10.1093/toxsci/kfn084 [PubMed] - 34. Howdeshell KL, Furr J, Lambright CR, Wilson VS, Ryan BC, Gray LE
Jr . Gestational and lactational exposure to ethinyl estradiol, but not bisphenol A, decreases androgen-dependent reproductive organ weights and epididymal sperm abundance in the male long evans hooded rat. Toxicol Sci. 2008; 102:371–82. https://doi.org/10.1093/toxsci/kfm306 [PubMed] - 35. Kato H, Furuhashi T, Tanaka M, Katsu Y, Watanabe H, Ohta Y, Iguchi T. Effects of bisphenol a given neonatally on reproductive functions of male rats. Reprod Toxicol. 2006; 22:20–29. https://doi.org/10.1016/j.reprotox.2005.10.003 [PubMed]
- 36. Ichihara T, Yoshino H, Imai N, Tsutsumi T, Kawabe M, Tamano S, Inaguma S, Suzuki S, Shirai T. Lack of carcinogenic risk in the prostate with transplacental and lactational exposure to bisphenol a in rats. J Toxicol Sci. 2003; 28:165–71. https://doi.org/10.2131/jts.28.165 [PubMed]
- 37. Tinwell H, Haseman J, Lefevre PA, Wallis N, Ashby J. Normal sexual development of two strains of rat exposed in utero to low doses of bisphenol a. Toxicol Sci. 2002; 68:339–48. https://doi.org/10.1093/toxsci/68.2.339 [PubMed]
- 38. Yoshino H, Ichihara T, Kawabe M, Imai N, Hagiwara A, Asamoto M, Shirai T. Lack of significant alteration in the prostate or testis of F344 rat offspring after transplacental and lactational exposure to bisphenol a. J Toxicol Sci. 2002; 27:433–39. https://doi.org/10.2131/jts.27.433 [PubMed]
- 39. Nagao T, Saito Y, Usumi K, Yoshimura S, Ono H. Low-dose bisphenol a does not affect reproductive organs in estrogen-sensitive C57BL/6N mice exposed at the sexually mature, juvenile, or embryonic stage. Reprod Toxicol. 2002; 16:123–30. https://doi.org/10.1016/s0890-6238(02)00003-5 [PubMed]
- 40. Ema M, Fujii S, Furukawa M, Kiguchi M, Ikka T, Harazono A. Rat two-generation reproductive toxicity study of bisphenol a. Reprod Toxicol. 2001; 15:505–23. https://doi.org/10.1016/s0890-6238(01)00160-5 [PubMed]
- 41. Kwon S, Stedman DB, Elswick BA, Cattley RC, Welsch F. Pubertal development and reproductive functions of Crl:CD BR sprague-dawley rats exposed to bisphenol a during prenatal and postnatal development. Toxicol Sci. 2000; 55:399–406. https://doi.org/10.1093/toxsci/55.2.399 [PubMed]
- 42. Cagen SZ, Waechter JM
Jr , Dimond SS, Breslin WJ, Butala JH, Jekat FW, Joiner RL, Shiotsuka RN, Veenstra GE, Harris LR. Normal reproductive organ development in CF-1 mice following prenatal exposure to bisphenol a. Toxicol Sci. 1999; 50:36–44. https://doi.org/10.1093/toxsci/50.1.36 [PubMed] - 43. Ashby J, Tinwell H, Haseman J. Lack of effects for low dose levels of bisphenol a and diethylstilbestrol on the prostate gland of CF1 mice exposed in utero. Regul Toxicol Pharmacol. 1999; 30:156–66. https://doi.org/10.1006/rtph.1999.1317 [PubMed]
- 44. vom Saal FS, Cooke PS, Buchanan DL, Palanza P, Thayer KA, Nagel SC, Parmigiani S, Welshons WV. A physiologically based approach to the study of bisphenol a and other estrogenic chemicals on the size of reproductive organs, daily sperm production, and behavior. Toxicol Ind Health. 1998; 14:239–60. https://doi.org/10.1177/074823379801400115 [PubMed]
- 45. Seachrist DD, Bonk KW, Ho SM, Prins GS, Soto AM, Keri RA. A review of the carcinogenic potential of bisphenol a. Reprod Toxicol. 2016; 59:167–82. https://doi.org/10.1016/j.reprotox.2015.09.006 [PubMed]
- 46. Wassenaar PN, Trasande L, Legler J. Systematic review and meta-analysis of early-life exposure to bisphenol a and obesity-related outcomes in rodents. Environ Health Perspect. 2017; 125:106001. https://doi.org/10.1289/EHP1233 [PubMed]
- 47. Tarapore P, Ying J, Ouyang B, Burke B, Bracken B, Ho SM. Exposure to bisphenol a correlates with early-onset prostate cancer and promotes centrosome amplification and anchorage-independent growth in vitro. PLoS One. 2014; 9:e90332. https://doi.org/10.1371/journal.pone.0090332 [PubMed]
- 48. Hu WY, Shi GB, Lam HM, Hu DP, Ho SM, Madueke IC, Kajdacsy-Balla A, Prins GS. Estrogen-initiated transformation of prostate epithelium derived from normal human prostate stem-progenitor cells. Endocrinology. 2011; 152:2150–63. https://doi.org/10.1210/en.2010-1377 [PubMed]
- 49. Huang D, Wu J, Su X, Yan H, Sun Z. Effects of low dose of bisphenol a on the proliferation and mechanism of primary cultured prostate epithelial cells in rodents. Oncol Lett. 2017; 14:2635–42. https://doi.org/10.3892/ol.2017.6469 [PubMed]
- 50. Iida H, Maehara K, Doiguchi M, Mōri T, Yamada F. Bisphenol a-induced apoptosis of cultured rat sertoli cells. Reprod Toxicol. 2003; 17:457–64. https://doi.org/10.1016/s0890-6238(03)00034-0 [PubMed]
- 51. Vandenberg LN, Maffini MV, Sonnenschein C, Rubin BS, Soto AM. Bisphenol-a and the great divide: a review of controversies in the field of endocrine disruption. Endocr Rev. 2009; 30:75–95. https://doi.org/10.1210/er.2008-0021 [PubMed]
- 52. Taylor JA, Richter CA, Ruhlen RL, vom Saal FS. Estrogenic environmental chemicals and drugs: mechanisms for effects on the developing male urogenital system. J Steroid Biochem Mol Biol. 2011; 127:83–95. https://doi.org/10.1016/j.jsbmb.2011.07.005 [PubMed]
- 53. Hess-Wilson JK, Webb SL, Daly HK, Leung YK, Boldison J, Comstock CE, Sartor MA, Ho SM, Knudsen KE. Unique bisphenol a transcriptome in prostate cancer: novel effects on ERbeta expression that correspond to androgen receptor mutation status. Environ Health Perspect. 2007; 115:1646–53. https://doi.org/10.1289/ehp.10283 [PubMed]
- 54. Wadia PR, Vandenberg LN, Schaeberle CM, Rubin BS, Sonnenschein C, Soto AM. Perinatal bisphenol a exposure increases estrogen sensitivity of the mammary gland in diverse mouse strains. Environ Health Perspect. 2007; 115:592–98. https://doi.org/10.1289/ehp.9640 [PubMed]
- 55. Wetherill YB, Akingbemi BT, Kanno J, McLachlan JA, Nadal A, Sonnenschein C, Watson CS, Zoeller RT, Belcher SM. In vitro molecular mechanisms of bisphenol a action. Reprod Toxicol. 2007; 24:178–98. https://doi.org/10.1016/j.reprotox.2007.05.010 [PubMed]
- 56. Goldstein KM, Seyler DE, Durand P, Perrard MH, Baker TK. Use of a rat ex-vivo testis culture method to assess toxicity of select known male reproductive toxicants. Reprod Toxicol. 2016; 60:92–103. https://doi.org/10.1016/j.reprotox.2016.01.003 [PubMed]
- 57. Minamiyama Y, Ichikawa H, Takemura S, Kusunoki H, Naito Y, Yoshikawa T. Generation of reactive oxygen species in sperms of rats as an earlier marker for evaluating the toxicity of endocrine-disrupting chemicals. Free Radic Res. 2010; 44:1398–406. https://doi.org/10.3109/10715762.2010.510523 [PubMed]
- 58. Rahman MS, Kwon WS, Lee JS, Yoon SJ, Ryu BY, Pang MG. Bisphenol-a affects male fertility via fertility-related proteins in spermatozoa. Sci Rep. 2015; 5:9169. https://doi.org/10.1038/srep09169 [PubMed]
- 59. Kohn MC, Melnick RL. Biochemical origins of the non-monotonic receptor-mediated dose-response. J Mol Endocrinol. 2002; 29:113–23. https://doi.org/10.1677/jme.0.0290113 [PubMed]
- 60. Myers JP, Zoeller RT, vom Saal FS. A clash of old and new scientific concepts in toxicity, with important implications for public health. Environ Health Perspect. 2009; 117:1652–55. https://doi.org/10.1289/ehp.0900887 [PubMed]
- 61. Vandenberg LN. Non-monotonic dose responses in studies of endocrine disrupting chemicals: bisphenol a as a case study. Dose Response. 2013; 12:259–76. https://doi.org/10.2203/dose-response.13-020.Vandenberg [PubMed]
- 62. Kilkenny C, Browne WJ, Cuthill IC, Emerson M, Altman DG. Improving bioscience research reporting: the ARRIVE guidelines for reporting animal research. PLoS Biol. 2010; 8:e1000412. https://doi.org/10.1371/journal.pbio.1000412 [PubMed]


